Advanced Rhodium-Catalyzed Synthesis of Benzocycloheptenones for High-Value Pharmaceutical Intermediates
Advanced Rhodium-Catalyzed Synthesis of Benzocycloheptenones for High-Value Pharmaceutical Intermediates
The development of efficient synthetic routes for complex polycyclic frameworks remains a cornerstone of modern medicinal chemistry. Patent CN113620795A introduces a groundbreaking transition metal-catalyzed methodology for the synthesis of benzocycloheptenone compounds, a privileged scaffold found in numerous natural products and drug candidates exhibiting antifungal, antibacterial, and antitumor activities. This innovation leverages a rhodium(III) catalytic system to couple 2-biphenylboronic acid derivatives with disubstituted cyclopropenones, achieving the construction of the challenging seven-membered ring in a single operational step. For R&D directors and process chemists, this represents a significant leap forward in atom economy and step efficiency, bypassing the multi-step sequences traditionally required to access these sterically congested structures.
From a commercial manufacturing perspective, the robustness of this protocol is particularly noteworthy. The reaction proceeds efficiently under an air atmosphere at moderate temperatures (60°C), utilizing ethyl acetate as a green solvent. This eliminates the stringent requirement for inert gas protection (such as nitrogen or argon gloveboxes) often associated with organometallic catalysis, thereby drastically simplifying the engineering controls needed for scale-up. The broad substrate scope, accommodating various electronic and steric environments on both the biphenyl and cyclopropenone partners, ensures that this technology is not merely a niche academic curiosity but a versatile platform for generating diverse libraries of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzocycloheptenone derivatives has been plagued by inefficiencies inherent to constructing seven-membered carbocycles. Traditional approaches often rely on intramolecular cyclizations of linear precursors, which can suffer from unfavorable entropy factors leading to low yields and the formation of oligomeric by-products. Furthermore, many existing methods require harsh reaction conditions, such as strong Lewis acids or high temperatures, which are incompatible with sensitive functional groups frequently present in advanced drug intermediates. The necessity for pre-functionalized starting materials and multiple protection-deprotection steps further inflates the cost of goods sold (COGS) and extends the lead time for process development. These limitations create a bottleneck for procurement managers seeking reliable sources of complex scaffolds, as the supply chain becomes vulnerable to the yield losses and purification challenges associated with these archaic synthetic routes.
The Novel Approach
The methodology disclosed in CN113620795A fundamentally reimagines the construction of the benzocycloheptenone core through a strategic C-H activation and annulation cascade. By employing 2-biphenylboronic acids and cyclopropenones as building blocks, the process harnesses the strain energy of the three-membered cyclopropenone ring to drive the formation of the new carbon-carbon bonds. 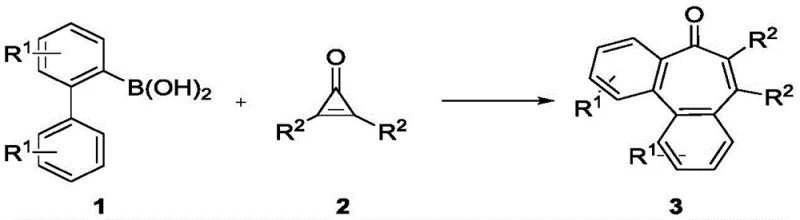 As illustrated in the general reaction scheme, this transformation is catalyzed by a Cp*Rh(III) complex in the presence of a silver salt oxidant. The novelty lies in the seamless integration of C-H activation, alkyne insertion (from the strained ring), and subsequent ring expansion, all occurring in a single pot. This approach not only reduces the number of synthetic steps but also improves the overall mass balance, offering a direct pathway to high-value targets that were previously difficult to access economically.
As illustrated in the general reaction scheme, this transformation is catalyzed by a Cp*Rh(III) complex in the presence of a silver salt oxidant. The novelty lies in the seamless integration of C-H activation, alkyne insertion (from the strained ring), and subsequent ring expansion, all occurring in a single pot. This approach not only reduces the number of synthetic steps but also improves the overall mass balance, offering a direct pathway to high-value targets that were previously difficult to access economically.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Annulation
Understanding the catalytic cycle is crucial for R&D teams aiming to optimize this process for specific substrates or troubleshoot potential impurities. The proposed mechanism initiates with the generation of the active cationic Rh(III) species, likely facilitated by the silver oxidant which abstracts a chloride ligand. This active catalyst then coordinates to the oxygen atom of the ketone or directs C-H activation on the biphenyl moiety, forming a five-membered rhodacycle intermediate. 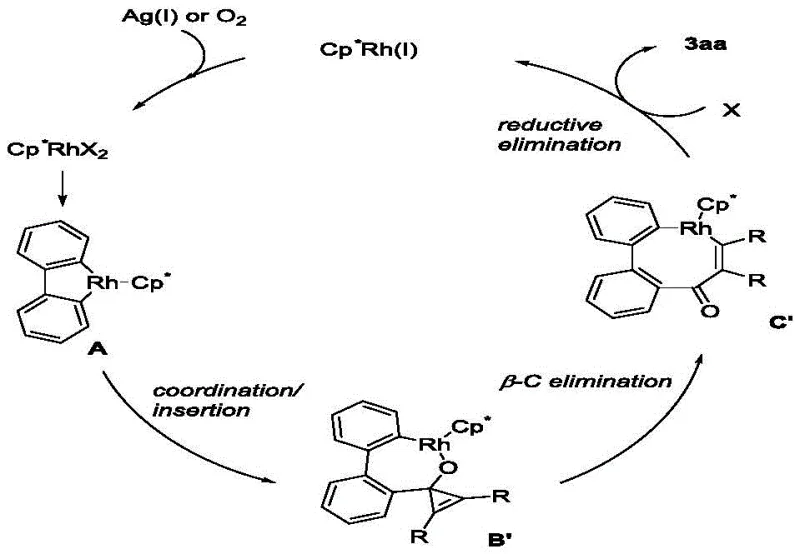 Following this, the strained cyclopropenone undergoes coordination and migratory insertion into the Rh-C bond. A critical step follows: the beta-carbon elimination of the cyclopropenone ring. This unique step releases the ring strain and expands the metallacycle, setting the stage for the final seven-membered ring formation. The cycle concludes with reductive elimination to release the benzocycloheptenone product and regenerate the Rh(I) species, which is subsequently re-oxidized to Rh(III) by the silver salt or molecular oxygen to close the catalytic loop.
Following this, the strained cyclopropenone undergoes coordination and migratory insertion into the Rh-C bond. A critical step follows: the beta-carbon elimination of the cyclopropenone ring. This unique step releases the ring strain and expands the metallacycle, setting the stage for the final seven-membered ring formation. The cycle concludes with reductive elimination to release the benzocycloheptenone product and regenerate the Rh(I) species, which is subsequently re-oxidized to Rh(III) by the silver salt or molecular oxygen to close the catalytic loop.
For quality control and impurity profiling, it is vital to note that the selectivity of the beta-C elimination step dictates the regiochemistry of the final product. The patent data indicates high selectivity for the desired benzocycloheptenone isomers, minimizing the formation of regioisomeric by-products that could complicate downstream purification. The tolerance of the catalytic system towards various substituents suggests that electronic effects play a role in the rate-determining steps, likely the C-H activation or the insertion event. This mechanistic understanding allows process chemists to fine-tune reaction parameters, such as temperature and oxidant stoichiometry, to maximize yield and purity, ensuring that the final API intermediate meets stringent regulatory specifications.
How to Synthesize Benzocycloheptenone Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it highly attractive for both laboratory discovery and pilot plant production. The general procedure involves charging a reaction vessel with the 2-biphenylboronic acid derivative, the disubstituted cyclopropenone, the rhodium catalyst precursor [Cp*RhCl2]2, and silver acetate. 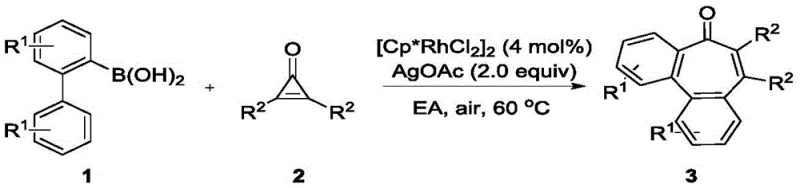 Ethyl acetate is added as the solvent, and the mixture is heated to 60°C under an open air atmosphere. The reaction progress is monitored by TLC until the starting material is consumed. Workup typically involves aqueous quenching, extraction with organic solvents like diethyl ether or ethyl acetate, and purification via silica gel column chromatography. This straightforward protocol minimizes the need for specialized handling equipment.
Ethyl acetate is added as the solvent, and the mixture is heated to 60°C under an open air atmosphere. The reaction progress is monitored by TLC until the starting material is consumed. Workup typically involves aqueous quenching, extraction with organic solvents like diethyl ether or ethyl acetate, and purification via silica gel column chromatography. This straightforward protocol minimizes the need for specialized handling equipment.
- Combine 2-biphenylboronic acid derivatives and disubstituted cyclopropenones in a reaction vessel with [Cp*RhCl2]2 catalyst.
- Add silver acetate (AgOAc) as the oxidant and ethyl acetate as the solvent under an air atmosphere.
- Heat the mixture to 60°C with stirring until completion, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic and logistical implications of this patent are profound. The shift towards a one-step synthesis from commercially available or easily prepared starting materials significantly compresses the manufacturing timeline. By eliminating multiple isolation and purification stages associated with traditional multi-step routes, the process inherently reduces solvent consumption, waste generation, and labor costs. The use of ethyl acetate, a widely available and environmentally benign solvent, further aligns with green chemistry principles and simplifies waste disposal compliance. Moreover, the ability to run the reaction in air removes the capital expenditure and operational overhead associated with maintaining large-scale inert atmosphere reactors, directly contributing to cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency here is the dramatic reduction in process complexity. Traditional routes to seven-membered rings often involve low-yielding cyclization steps that require recycling of unreacted material or extensive chromatography. In contrast, this rhodium-catalyzed method delivers high yields (often exceeding 80-90% in optimized examples) with minimal by-product formation. Although rhodium is a precious metal, the catalyst loading is low (4 mol%), and the high turnover number ensures that the metal cost per kilogram of product is negligible compared to the savings gained from reduced processing time and higher throughput. The elimination of cryogenic conditions or high-pressure hydrogenation steps further lowers the utility costs of the manufacturing facility.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The tolerance for air and moisture means that raw material handling is less critical, reducing the risk of batch failures due to environmental exposure. The starting materials, 2-biphenylboronic acids and cyclopropenones, are part of a well-established supply chain for Suzuki coupling and other organometallic transformations, ensuring consistent availability. This reliability is crucial for maintaining continuous production schedules for critical drug substances. Furthermore, the broad substrate scope means that a single manufacturing platform can be adapted to produce a variety of analogues, allowing for agile responses to changing market demands or clinical trial requirements without requalifying entirely new processes.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of this method, with examples showing successful gram-scale synthesis.
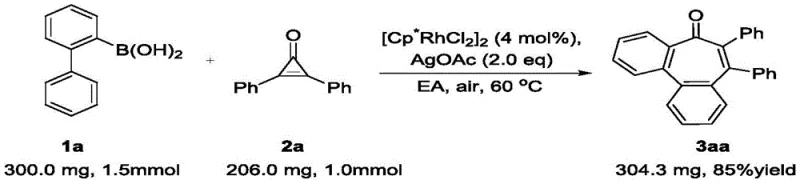 The transition from milligram to kilogram scale is facilitated by the homogeneous nature of the catalysis and the absence of hazardous reagents. From an environmental standpoint, the atom economy is superior to classical methods, and the use of silver acetate as a terminal oxidant generates manageable silver waste that can potentially be recovered and recycled. This aligns with the increasing regulatory pressure on pharmaceutical manufacturers to reduce their environmental footprint, making this technology a sustainable choice for long-term commercial production of complex organic intermediates.
The transition from milligram to kilogram scale is facilitated by the homogeneous nature of the catalysis and the absence of hazardous reagents. From an environmental standpoint, the atom economy is superior to classical methods, and the use of silver acetate as a terminal oxidant generates manageable silver waste that can potentially be recovered and recycled. This aligns with the increasing regulatory pressure on pharmaceutical manufacturers to reduce their environmental footprint, making this technology a sustainable choice for long-term commercial production of complex organic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed technology. These insights are derived directly from the experimental data and scope limitations described in the patent documentation, providing clarity for technical teams evaluating this route for their specific projects.
Q: What are the key advantages of this rhodium-catalyzed method over traditional synthesis?
A: This method utilizes readily available 2-biphenylboronic acids and cyclopropenones to construct the seven-membered ring in a single step. It operates under mild conditions (60°C) in air, eliminating the need for inert gas protection and expensive specialized equipment.
Q: Is this process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the patent demonstrates successful scale-up capabilities. The use of stable catalysts and common solvents like ethyl acetate facilitates transition from laboratory to pilot and commercial scales without significant re-optimization.
Q: What is the substrate scope for the R1 and R2 groups?
A: The reaction tolerates a wide range of functional groups including halogens (F, Cl, Br), alkyl, alkoxy, trifluoromethyl, and nitro groups. This versatility allows for the synthesis of diverse libraries of bioactive benzocycloheptenone derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzocycloheptenone Supplier
The technological breakthroughs detailed in CN113620795A underscore the immense potential of benzocycloheptenone derivatives in next-generation therapeutics. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative academic research into robust, commercial-grade manufacturing processes. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzocycloheptenone intermediate delivered adheres to the highest international standards required for pharmaceutical applications.
We invite you to leverage our technical expertise to accelerate your drug development programs. Whether you require custom synthesis of novel analogues or scale-up of the standard protocol, our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us collaborate to bring your high-purity benzocycloheptenone projects from the bench to the market with speed, efficiency, and economic viability.
