Advanced Catalytic Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
Advanced Catalytic Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
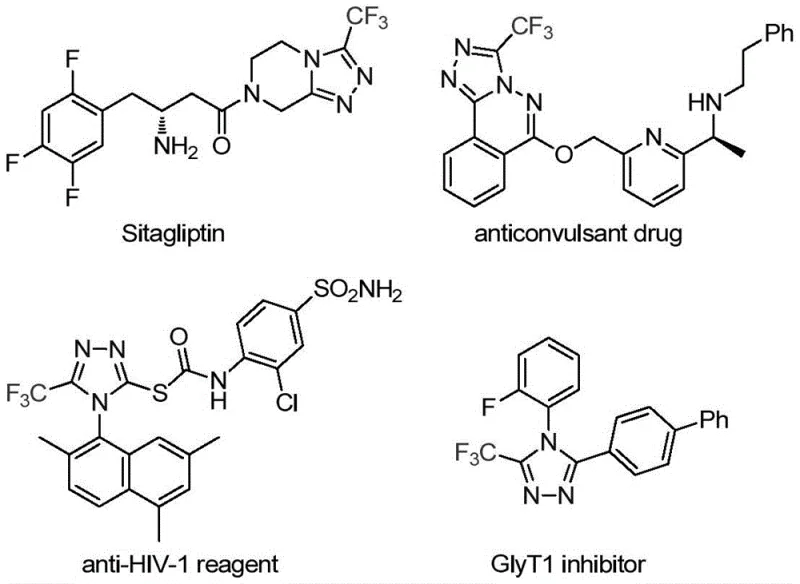
The landscape of modern pharmaceutical synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and versatile methodologies to construct complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN113307778A, which discloses a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These nitrogen-containing heterocycles are not merely academic curiosities; they are foundational structures in numerous bioactive molecules, including prominent drugs like Sitagliptin, Deferasirox, and Maraviroc. The introduction of a trifluoromethyl group into these frameworks is particularly strategic, as it significantly enhances key physicochemical properties such as metabolic stability, lipophilicity, and bioavailability, thereby optimizing the pharmacokinetic profile of the final active pharmaceutical ingredient (API).
This patented technology represents a paradigm shift from conventional synthetic routes, offering a robust pathway for the construction of polysubstituted 1,2,4-triazoles. By leveraging a synergistic co-catalytic system involving molybdenum and copper, the method achieves high reaction efficiency under remarkably mild conditions. For R&D directors and process chemists seeking reliable pharmaceutical intermediate suppliers, this development signals a new standard for accessing high-purity triazole derivatives with reduced operational complexity and enhanced safety profiles compared to legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant challenges that hinder large-scale manufacturing and rapid library generation for drug discovery. Traditional literature reports primarily rely on the cyclization reaction of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These pathways often necessitate harsh reaction conditions, multiple synthetic steps, and the use of sensitive reagents that complicate purification and lower overall yields. Furthermore, alternative approaches involving copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane introduce severe safety hazards due to the explosive nature of diazo compounds, posing unacceptable risks in a commercial production environment.
Another prevalent method involves the cyclization of trifluoroethylimidoyl chloride with aldehyde hydrazones and hydrazides. While effective in some contexts, these routes frequently suffer from limited substrate scope and poor atom economy. Most existing developed methods focus on obtaining fully substituted 1,2,4-triazoles, leaving a gap in the efficient synthesis of 3,4-disubstituted variants which are crucial for specific biological activities. The reliance on expensive catalysts, toxic solvents, or difficult-to-handle precursors in these conventional protocols creates substantial bottlenecks in cost reduction in API manufacturing, forcing procurement teams to seek more sustainable and economically viable alternatives.
The Novel Approach
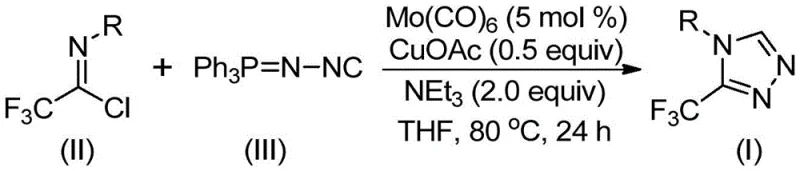
In stark contrast to these legacy techniques, the invention disclosed in patent CN113307778A introduces a streamlined, one-pot synthesis strategy that utilizes cheap and easily obtainable functionalized isonitriles and trifluoroethylimidoyl chloride as starting raw materials. The core innovation lies in the employment of a molybdenum and copper co-catalyzed cycloaddition reaction, which proceeds smoothly at temperatures between 70°C and 90°C. This mild thermal requirement not only reduces energy consumption but also minimizes the formation of thermal degradation byproducts, thereby simplifying the downstream purification process. The reaction system is remarkably tolerant, allowing for the incorporation of diverse functional groups without the need for extensive protecting group strategies.
The operational simplicity of this novel approach cannot be overstated. The reaction mixture typically comprises molybdenum hexacarbonyl, cuprous acetate, triethylamine, and molecular sieves in an organic solvent such as THF. Post-reaction processing is straightforward, involving filtration followed by standard column chromatography to isolate the target 3-trifluoromethyl-substituted 1,2,4-triazole compound in high purity. This method effectively bridges the gap between laboratory-scale discovery and industrial application, offering a scalable solution that addresses the critical need for commercial scale-up of complex pharmaceutical intermediates. By eliminating the need for hazardous diazonium salts and harsh cyclization conditions, this technology provides a safer, greener, and more cost-effective route for producing high-value heterocyclic building blocks.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this synthetic methodology is rooted in the sophisticated interplay between the molybdenum and copper catalysts, which orchestrate a precise sequence of bond-forming events. Mechanistically, the reaction is believed to initiate with the activation of the functionalized isonitrile by molybdenum hexacarbonyl, forming a reactive metal-isocyanide complex. This activation step is crucial as it enhances the nucleophilicity of the isonitrile carbon, priming it for subsequent attack. Concurrently, the cuprous acetate acts as a promoter for the [3+2] cycloaddition reaction between the activated isonitrile species and the trifluoroethylimidoyl chloride. This concerted cycloaddition leads to the formation of a five-membered ring intermediate, establishing the core 1,2,4-triazole skeleton with high regioselectivity.
Following the ring closure, the reaction pathway involves the elimination of triphenylphosphine oxide, a byproduct derived from the functionalized isonitrile precursor (Ph3P=N-NC), under the influence of water present in the system or generated during the process. This elimination step drives the equilibrium towards the formation of the final 3-trifluoromethyl-substituted 1,2,4-triazole product. The choice of triethylamine as a base further facilitates this transformation by neutralizing acidic byproducts and maintaining the optimal pH for catalyst turnover. Understanding this mechanistic nuance is vital for process optimization, as it highlights the importance of maintaining strict stoichiometric ratios—preferably 1:1.5 for the imidoyl chloride to isonitrile—to maximize yield and minimize impurity profiles.
From an impurity control perspective, the use of molecular sieves in the reaction mixture plays a pivotal role in managing water content, which can otherwise lead to hydrolysis of the sensitive imidoyl chloride starting material. The robustness of the Mo/Cu catalytic system ensures that even with substrates bearing electron-withdrawing groups like nitro or halogens, the reaction proceeds with commendable efficiency. This mechanistic resilience translates directly to product quality, ensuring that the resulting high-purity pharmaceutical intermediates meet the stringent specifications required for downstream drug synthesis. The ability to tune the electronic properties of the R-group on the aryl ring without compromising reaction efficiency underscores the versatility of this catalytic manifold.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, precise adherence to the optimized reaction parameters is essential. The process begins with the careful weighing and mixing of molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), and triethylamine (2.0 equiv) along with the specific trifluoroethylimidoyl chloride and functionalized isonitrile substrates. The reaction is conducted in anhydrous THF, heated to 80°C, and stirred for approximately 24 hours to ensure complete conversion. Detailed standardized operating procedures regarding reagent addition rates, temperature ramping, and quenching protocols are critical for reproducibility and safety.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the crude sample with silica gel, and perform column chromatography purification to isolate the pure 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling strategic advantages that extend beyond mere technical feasibility. The primary driver for adoption is the significant potential for cost optimization throughout the manufacturing value chain. By utilizing starting materials that are commercially available and inexpensive, such as trifluoroethylimidoyl chloride and functionalized isonitriles, the raw material cost base is substantially lowered compared to routes requiring specialized or custom-synthesized precursors. Furthermore, the elimination of hazardous reagents like diazonium salts reduces the regulatory burden and insurance costs associated with handling explosive materials, contributing to a leaner operational expenditure model.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming from both material and operational efficiencies. The use of relatively inexpensive catalysts like cuprous acetate and molybdenum hexacarbonyl, which are used in catalytic or sub-stoichiometric amounts, avoids the high costs associated with precious metal catalysts like palladium or rhodium. Additionally, the mild reaction conditions (70-90°C) imply lower energy consumption for heating and cooling cycles compared to cryogenic or high-temperature processes. The simplified workup procedure, which avoids complex extraction or distillation steps in favor of direct filtration and chromatography, reduces solvent usage and labor hours, leading to substantial overall cost savings in the production of these valuable intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the high availability of the requisite raw materials. Since the key reagents are commodity chemicals or easily synthesized from common precursors like aromatic amines and trifluoroacetic acid, the risk of supply disruption is minimized. This reliability is crucial for maintaining continuous production schedules for critical API intermediates. Moreover, the robustness of the reaction across a wide range of substrates means that a single manufacturing line can be adapted to produce various derivatives simply by changing the starting amine, providing flexibility to respond to shifting market demands without significant capital investment in new equipment or process validation.
- Scalability and Environmental Compliance: The patent explicitly notes that the method can be expanded to gram-level reactions with high efficiency, indicating a clear path towards kilogram and ton-scale production. The use of THF as a solvent, while requiring recovery systems, is well-established in the industry, and the absence of heavy metal waste streams (other than trace Mo and Cu which are easier to manage than Pd) simplifies wastewater treatment. The high atom economy of the cycloaddition reaction minimizes waste generation, aligning with green chemistry principles and helping manufacturers meet increasingly stringent environmental regulations, thus avoiding potential fines and reputational damage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when integrating this methodology into their supply chains.
Q: What are the primary advantages of the Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: The novel method utilizes cheap and readily available starting materials like functionalized isonitriles and trifluoroethylimidoyl chloride under mild conditions (70-90°C). Unlike traditional methods requiring harsh cyclization or unstable diazonium salts, this approach offers high reaction efficiency, simple operation, and excellent functional group tolerance, making it ideal for complex API intermediate manufacturing.
Q: Can this synthesis method be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the patent explicitly demonstrates that the reaction can be expanded to gram-level scales with high efficiency. The use of stable catalysts like molybdenum hexacarbonyl and cuprous acetate, combined with standard post-processing techniques like column chromatography, suggests strong potential for commercial scale-up in industrial settings.
Q: What types of substituents are tolerated on the aryl ring of the final triazole product?
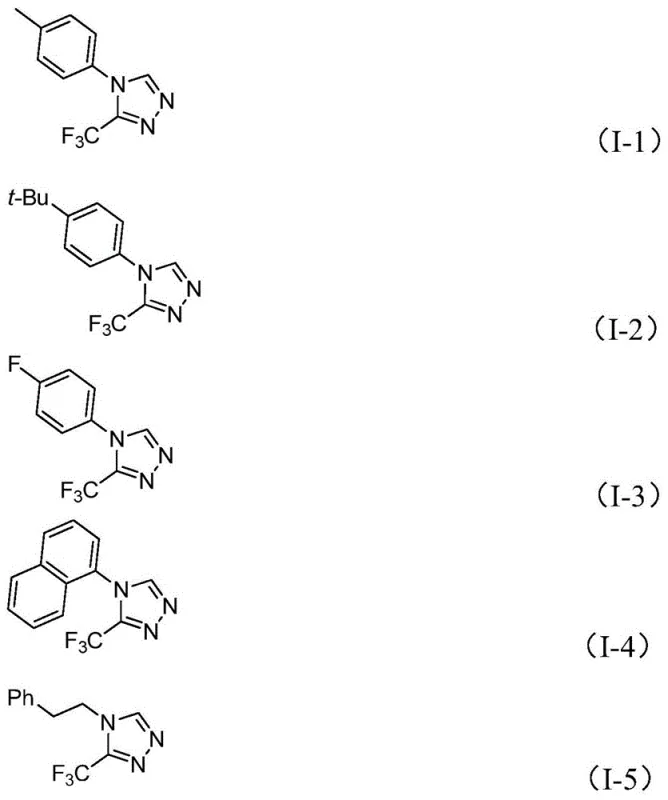
A: The method exhibits broad substrate scope, successfully accommodating various substituents on the aryl ring including alkyl groups (methyl, ethyl, t-butyl), alkoxy groups (methoxy), halogens (fluoro, chloro), and electron-withdrawing groups like nitro and methylthio. This versatility allows for the design of diverse molecular frameworks for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methods like the Mo/Cu co-catalyzed synthesis of 3-trifluoromethyl-1,2,4-triazoles. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative patent technologies into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of intermediate meets the highest quality standards required for global pharmaceutical applications.
We invite you to collaborate with us to leverage this efficient synthesis route for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data for our triazole derivatives and discuss route feasibility assessments that can accelerate your drug development timeline while optimizing your overall manufacturing budget.
