Advanced Rhodium-Catalyzed Synthesis of Benzocycloheptenones for Commercial API Manufacturing
Introduction to Novel Benzocycloheptenone Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex polycyclic scaffolds efficiently. Patent CN113620795B introduces a groundbreaking transition metal-catalyzed approach for synthesizing benzocycloheptenone compounds, a privileged structural motif found in numerous bioactive natural products and drug candidates. This innovation leverages a rhodium-catalyzed annulation strategy that couples 2-biphenylboronic acid derivatives with disubstituted cyclopropenones. Unlike traditional multi-step sequences that often suffer from low atom economy and harsh conditions, this protocol offers a streamlined, one-pot solution. The reaction proceeds under remarkably mild conditions, utilizing air as the terminal oxidant environment and common organic solvents, which significantly lowers the barrier for adoption in both academic research and industrial settings. By enabling the direct construction of the seven-membered ring through a cascade involving C-H activation and ring expansion, this technology represents a significant leap forward in synthetic efficiency.
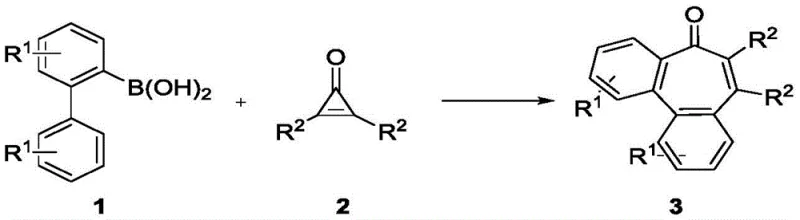
The core transformation involves the strategic use of cyclopropenones as unique three-carbon building blocks. In this process, the strained three-membered ring of the cyclopropenone undergoes a controlled cleavage and reorganization to form the target seven-membered benzocycloheptenone core. This methodology is not merely a laboratory curiosity but a viable pathway for generating diverse libraries of compounds for drug discovery. The versatility of the reaction is underscored by its broad substrate scope, accommodating various electronic and steric environments on both the biphenyl and cyclopropenone partners. For procurement and supply chain professionals, the simplicity of the starting materials—commercially available boronic acids and synthetically accessible cyclopropenones—suggests a highly stable and cost-effective supply chain for the resulting high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzocycloheptenone frameworks has been fraught with challenges that hinder large-scale production. Conventional routes often rely on intramolecular Friedel-Crafts acylations or transition-metal-mediated cyclizations that require pre-functionalized substrates, such as halides or triflates, which add significant cost and waste to the process. Furthermore, many existing methods necessitate stringent anhydrous and anaerobic conditions, requiring expensive inert gas manifolds and specialized reactor setups that increase capital expenditure. The use of stoichiometric amounts of toxic heavy metals or harsh Lewis acids in older protocols also complicates downstream processing, particularly regarding the removal of metal residues to meet stringent regulatory standards for API intermediates. Additionally, traditional approaches frequently suffer from poor regioselectivity and limited functional group tolerance, forcing chemists to employ extensive protecting group strategies that lengthen the synthetic timeline and reduce overall yield. These inefficiencies collectively drive up the cost of goods sold (COGS) and extend lead times for critical drug development projects.
The Novel Approach
The methodology disclosed in CN113620795B effectively dismantles these barriers through a clever design centered on C-H activation and strain-release driving forces. By utilizing 2-biphenylboronic acids, the process bypasses the need for pre-halogenated precursors, directly engaging the C-H bond adjacent to the biaryl system. The use of cyclopropenones serves a dual purpose: acting as both the carbon source and the driving force for the reaction due to the release of ring strain upon opening. Crucially, this new approach operates efficiently under an air atmosphere, eliminating the need for costly nitrogen or argon protection. The reaction conditions are温和 (mild), typically proceeding at 60°C in ethyl acetate, a green solvent preferred in modern green chemistry metrics. This shift from complex, multi-step, anaerobic protocols to a simple, aerobic, one-pot reaction drastically simplifies the operational workflow. For manufacturing teams, this translates to reduced batch times, lower energy consumption for heating and cooling, and simplified waste management, positioning this technology as a superior alternative for the cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Ring Expansion
Understanding the mechanistic underpinnings of this transformation is vital for R&D directors aiming to optimize the process for specific analogs. The reaction is initiated by the active cationic Rh(III) species generated in situ from the dimeric precursor [Cp*RhCl2]2 and the silver salt oxidant. The first critical step involves the coordination of the rhodium center to the oxygen atom of the carbonyl group or the pi-system, followed by concerted metalation-deprotonation (CMD) to activate the ortho-C-H bond of the biphenyl moiety. This forms a stable five-membered rhodacycle intermediate, which is the resting state of the catalyst before substrate insertion. The subsequent step involves the coordination and insertion of the cyclopropenone into the Rh-C bond. This insertion is highly regioselective, driven by both steric and electronic factors inherent to the substituents on the cyclopropenone ring.
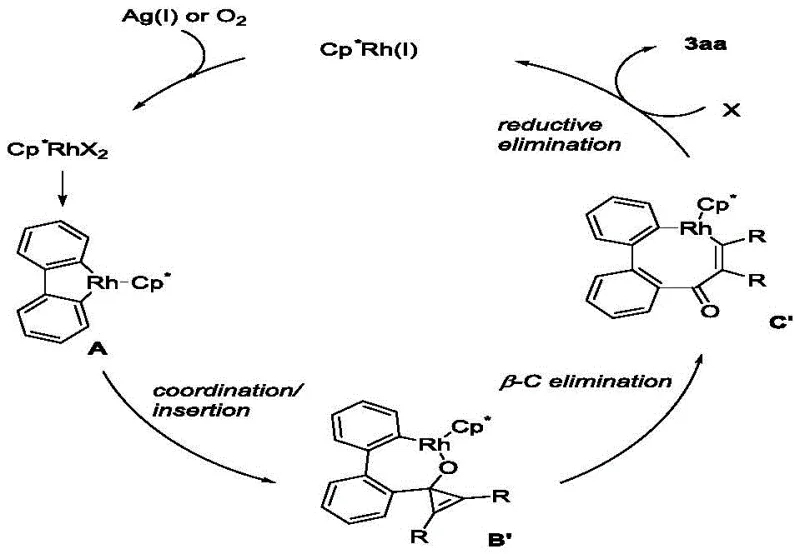
Following the insertion event, the key step that differentiates this chemistry is the beta-carbon elimination. The strained three-membered ring of the inserted cyclopropenone undergoes cleavage, expanding the metallacycle from five members to seven members. This ring-expansion step releases the significant angle strain of the cyclopropenone, providing the thermodynamic driving force for the reaction. Finally, reductive elimination occurs to release the desired benzocycloheptenone product and regenerate the Rh(I) species. The silver oxidant then re-oxidizes Rh(I) back to the active Rh(III) state, closing the catalytic cycle. This mechanism explains the high efficiency and the specific requirement for the silver salt. From an impurity control perspective, the well-defined nature of the rhodacycle intermediates minimizes side reactions such as homocoupling of the boronic acid, ensuring a clean impurity profile that simplifies purification and enhances the quality of the final high-purity OLED material or pharmaceutical intermediate.
How to Synthesize Benzocycloheptenone Efficiently
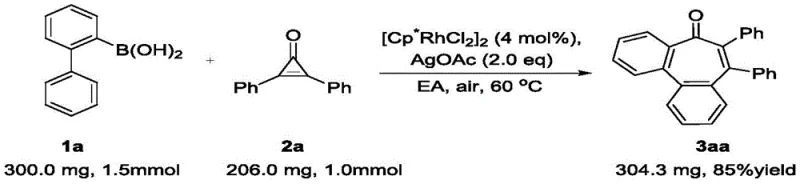
Implementing this synthesis requires attention to specific stoichiometric ratios and reaction parameters to maximize yield and reproducibility. The patent outlines a generalized procedure that has been validated across a wide array of substrates, demonstrating the robustness of the protocol. The typical operation involves charging a reaction vessel with the 2-biphenylboronic acid derivative and the disubstituted cyclopropenone in a molar ratio favoring the boronic acid slightly (e.g., 1.2:1.0 to 1.5:1.0) to ensure complete consumption of the more valuable or difficult-to-handle cyclopropenone. The catalyst loading is kept low at 4 mol% of [Cp*RhCl2]2, which is economically favorable for scale-up. The choice of solvent is critical; ethyl acetate is identified as the optimal medium, balancing solubility and reaction rate while adhering to safety and environmental guidelines. The reaction is heated to 60°C, a temperature that provides sufficient energy for the C-H activation and ring expansion steps without promoting thermal decomposition of sensitive functional groups.
- Combine 2-biphenylboronic acid derivatives and disubstituted cyclopropenones in a reaction vessel with [Cp*RhCl2]2 catalyst.
- Add Silver Acetate (AgOAc) as the oxidant and Ethyl Acetate as the solvent under ambient air conditions.
- Heat the mixture to 60°C and stir until completion, followed by standard aqueous workup and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this rhodium-catalyzed methodology offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to substantial cost savings. By eliminating the need for inert gas protection and specialized high-pressure equipment, facilities can utilize standard glass-lined or stainless steel reactors, thereby reducing capital investment and maintenance costs. Furthermore, the use of ethyl acetate, a commodity solvent with a well-established global supply chain, mitigates the risks associated with sourcing exotic or regulated solvents. The high atom economy of the reaction, where the majority of the starting material atoms are incorporated into the final product, minimizes waste generation and lowers disposal costs, aligning with increasingly strict environmental regulations.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of expensive protecting groups and the reduction in synthetic steps. Traditional routes often require 4-5 steps to achieve the same scaffold, whereas this method accomplishes it in a single pot. This consolidation reduces labor costs, utility consumption, and equipment occupancy time. Additionally, the catalyst loading is optimized to be low (4 mol%), and the silver oxidant, while a cost factor, is used in stoichiometric amounts that are manageable compared to the savings gained from shortened cycle times. The ability to run the reaction under air further reduces operational expenditures by removing the need for nitrogen generators or liquid nitrogen supplies, leading to significant overall cost reduction in API intermediate manufacturing.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically 2-biphenylboronic acids and cyclopropenones, are either commercially available or can be synthesized from abundant feedstocks like biphenyls and alkynes. This ensures a stable and reliable supply chain, reducing the risk of production delays caused by raw material shortages. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or moisture, which enhances batch-to-b consistency. For supply chain planners, this reliability translates to predictable lead times and the ability to maintain lower safety stock levels, optimizing working capital. The method's compatibility with diverse functional groups also allows for a flexible sourcing strategy, where different substituted boronic acids can be swapped in without re-validating the entire process.
- Scalability and Environmental Compliance: Scaling chemical processes from milligram to ton scale often reveals hidden bottlenecks, but this technology demonstrates excellent scalability potential. The patent includes data on scale-up trials where the reaction maintained high efficiency even when the batch size was increased, indicating good heat and mass transfer characteristics. The exothermic nature of the ring-opening step is manageable at 60°C, preventing runaway reactions. From an environmental standpoint, the process generates minimal hazardous waste. The primary byproducts are silver salts and boron species, which can be recovered or treated using standard wastewater protocols. The use of air as an oxidant avoids the generation of toxic stoichiometric oxidant waste streams common in other oxidation reactions. This green profile facilitates easier permitting and compliance with environmental, health, and safety (EHS) standards, smoothing the path for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline candidates. The answers provided reflect the current state of the art as described in CN113620795B and serve as a foundation for further process development discussions.
Q: What are the key advantages of this Rhodium-catalyzed method over traditional synthesis?
A: This method utilizes readily available 2-biphenylboronic acids and cyclopropenones, operates under mild air conditions without inert gas protection, and achieves high yields (up to 99%) with excellent functional group tolerance.
Q: Is this process scalable for industrial production?
A: Yes, the patent explicitly demonstrates successful scale-up trials maintaining high efficiency. The use of common solvents like ethyl acetate and the absence of strict anaerobic requirements facilitate large-scale manufacturing.
Q: What is the role of the silver salt oxidant in this reaction?
A: The silver salt, specifically Silver Acetate (AgOAc), acts as a crucial oxidant to regenerate the active Rh(III) species from Rh(I) during the catalytic cycle, ensuring continuous turnover and high conversion rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzocycloheptenone Supplier
The emergence of efficient synthetic routes like the one described in CN113620795B highlights the dynamic nature of modern organic synthesis, yet translating patent literature into commercial reality requires deep expertise. NINGBO INNO PHARMCHEM stands at the forefront of this translation, offering comprehensive CDMO services tailored to complex molecule manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of handling sensitive organometallic chemistry, guaranteeing that every batch meets stringent purity specifications required by global regulatory bodies. We understand that the devil is in the details, from catalyst recovery to solvent recycling, and our engineering team is dedicated to optimizing these parameters to maximize your ROI.
We invite you to leverage our technical prowess to accelerate your drug development timelines. Whether you require custom synthesis of specific benzocycloheptenone analogs or full-scale production of the core scaffold, our team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis for your project. We are prepared to provide specific COA data from our pilot runs and detailed route feasibility assessments to demonstrate how our capabilities align with your supply chain goals. Let us partner with you to turn this innovative chemistry into a commercial success story.
