Advanced Synthesis of 2-Alkynyl Indoles via Primary Amine Guidance for Commercial Scale-Up
Advanced Synthesis of 2-Alkynyl Indoles via Primary Amine Guidance for Commercial Scale-Up
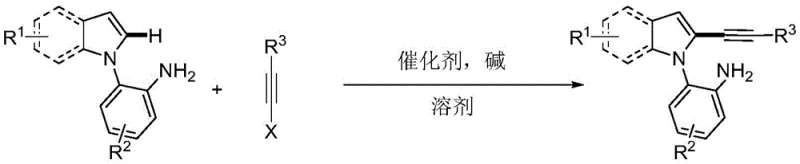
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds with high precision and efficiency. A significant breakthrough in this domain is detailed in patent CN108864164B, which discloses a novel synthesis method for primary amine-guided 2-alkynyl indole compounds. This technology represents a paradigm shift from traditional multi-step functionalization strategies to a direct, catalytic cross-coupling approach. By leveraging the unique coordinating ability of a primary amine group, this method achieves exceptional regioselectivity at the C2 position of the indole ring, a site that is historically challenging to functionalize due to the inherent electronic preference for the C3 position. For R&D directors and process chemists, this patent offers a compelling solution for generating highly functionalized indole derivatives, which are critical building blocks in the development of bioactive molecules, agrochemicals, and advanced materials.
The core innovation lies in the use of simple 2-(1H-indol-1-yl)aniline compounds reacting with alkyne halides under palladium catalysis. What makes this particularly attractive for industrial application is the reaction's tolerance for a wide range of substrates and its compatibility with green solvents. The process eliminates the need for pre-functionalization steps such as halogenation, thereby streamlining the synthetic route. As a reliable pharmaceutical intermediate supplier, understanding such technological advancements allows us to offer clients superior routes that minimize waste and maximize yield. The ability to synthesize these complex structures in a single pot under relatively mild conditions underscores the potential for significant cost reduction in API manufacturing and process intensification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of functionalized indole derivatives has relied heavily on classical cross-coupling reactions that require pre-halogenated substrates. This conventional approach typically involves an initial C-H halogenation step to introduce a leaving group, followed by a metal-catalyzed coupling. However, this strategy suffers from several inherent drawbacks that impact both cost and efficiency. Firstly, indole compounds possess higher electron cloud density at the C3 position compared to the C2 position, making selective halogenation at C2 difficult without protecting groups or harsh conditions. Secondly, the multi-step nature of these traditional routes leads to lower overall atom economy and increased generation of chemical waste. Furthermore, the use of stoichiometric amounts of halogenating agents and the subsequent purification of halogenated intermediates add substantial complexity and cost to the supply chain. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The method described in patent CN108864164B overcomes these historical challenges by employing a direct C-H activation strategy guided by a primary amine. Instead of pre-installing a halogen, the reaction utilizes the amino group on the adjacent aniline ring to direct the palladium catalyst specifically to the C2-H bond of the indole. This direct alkynylation bypasses the need for prior halogenation, effectively shortening the synthetic sequence. The reaction proceeds smoothly in the presence of a palladium salt catalyst and a base, such as cesium pivalate, often using water as the solvent. This shift not only simplifies the operation but also drastically improves the safety profile of the process by avoiding hazardous halogenating reagents. The result is a streamlined pathway to 2-alkynyl indoles that offers superior regioselectivity and operational simplicity, making it an ideal candidate for the commercial scale-up of complex polymer additives or drug candidates.
Mechanistic Insights into Pd-Catalyzed C-H Alkynylation
The success of this synthesis relies on a sophisticated catalytic cycle driven by the coordination between the palladium center and the primary amine directing group. Mechanistically, the reaction initiates with the coordination of the palladium catalyst to the nitrogen atom of the primary amine on the 2-(1H-indol-1-yl)aniline substrate. This coordination facilitates the formation of a thermodynamically stable six-membered palladacycle intermediate. This cyclometalation step is crucial as it activates the proximal C2-H bond of the indole ring while simultaneously blocking the more reactive C3 position, thereby ensuring exclusive C2 selectivity. Following the formation of this organopalladium intermediate, the alkyne halide undergoes oxidative addition to the palladium center. This step introduces the alkynyl moiety into the coordination sphere of the metal.
The final stage of the catalytic cycle involves reductive elimination, which releases the desired 2-alkynyl indole product and regenerates the active palladium catalyst for the next turnover. This mechanism explains the broad substrate scope observed in the patent examples, where various substituents on both the indole and aniline rings are well-tolerated. From an impurity control perspective, this mechanism is highly advantageous. By enforcing a specific geometric arrangement through the six-membered ring intermediate, the formation of C3-alkynylated byproducts is effectively suppressed. This intrinsic selectivity reduces the burden on downstream purification processes, ensuring that the final high-purity OLED material or pharmaceutical intermediate meets stringent quality specifications with minimal effort. The use of triisopropylsilyl (TIPS) protected alkynes further enhances stability and handling during the reaction.
How to Synthesize 2-Alkynyl Indole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The patent provides detailed embodiments demonstrating the robustness of the method across various substrates. The general procedure involves charging a reactor with the indole-aniline substrate, the alkyne halide (typically a TIPS-protected bromoalkyne), a palladium source like palladium tetranitrate tetrafluoroborate, and a cesium base. The choice of solvent is flexible, with water or water-toluene mixtures proving effective, highlighting the green chemistry credentials of the process. Heating the mixture to temperatures between 80°C and 110°C drives the reaction to completion within 12 to 24 hours. Detailed standardized synthesis steps are provided in the guide below.
- Charge a reactor with 2-(1H-indol-1-yl)aniline substrate, alkyne halide (e.g., TIPS-protected bromoalkyne), palladium salt catalyst, and cesium base.
- Add water or a water-toluene mixture as the solvent and heat the reaction mixture to 80-110°C with stirring for 12-24 hours.
- Upon completion, cool to room temperature, extract with ethyl acetate, dry over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible strategic advantages beyond mere technical novelty. The shift towards a direct C-H functionalization route fundamentally alters the cost structure of producing 2-alkynyl indoles. By eliminating the need for separate halogenation steps and the associated reagents, the process reduces the total number of unit operations required. This consolidation of steps leads to a significant reduction in labor, energy consumption, and equipment usage time. Furthermore, the ability to use water as a primary solvent removes the dependency on expensive and regulated organic solvents, simplifying waste management and lowering environmental compliance costs. These factors collectively contribute to a more resilient and cost-effective supply chain for critical chemical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by its high atom economy and simplified workflow. Traditional routes often suffer from yield losses at each discrete step; by condensing the synthesis into a single catalytic event, the overall yield is preserved and improved. The use of inexpensive palladium salts and readily available cesium bases, combined with the elimination of costly halogenating agents, results in substantial raw material savings. Additionally, the simplified workup procedure, which involves standard extraction and chromatography, reduces the operational overhead associated with complex purification protocols. This efficiency translates directly into a lower cost of goods sold (COGS) for the final product.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized or hazardous reagents. This method utilizes 2-(1H-indol-1-yl)aniline compounds and alkyne halides, which are commercially available and easy to source in bulk quantities. The robustness of the reaction conditions, which tolerate a wide range of functional groups including halogens, methoxy, and cyano groups, ensures consistent production even with slight variations in feedstock quality. This flexibility reduces the risk of production stoppages and ensures a steady flow of materials for downstream applications, making it a reliable choice for long-term sourcing strategies.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces safety and environmental challenges, particularly when dealing with exothermic halogenation reactions. This aqueous-based palladium-catalyzed method operates under mild thermal conditions (80-110°C), which are easily manageable in large-scale reactors. The use of water as a solvent significantly reduces the fire hazard and VOC emissions associated with traditional organic synthesis. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing facility. The process is inherently safer and more sustainable, supporting corporate sustainability goals while maintaining high production throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2-alkynyl indole compounds using this primary amine-guided strategy. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature. Understanding these details helps stakeholders make informed decisions about process adoption and material sourcing. The answers reflect the practical realities of implementing this chemistry in a GMP or industrial setting.
Q: How does this method achieve high regioselectivity at the C2 position?
A: The method utilizes the primary amine group on the aniline ring as a directing group. This coordinates with the palladium catalyst to form a stable six-membered ring intermediate, effectively blocking the more reactive C3 position and guiding the alkynylation specifically to the C2 position of the indole ring.
Q: What are the environmental benefits of this synthesis route?
A: Unlike traditional methods requiring toxic organic solvents, this process can utilize water as the primary solvent. This significantly reduces volatile organic compound (VOC) emissions, lowers waste disposal costs, and aligns with green chemistry principles for sustainable API manufacturing.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under mild temperatures (80-110°C) and uses commercially available, inexpensive starting materials. The simple workup procedure involving extraction and chromatography makes it highly scalable for commercial production of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkynyl Indole Supplier
The technological potential of primary amine-guided C-H activation is immense, offering a pathway to complex heterocycles that were previously difficult to access economically. At NINGBO INNO PHARMCHEM, we specialize in translating such innovative academic and patent technologies into viable commercial processes. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of 2-alkynyl indole delivered meets the exacting standards required by the global pharmaceutical and agrochemical industries. We are committed to bridging the gap between novel synthetic methodology and reliable industrial supply.
We invite R&D directors and procurement specialists to collaborate with us to evaluate the feasibility of this route for your specific projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this green chemistry approach can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable and cost-efficient supply of high-value indole intermediates for your next generation of products.
