Advanced Molybdenum-Copper Catalysis for Scalable 3-Trifluoromethyl-1,2,4-Triazole Production
Advanced Molybdenum-Copper Catalysis for Scalable 3-Trifluoromethyl-1,2,4-Triazole Production
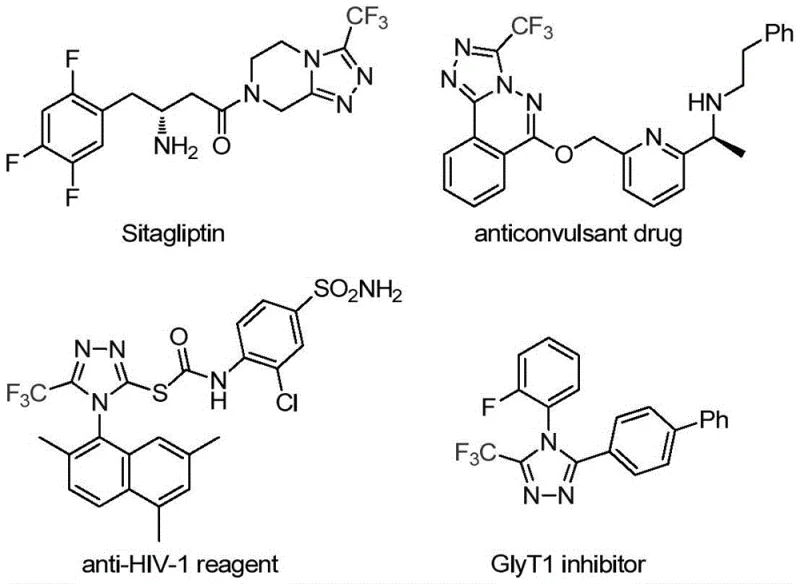
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are pivotal for enhancing metabolic stability and bioavailability. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current synthetic routes. This technology leverages a dual catalytic system involving molybdenum hexacarbonyl and cuprous acetate to facilitate a highly efficient cycloaddition reaction. The significance of this development cannot be overstated, as the 1,2,4-triazole scaffold is a privileged structure found in numerous blockbuster drugs, including antidiabetic agents like Sitagliptin and antiviral compounds. By enabling the direct assembly of these complex architectures from readily available trifluoroethylimidoyl chloride and functionalized isonitriles, this invention offers a streamlined pathway that aligns perfectly with the demands of modern green chemistry and industrial scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with synthetic challenges that hinder efficient commercial production. Traditional literature reports predominantly rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These classical approaches often suffer from harsh reaction conditions, requiring extreme temperatures or strong acidic/basic environments that can degrade sensitive functional groups. Furthermore, alternative multi-component reactions utilizing diazonium salts or trifluorodiazoethane introduce significant safety hazards due to the explosive nature of diazo compounds, making them unsuitable for large-scale manufacturing. The limited scope of substrates in these older methods also restricts the ability to generate diverse libraries of analogs for structure-activity relationship studies, thereby slowing down the drug discovery process considerably.
The Novel Approach
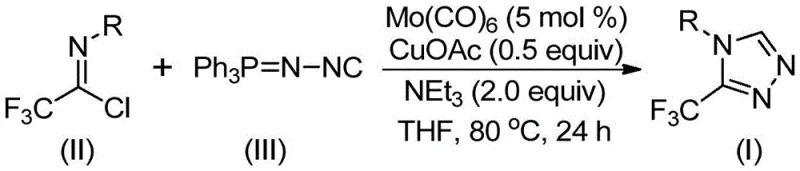
In stark contrast, the novel methodology disclosed in the patent utilizes a transition metal co-catalyzed strategy that operates under remarkably mild conditions. By employing trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as starting materials, the reaction proceeds smoothly in common organic solvents like THF at moderate temperatures ranging from 70°C to 90°C. This approach eliminates the need for hazardous diazo reagents and avoids the use of expensive or toxic noble metals often seen in cross-coupling reactions. The reaction mechanism involves the activation of the isonitrile by molybdenum hexacarbonyl followed by a copper-promoted [3+2] cycloaddition, leading to the formation of the five-membered triazole ring with high atom economy. This strategic shift not only simplifies the operational procedure but also significantly enhances the safety profile of the synthesis, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into Mo/Cu Co-catalyzed Cycloaddition
The success of this transformation lies in the synergistic interaction between the molybdenum and copper catalysts, which orchestrate the bond-forming events with precision. Mechanistically, the molybdenum hexacarbonyl serves as a crucial activator for the functionalized isonitrile, forming a transient metal-isocyanide complex that increases the nucleophilicity of the carbon center. Simultaneously, the cuprous acetate facilitates the coordination and activation of the trifluoroethylimidoyl chloride, promoting the initial attack of the isonitrile species. This cooperative catalysis lowers the activation energy barrier for the [3+2] cycloaddition step, allowing the reaction to proceed efficiently even with sterically hindered or electronically diverse substrates. The subsequent elimination of triphenylphosphine oxide, driven by the presence of water or molecular sieves in the system, ensures the irreversible formation of the aromatic triazole ring, driving the equilibrium towards the desired product.
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid-base cyclizations. The specificity of the metal-ligand interactions minimizes side reactions such as polymerization of the isonitrile or hydrolysis of the imidoyl chloride, which are common pitfalls in non-catalyzed processes. The use of molecular sieves further aids in scavenging trace moisture that could otherwise lead to the decomposition of sensitive intermediates, thereby ensuring a cleaner crude reaction profile. This high level of chemoselectivity is paramount for R&D directors focused on purity specifications, as it reduces the burden on downstream purification steps and ensures that the final high-purity pharmaceutical intermediates meet stringent regulatory standards without extensive recrystallization or chromatographic efforts.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and reproducibility. The protocol dictates a specific molar ratio where the functionalized isonitrile is used in slight excess relative to the trifluoroethylimidoyl chloride to drive the reaction to completion. The catalytic loading is kept low, with molybdenum hexacarbonyl at approximately 5 mol % and cuprous acetate at 0.5 equivalents, balancing cost efficiency with catalytic activity. The detailed standardized synthesis steps, including precise addition sequences and workup procedures, are outlined below to guide technical teams in replicating these results effectively.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the crude sample with silica gel, and purify via column chromatography to isolate the final 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route presents a compelling value proposition centered around cost stability and operational reliability. The primary advantage stems from the utilization of commodity chemicals; both trifluoroethylimidoyl chloride and the requisite isonitriles are derived from widely available industrial feedstocks, insulating the supply chain from the volatility associated with exotic or proprietary reagents. This accessibility ensures consistent raw material availability, reducing the risk of production delays caused by sourcing bottlenecks. Furthermore, the elimination of hazardous diazo compounds removes the need for specialized explosion-proof infrastructure, allowing for production in standard chemical manufacturing facilities and thereby lowering capital expenditure requirements for scale-up.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the replacement of expensive noble metal catalysts with abundant and inexpensive copper and molybdenum salts. Additionally, the mild reaction conditions translate directly into lower energy consumption, as the process does not require cryogenic cooling or extreme heating, resulting in substantial cost savings in utility usage. The high reaction efficiency and selectivity minimize waste generation, reducing the costs associated with solvent recovery and waste disposal, which collectively contribute to a more lean and cost-effective manufacturing model for complex heterocyclic intermediates.
- Enhanced Supply Chain Reliability: By relying on a robust catalytic system that tolerates a wide range of functional groups, manufacturers can source diverse substrates without needing to qualify new synthetic routes for each analog. This flexibility allows for rapid response to changing market demands or custom synthesis requests, significantly reducing lead time for high-purity pharmaceutical intermediates. The simplicity of the post-treatment process, involving basic filtration and chromatography, ensures faster turnaround times from reaction completion to final product release, enhancing overall supply chain agility and responsiveness to client needs.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be effective at the gram level with clear potential for kilogram-to-ton scale-up, supported by the use of stable catalysts and safe solvents like THF. The absence of toxic heavy metals and explosive reagents simplifies environmental compliance and regulatory approval processes, facilitating smoother technology transfer from R&D to commercial production. This alignment with green chemistry principles not only mitigates environmental risk but also future-proofs the manufacturing process against increasingly stringent global regulations on chemical safety and sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology, providing clarity for potential partners evaluating its feasibility for their specific applications. These insights are derived directly from the experimental data and embodiments described in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The optimal conditions involve using THF as the solvent at temperatures between 70°C and 90°C for 18 to 30 hours, utilizing a catalytic system of Mo(CO)6 and CuOAc with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates broad substrate tolerance, successfully accommodating various substituents on the aryl ring including methyl, methoxy, fluoro, chloro, and nitro groups without significant loss in efficiency.
Q: Is this process suitable for large-scale manufacturing?
A: The patent explicitly states that the method can be expanded to gram-level reactions and potentially further, owing to the use of cheap, commercially available starting materials and simple post-treatment procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Mo/Cu co-catalyzed synthesis in accelerating the development of next-generation therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate delivered meets the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced chemistry for your pipeline projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and reduce your overall cost of goods sold.
