Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles: Technical Breakthroughs and Commercial Scalability
Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles: Technical Breakthroughs and Commercial Scalability
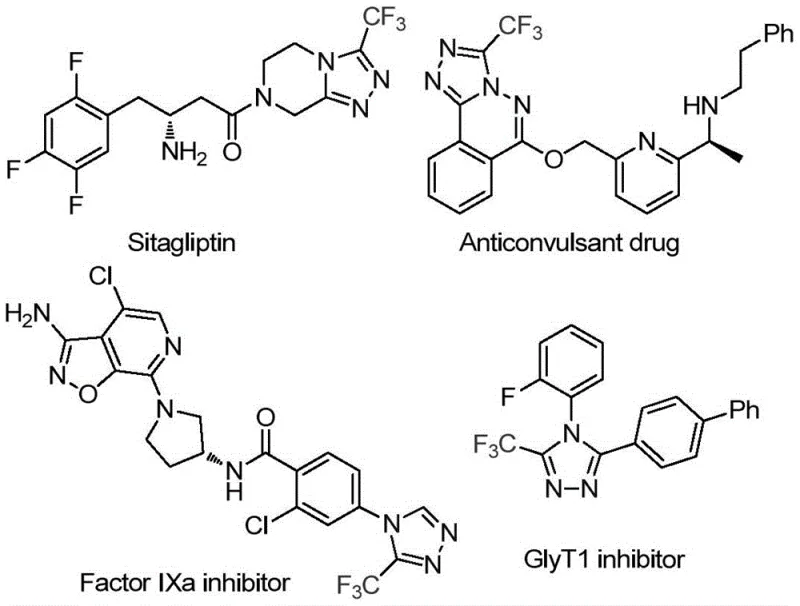
The pharmaceutical and agrochemical industries continuously seek robust methodologies for constructing fluorinated heterocyclic scaffolds, particularly 1,2,4-triazoles, due to their profound impact on metabolic stability and bioactivity. Patent CN114920707B introduces a transformative preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds that leverages the ubiquitous solvent DMF as a critical carbon source. This innovation addresses long-standing challenges in heterocyclic synthesis by utilizing a molecular iodine-promoted tandem cyclization that operates efficiently under air atmosphere. The significance of this chemical motif is underscored by its presence in high-value therapeutic agents, ranging from antidiabetic medications to potent enzyme inhibitors, highlighting the critical need for reliable pharmaceutical intermediate supplier networks capable of delivering these complex structures at scale.
The strategic value of this patent lies in its ability to streamline the supply chain for high-purity triazole compounds. By integrating the carbon source directly into the solvent system, the process minimizes the number of discrete reagent additions and reduces the overall chemical footprint of the reaction. For R&D Directors evaluating new routes, this represents a significant opportunity to enhance process mass intensity (PMI) metrics while maintaining rigorous purity standards. The method's compatibility with diverse functional groups ensures that it can be adapted for the commercial scale-up of complex polymer additives or specialty chemical derivatives, providing a versatile platform for downstream application development in both medicinal chemistry and material science sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing trifluoromethyl-substituted triazoles often rely on pre-functionalized C1 synthons or harsh cyclization conditions that necessitate stringent exclusion of moisture and oxygen. These conventional approaches frequently involve multiple synthetic steps, requiring the isolation of unstable intermediates which increases production time and introduces potential yield losses at each stage. Furthermore, the reliance on specialized reagents for introducing the trifluoromethyl group can lead to substantial cost reduction barriers, as these materials are often subject to volatile market pricing and complex logistics. The need for inert atmosphere handling also imposes significant capital expenditure on reactor infrastructure, making it difficult for manufacturers to achieve cost reduction in fine chemical manufacturing without compromising on safety or quality control protocols.
The Novel Approach
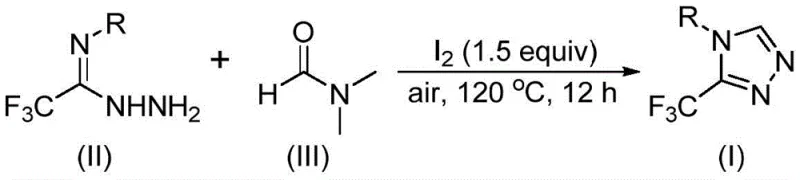
In stark contrast, the methodology disclosed in CN114920707B utilizes a direct tandem cyclization strategy where dimethylformamide (DMF) acts simultaneously as the reaction medium and the carbon donor. This dual functionality eliminates the need for external C1 sources, thereby simplifying the reaction setup and reducing the overall inventory of hazardous chemicals required on site. The use of molecular iodine as a promoter facilitates the activation of the DMF molecule under relatively mild thermal conditions, enabling the reaction to proceed efficiently in the presence of air. This operational simplicity translates directly into enhanced supply chain reliability, as the process is less susceptible to failures caused by equipment leaks or environmental fluctuations, ensuring consistent batch-to-batch quality for high-purity OLED material or pharmaceutical intermediate production.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation offers fascinating insights into the reactivity of amide solvents under oxidative conditions. The reaction initiates with the activation of DMF by molecular iodine, which generates a reactive electrophilic species capable of engaging with the nucleophilic hydrazide moiety of the starting material. Depending on the specific reaction trajectory, either the formyl group or the N-methyl group of DMF can serve as the carbon source for the triazole ring construction. When the formyl group participates, it undergoes condensation with the hydrazide to form a hydrazone intermediate, which subsequently cyclizes with the elimination of dimethylamine. Alternatively, the N-methyl pathway involves the formation of an amine salt followed by nucleophilic addition and oxidative aromatization, showcasing the versatility of this chemical system in generating diverse structural analogues through subtle modifications in reaction parameters.
From an impurity control perspective, this mechanism is particularly advantageous because the byproducts generated, such as dimethylamine or iodine salts, are generally volatile or water-soluble, facilitating their removal during the workup phase. This inherent cleanliness of the reaction profile reduces the burden on downstream purification processes, allowing for the attainment of stringent purity specifications with minimal effort. For quality assurance teams, understanding this mechanistic nuance is crucial for establishing robust in-process controls that monitor the consumption of iodine and the formation of the triazole core. The ability to tune the electronic properties of the substrate without disrupting the core cyclization mechanism ensures that reducing lead time for high-purity intermediates does not come at the expense of chemical integrity or regulatory compliance.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing manufacturing workflows, requiring only standard glassware or stainless steel reactors capable of withstanding moderate temperatures. The procedure begins with the precise charging of trifluoroethyliminohydrazide and molecular iodine into the reaction vessel, followed by the addition of DMF which serves as both solvent and reactant in significant excess to drive the equilibrium towards product formation. The mixture is then heated to a controlled temperature range of 110-130°C and maintained under stirring for a period of 10 to 15 hours, allowing the tandem cyclization to reach completion without the need for complex monitoring equipment. Detailed standardized synthesis steps see the guide below.
- Combine trifluoroethyliminohydrazide, molecular iodine, and excess DMF solvent in a reaction vessel under standard air atmosphere conditions.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for a duration of 10 to 15 hours to ensure complete conversion.
- Perform post-treatment involving filtration, washing, and silica gel column chromatography to isolate the high-purity 3-trifluoromethyl-1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary driver of value is the utilization of DMF, a commodity chemical that is readily available globally at stable price points, effectively decoupling the production cost from the volatility associated with specialized fluorinating agents. This stability allows for more accurate long-term budgeting and contract negotiation, ensuring that cost reduction in electronic chemical manufacturing or pharmaceutical production is sustainable over the product lifecycle. Additionally, the elimination of inert gas requirements reduces utility consumption and simplifies the safety protocols required for plant operation, further contributing to overall operational expenditure savings.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by consolidating the solvent and reagent functions into a single chemical entity, thereby reducing the total volume of materials purchased and handled. The avoidance of expensive transition metal catalysts or exotic ligands means that the raw material bill of materials is drastically simplified, leading to substantial cost savings per kilogram of active ingredient produced. Furthermore, the mild reaction conditions minimize energy consumption compared to high-pressure or cryogenic alternatives, enhancing the overall energy efficiency of the manufacturing plant while maintaining high throughput capabilities.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved as the key reagents, including iodine and DMF, are produced by multiple suppliers worldwide, mitigating the risk of single-source bottlenecks. The robustness of the reaction under air atmosphere means that production schedules are less vulnerable to disruptions caused by nitrogen supply failures or glovebox maintenance issues. This resilience ensures consistent delivery timelines for clients relying on just-in-time inventory models, reinforcing the reputation of the manufacturer as a dependable partner for critical API intermediate supply chains.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, which typically involves filtration and standard chromatography, facilitates easy scale-up from laboratory benchtop to multi-ton commercial production without encountering non-linear engineering challenges. The waste stream generated is primarily composed of organic solvents and inorganic salts that can be managed through established recovery and treatment protocols, aligning with increasingly stringent environmental regulations. This compliance readiness reduces the regulatory burden on the manufacturing site and ensures that the production process remains viable in jurisdictions with strict ecological oversight.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and optimization of this trifluoromethyl triazole synthesis platform. These answers are derived directly from the experimental data and scope defined within the patent documentation, providing a factual basis for feasibility assessments. Understanding these nuances helps technical teams evaluate the fit of this technology within their specific portfolio requirements and identify potential areas for process intensification or customization.
Q: What are the primary advantages of using DMF as a carbon source in this synthesis?
A: Using DMF serves a dual purpose as both the reaction solvent and the carbon synthon, which drastically simplifies the reagent list, reduces raw material costs, and eliminates the need for specialized C1 building blocks that are often expensive or hazardous to handle.
Q: Does this reaction require strict anhydrous or anaerobic conditions?
A: No, one of the significant technical breakthroughs of this method is its robustness under air atmosphere without the need for anhydrous conditions, which lowers operational complexity and infrastructure costs for commercial scale-up.
Q: What is the substrate scope for the R-group in the trifluoroethyliminohydrazide starting material?
A: The method demonstrates broad substrate tolerance, successfully accommodating various substituted aryl groups including those with electron-donating or electron-withdrawing substituents such as halogens, alkyl, alkoxy, and trifluoromethyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient heterocyclic synthesis plays in accelerating drug discovery and commercialization timelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and compliant. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole meets the exacting standards required by global regulatory bodies. Our infrastructure is designed to handle complex fluorinated chemistries safely and efficiently, providing a secure foundation for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this DMF-based methodology for your specific targets. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence and our proven track record in delivering high-quality pharmaceutical intermediates.
