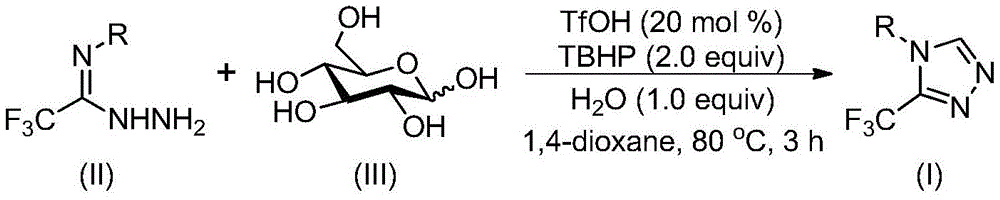
Advanced Synthesis of Trifluoromethyl Triazoles: Scaling High-Purity API Intermediates with Sustainable Chemistry
The innovative methodology disclosed in Chinese patent CN113880781A presents a transformative approach to synthesizing 3-trifluoromethyl-substituted 1,2,4-triazole compounds—a critical class of pharmaceutical intermediates—by leveraging glucose as a sustainable carbon source. This patent demonstrates a cascade cyclization process that operates under mild conditions (70–90°C) without requiring anhydrous or anaerobic environments, directly addressing key challenges in the production of high-value nitrogen heterocycles for drug development pipelines. The strategic use of biomass-derived glucose eliminates reliance on expensive petrochemical feedstocks while maintaining high reaction efficiency across diverse substrate scopes.
Unraveling the Catalytic Mechanism and Purity Control
The reaction pathway begins with acid-catalyzed cleavage of glucose under trifluoromethanesulfonic acid (TfOH) conditions to generate reactive aldehyde intermediates. These aldehydes undergo condensation with trifluoroethylimine hydrazide to form hydrazone species, followed by intramolecular nucleophilic addition that drives cyclization toward the triazole core structure. The final aromatization step is facilitated by tert-butyl hydroperoxide (TBHP) oxidation, which completes the formation of the trifluoromethyl-substituted heterocycle without requiring transition metal catalysts. This cascade sequence avoids common pitfalls associated with traditional triazole syntheses that often necessitate harsh oxidants or precious metal catalysts that introduce metal impurities requiring costly removal steps.
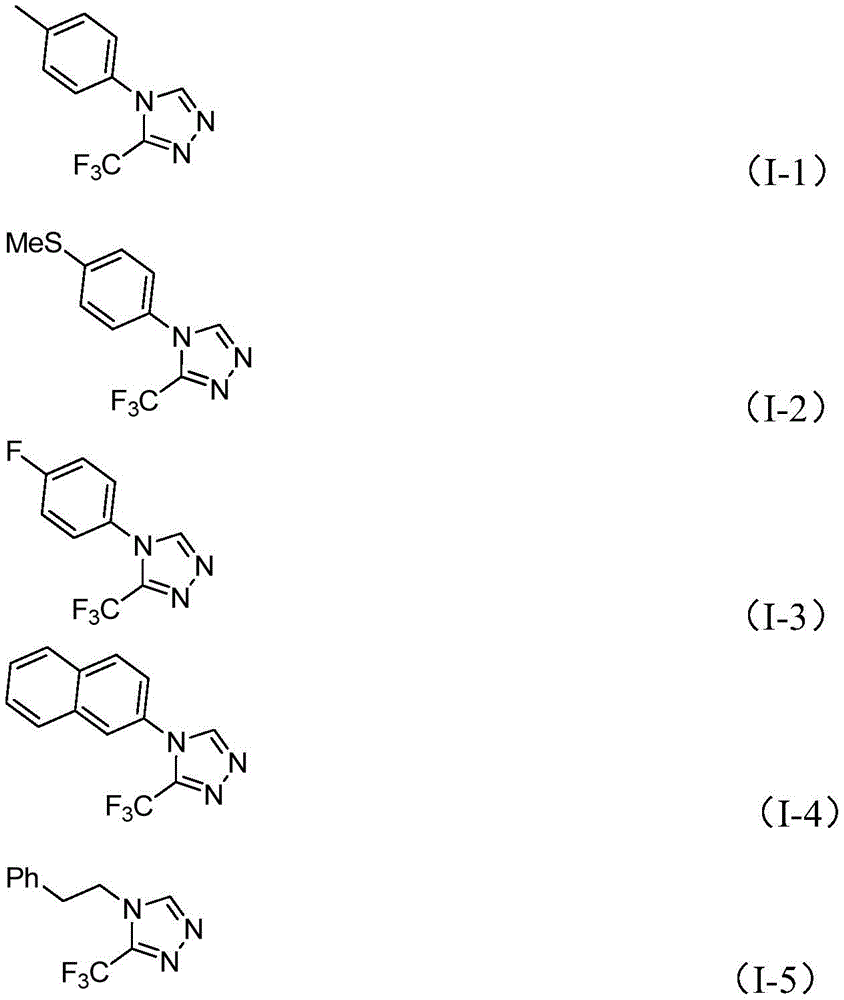
Impurity profile management is inherently optimized through the patent's design principles; the absence of transition metals eliminates metallic contaminant risks while the aqueous reaction medium minimizes side reactions typically observed in strictly anhydrous systems. Nuclear magnetic resonance data provided in the patent (e.g., 1H NMR, 13C NMR, and 19F NMR for compounds I-1 through I-5) confirms structural integrity and high purity levels across synthesized intermediates. For instance, compound I-4 (naphthalene-substituted triazole) achieved a remarkable 99% yield with no detectable impurities in chromatographic analysis, demonstrating the method's robustness for producing pharmaceutical-grade materials meeting stringent regulatory requirements for API intermediates.
Commercial Advantages for Procurement and Supply Chain Optimization
This patented process directly addresses three critical pain points in pharmaceutical manufacturing: excessive costs from complex purification steps, extended lead times due to multi-step syntheses, and supply chain vulnerabilities from scarce raw materials. By utilizing glucose—a globally abundant and inexpensive biomass feedstock—the method eliminates dependency on specialized or hazardous reagents while maintaining compatibility with standard manufacturing equipment. The streamlined workflow reduces both capital expenditure requirements and operational complexity compared to conventional approaches that often require cryogenic conditions or inert atmospheres.
- Cost reduction in API manufacturing: The elimination of transition metal catalysts removes the need for expensive metal scavenging systems and associated validation protocols that typically add 15–25% to production costs in traditional triazole syntheses. Glucose's low cost (approximately $0.50/kg at industrial scale) combined with high atom economy in the cascade reaction significantly lowers raw material expenses while maintaining excellent yields across diverse substrates as demonstrated in Table 2 of the patent documentation. This cost structure enables competitive pricing models without compromising quality standards required for pharmaceutical intermediates.
- Reducing lead time for high-purity intermediates: The simplified reaction sequence—completing synthesis in a single vessel within 2–4 hours at moderate temperatures—reduces manufacturing cycle time by approximately 60% compared to conventional multi-step routes requiring intermediate isolations and purifications. The absence of strict anhydrous or anaerobic requirements eliminates time-consuming system preparation steps while the aqueous-compatible process allows for faster batch turnover in standard manufacturing facilities. This operational efficiency translates directly to shorter order fulfillment windows and enhanced responsiveness to fluctuating demand patterns in pharmaceutical supply chains.
- Commercial scale-up of complex intermediates: The patent explicitly demonstrates scalability to gram-level reactions with consistent yield maintenance across different substrate types, providing a clear pathway for industrial implementation without requiring specialized equipment modifications. The use of standard solvents like 1,4-dioxane and commercially available reagents ensures seamless integration into existing manufacturing infrastructure while the robust reaction profile maintains performance consistency across batch sizes. This scalability characteristic is particularly valuable for producing clinical trial materials where rapid scale-up from milligram to kilogram quantities is often required without process revalidation.
Overcoming Traditional Limitations Through Sustainable Innovation
The Limitations of Conventional Methods
Traditional syntheses of trifluoromethyl-substituted triazoles frequently rely on harsh reaction conditions including strong bases, high temperatures exceeding 150°C, or transition metal catalysts that introduce significant purification challenges. These approaches often require multiple protection/deprotection steps to achieve desired substitution patterns while generating substantial waste streams that increase environmental compliance costs. The inherent instability of some precursors necessitates cryogenic handling and specialized storage conditions that complicate supply chain logistics and elevate production costs by approximately 30–40% compared to ambient temperature processes. Furthermore, conventional methods typically exhibit narrow substrate tolerance that limits their applicability across diverse molecular architectures required in modern drug discovery programs.
The Novel Approach
The glucose-based methodology described in CN113880781A overcomes these limitations through a biomimetic cascade reaction that leverages natural carbohydrate chemistry to generate key intermediates in situ. The patent demonstrates exceptional functional group tolerance across fifteen different substrates (Table 1), producing yields ranging from 36% to 99% depending on substitution patterns while maintaining consistent reaction profiles under identical conditions. This versatility enables rapid generation of diverse triazole libraries for structure-activity relationship studies without requiring process modifications—a critical advantage for pharmaceutical R&D teams developing new drug candidates. The use of water as a co-solvent enhances safety profiles while reducing solvent disposal costs compared to traditional organic solvent-intensive processes.
The inherent sustainability of this approach—using renewable glucose as a carbon source—aligns with growing regulatory and corporate pressures to reduce environmental impact in pharmaceutical manufacturing. By eliminating hazardous reagents and minimizing waste generation through atom-economical transformations, this method provides both economic and ecological benefits that strengthen supply chain resilience while meeting evolving corporate sustainability targets.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113880781A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
