Advanced Synthesis of 2-Alkynyl Indoles via Primary Amine Guidance for Commercial Scale-Up
Advanced Synthesis of 2-Alkynyl Indoles via Primary Amine Guidance for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds with high precision and minimal environmental impact. A significant breakthrough in this domain is detailed in Chinese Patent CN108864164B, which discloses a novel synthesis method for primary amine-guided 2-alkynyl indole compounds. This technology represents a paradigm shift from traditional multi-step functionalization strategies to a direct, atom-economical cross-coupling approach. By leveraging the unique coordination capabilities of a primary amine directing group, this process achieves exceptional regioselectivity at the C2 position of the indole ring, a site that is notoriously difficult to functionalize directly due to the inherent electronic preference for the C3 position. The methodology employs common palladium salts as catalysts and, crucially, utilizes water as the reaction medium, aligning perfectly with the principles of green chemistry while maintaining high yields and broad substrate tolerance.
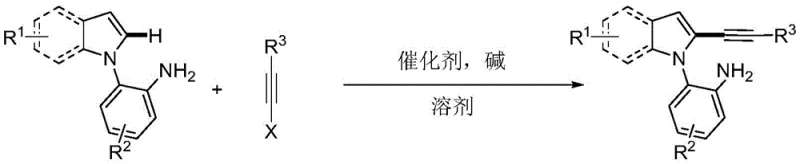
For R&D directors and process chemists evaluating new routes for API intermediates, the implications of this patent are profound. The ability to bypass pre-halogenation steps not only streamlines the synthetic pathway but also eliminates the generation of stoichiometric halogenated waste. The reaction conditions are mild, operating between 80°C and 110°C, which ensures compatibility with a wide range of sensitive functional groups including fluorine, chlorine, methoxy, and cyano substituents. This versatility allows for the rapid generation of diverse chemical libraries essential for drug discovery programs. Furthermore, the use of triisopropylsilyl-protected alkynes provides a stable handle for subsequent downstream transformations, such as Sonogashira couplings or click chemistry, making these intermediates highly valuable building blocks for medicinal chemistry campaigns targeting kinase inhibitors or GPCR modulators.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 2-substituted indoles has been plagued by significant regioselectivity challenges. The indole nucleus possesses a higher electron cloud density at the C3 position compared to the C2 position, making the C3 site inherently more reactive towards electrophilic attack. Consequently, conventional synthetic strategies often rely on indirect routes, such as the initial halogenation of the indole ring followed by protection-deprotection sequences to block the C3 position, or the use of specialized starting materials like 2-lithioindoles which require cryogenic conditions and strict anhydrous environments. These traditional approaches are not only operationally hazardous due to the use of strong bases and low temperatures but also suffer from poor atom economy. The necessity for multiple isolation and purification steps increases the overall production timeline and generates substantial quantities of chemical waste, thereby inflating the cost of goods sold (COGS) and complicating the supply chain for large-scale manufacturing.
The Novel Approach
In stark contrast, the methodology described in patent CN108864164B offers a streamlined solution by utilizing a primary amine as an intramolecular directing group. This strategy fundamentally alters the reaction trajectory by coordinating the palladium catalyst directly to the nitrogen atom of the aniline moiety attached to the indole nitrogen. This coordination facilitates the formation of a thermodynamically favorable six-membered palladacycle intermediate, which effectively overrides the natural electronic bias of the indole ring. As a result, the C-H activation occurs selectively at the C2 position, enabling direct cross-coupling with alkyne halides. This direct approach eliminates the need for pre-functionalization, reduces the number of synthetic steps, and operates under relatively mild thermal conditions. The transition from multi-step, hazard-prone protocols to a single-pot, water-mediated reaction signifies a major advancement in process efficiency and safety profiles for industrial applications.
Mechanistic Insights into Pd-Catalyzed C-H Activation
The mechanistic elegance of this transformation lies in the synergistic interaction between the palladium catalyst and the primary amine directing group. Upon introduction of the base, the 2-(1H-indol-1-yl)aniline substrate coordinates with the palladium species, likely undergoing a concerted metalation-deprotonation (CMD) pathway. The amino group acts as a Lewis base, anchoring the metal center in close proximity to the C2-H bond. This spatial arrangement lowers the activation energy required for C-H bond cleavage, leading to the formation of a cyclic organopalladium intermediate. Subsequent oxidative addition of the alkyne halide to this palladium(II) species generates a high-valent palladium(IV) or palladium(II) complex, depending on the specific oxidation state cycle involved. Finally, reductive elimination releases the desired 2-alkynyl indole product and regenerates the active palladium catalyst, completing the catalytic cycle. This mechanism ensures high turnover numbers and minimizes the loading of expensive precious metal catalysts required for the reaction.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or non-directed pathways. The rigid geometry of the six-membered palladacycle intermediate imposes strict steric constraints that prevent reaction at the C3 position or on the benzene ring of the indole moiety. This intrinsic selectivity drastically reduces the formation of regioisomeric byproducts, which are often difficult to separate and can pose significant risks in pharmaceutical manufacturing regarding genotoxicity or biological activity. Furthermore, the use of water as a solvent suppresses many side reactions associated with organic solvents, such as solvent participation or degradation of sensitive intermediates. The result is a cleaner reaction profile with a simplified impurity spectrum, facilitating easier downstream purification and ensuring that the final product meets the stringent purity specifications required for clinical grade materials.
How to Synthesize 2-Alkynyl Indole Efficiently
The practical implementation of this synthesis route is designed for scalability and operational simplicity, making it highly attractive for contract development and manufacturing organizations (CDMOs). The process begins with the charging of readily available starting materials into a standard reactor, avoiding the need for specialized equipment or inert atmosphere gloveboxes for the setup. The reaction proceeds efficiently in an aqueous environment, which simplifies heat transfer and temperature control during exothermic phases. Following the reaction period, the workup procedure involves standard liquid-liquid extraction techniques using ethyl acetate, a solvent that is easily recovered and recycled. The crude product is then subjected to column chromatography using petroleum ether and ethyl acetate mixtures, yielding the pure 2-alkynyl indole derivative. For detailed standardized operating procedures and specific parameter optimization, please refer to the technical guide below.
- Charge a reactor with 2-(1H-indol-1-yl)aniline substrate, alkyne halide, palladium salt catalyst, alkali base, and water solvent.
- Stir the reaction mixture at a temperature range of 80-110°C for a duration of 12 to 24 hours to ensure complete conversion.
- Upon completion, cool to room temperature, extract with ethyl acetate, dry the organic phase, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method translates into tangible strategic benefits beyond mere technical novelty. The shift towards a water-based solvent system represents a significant reduction in the procurement costs associated with volatile organic solvents and their subsequent disposal. Traditional organic solvents often require expensive incineration or specialized recycling infrastructure, whereas aqueous waste streams can be treated using standard wastewater facilities, leading to substantial operational expenditure (OPEX) savings. Additionally, the high atom economy of the direct C-H activation means that less raw material is wasted in the form of byproducts, maximizing the yield per kilogram of input. This efficiency is critical when sourcing expensive precursors like functionalized indoles or silyl-protected alkynes, as it ensures that every gram of purchased material contributes maximally to the final output.
- Cost Reduction in Manufacturing: The elimination of pre-halogenation steps and the reduction in catalyst loading directly lower the variable costs of production. By removing the need for cryogenic reagents and anhydrous conditions, the process also reduces energy consumption and equipment maintenance costs associated with handling hazardous chemicals. The simplified workflow decreases labor hours required for monitoring and intervention, further driving down the manufacturing overhead. These cumulative efficiencies allow for a more competitive pricing structure for the final API intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this reaction against various functional groups ensures a stable supply of diverse intermediates even when specific starting material batches vary slightly in quality. The use of common palladium salts and commercially available bases mitigates the risk of supply disruptions caused by niche reagent shortages. Furthermore, the scalability of the aqueous process from gram to ton scale is well-documented, providing confidence in the ability to meet sudden surges in demand for clinical trial materials or commercial launch volumes without the need for extensive process re-validation.
- Scalability and Environmental Compliance: Operating with water as the primary solvent aligns perfectly with increasingly stringent global environmental regulations regarding VOC emissions. This compliance future-proofs the supply chain against potential regulatory bans on certain chlorinated or aromatic solvents. The reduced hazard profile of the reaction mixture also lowers insurance premiums and safety training costs, while the simplified waste stream facilitates easier permitting for new manufacturing lines. This environmental stewardship enhances the corporate social responsibility (CSR) profile of the supply chain, a key metric for modern pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this primary amine-guided synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the process capabilities. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into existing production pipelines or for sourcing teams negotiating supply agreements for complex heterocyclic intermediates.
Q: Why is selective alkynylation at the C2 position of indole challenging?
A: Traditional electrophilic substitution favors the C3 position due to higher electron cloud density. Achieving C2 selectivity typically requires harsh pre-functionalization or specific directing groups to override natural electronic bias.
Q: What role does the primary amine play in this synthesis mechanism?
A: The primary amine acts as a powerful directing group, coordinating with the palladium catalyst to form a stable six-membered ring intermediate, which precisely guides the metal to the C2 position for activation.
Q: Is this synthesis method considered environmentally sustainable?
A: Yes, the process utilizes water as the primary solvent instead of toxic organic solvents, significantly reducing volatile organic compound (VOC) emissions and simplifying waste treatment protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkynyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality, cost-effective intermediates for the development of next-generation therapeutics. Our team of expert process chemists has extensively analyzed the potential of the primary amine-guided C-H activation pathway described in CN108864164B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from discovery to market. Our state-of-the-art facilities are designed to handle complex catalytic reactions with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to guarantee batch-to-batch consistency and regulatory compliance.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific drug development needs. Whether you require custom synthesis of novel 2-alkynyl indole derivatives or scale-up of existing routes, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume. Contact us today to request specific COA data, route feasibility assessments, and a comprehensive quotation that demonstrates how our optimized manufacturing processes can enhance your supply chain resilience and reduce overall project timelines.
