Advanced Palladium-Catalyzed Carbonylation for Scalable 2-Trifluoromethyl Quinazolinone Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2-Trifluoromethyl Quinazolinone Production
The pharmaceutical industry continuously seeks robust synthetic methodologies to access complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Patent CN112125856A discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone derivatives, addressing critical challenges in modern medicinal chemistry. Quinazolinone derivatives are privileged structures found in numerous bioactive molecules, exhibiting a broad spectrum of pharmacological activities including anti-inflammatory, antiviral, antifungal, anticonvulsant, and anticancer properties. The strategic introduction of a trifluoromethyl group at the 2-position further enhances these properties by improving metabolic stability, lipophilicity, and bioavailability. This patent presents a transition metal palladium-catalyzed carbonylation tandem reaction that utilizes readily available starting materials, offering a streamlined pathway for producing high-purity intermediates essential for drug discovery and development.
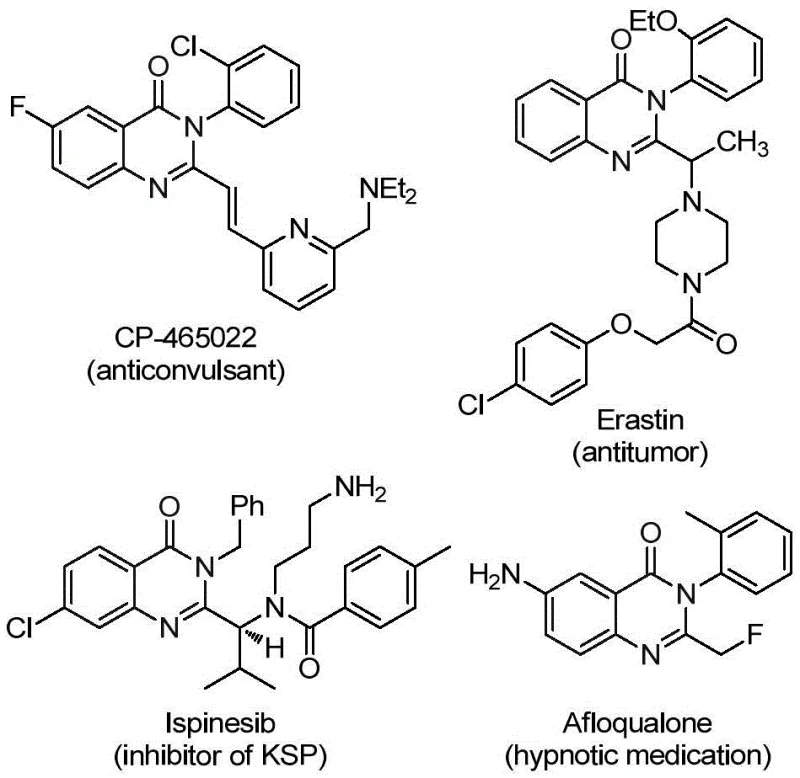
This innovative approach not only simplifies the synthetic route but also aligns with green chemistry principles by avoiding the use of toxic gaseous carbon monoxide. For R&D directors and process chemists, the ability to access these fluorinated heterocycles through a reliable and scalable process is invaluable. The method described leverages a solid carbon monoxide substitute, ensuring safer handling and easier implementation in standard laboratory and production environments. By focusing on the efficiency and versatility of this catalytic system, manufacturers can significantly reduce lead times for high-purity pharmaceutical intermediates, facilitating faster progression from bench-scale research to clinical candidate selection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted quinazolinone derivatives has been fraught with significant technical hurdles and safety concerns. Conventional literature reports typically rely on methods such as the cyclization of anthranilamide with ethyl trifluoroacetate, trifluoroacetic anhydride, or trifluoroacetic acid under varying conditions. Other approaches involve the cyclization of anthranilic esters with unstable trifluoroacetamides or the reaction of isatoic anhydride with trifluoroacetic anhydride. These traditional pathways often suffer from harsh reaction conditions that require extreme temperatures or pressures, posing risks to operational safety and equipment integrity. Furthermore, many of these methods necessitate the use of expensive or pre-activated substrates, which drastically increases the raw material costs and complicates the supply chain logistics. Additionally, the narrow substrate scope of these older techniques limits the structural diversity accessible to medicinal chemists, often resulting in low yields and difficult purification processes that hinder commercial viability.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a palladium-catalyzed carbonylation tandem reaction that fundamentally reshapes the synthetic landscape for these compounds. By employing o-iodoaniline and trifluoroacetimidoyl chloride as starting materials, the process capitalizes on the high reactivity of these precursors to drive the formation of the quinazolinone core efficiently. A key innovation is the use of 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide surrogate. This eliminates the need for handling toxic, colorless carbon monoxide gas, thereby removing a major safety bottleneck in chemical manufacturing. The reaction proceeds under relatively mild conditions at 90°C in common organic solvents like tetrahydrofuran (THF), making it highly compatible with existing infrastructure. This method not only improves yield and purity but also expands the range of compatible functional groups, allowing for the synthesis of diverse derivatives tailored to specific biological targets.
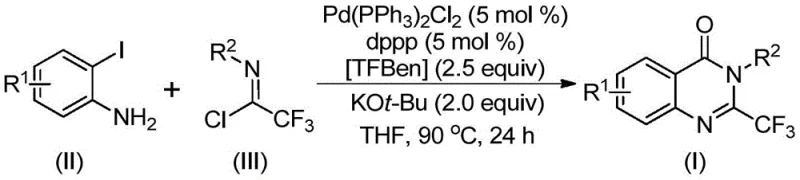
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Reaction
The mechanistic pathway of this transformation offers deep insights into its efficiency and selectivity, which are crucial for process optimization. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond coupling, facilitated by potassium tert-butoxide, to generate a trifluoroacetamidine derivative intermediate. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the aromatic ring, forming a divalent palladium intermediate. Under the heating conditions employed, the solid CO surrogate TFBen decomposes to release carbon monoxide in situ. This generated carbon monoxide then inserts into the carbon-palladium bond, creating an acyl palladium intermediate. The presence of the base further promotes the formation of a palladium-nitrogen species, leading to a seven-membered ring palladium intermediate. Finally, a reductive elimination step occurs, releasing the final 2-trifluoromethyl substituted quinazolinone derivative and regenerating the active palladium catalyst. This intricate cycle ensures high atom economy and minimizes waste generation.
From an impurity control perspective, the specificity of the palladium insertion and the controlled release of carbon monoxide from the solid surrogate play pivotal roles. Unlike gaseous CO, which can lead to uncontrolled side reactions or over-carbonylation if not managed precisely, the solid surrogate provides a steady, stoichiometric release of CO, ensuring that the acyl palladium intermediate forms selectively. This controlled environment significantly reduces the formation of by-products such as dehalogenated anilines or homocoupling products, which are common in traditional carbonylation reactions. The compatibility with various substituents on the aryl rings, including electron-withdrawing groups like nitro and halogens, as well as electron-donating alkyl groups, demonstrates the robustness of the catalytic system. This broad substrate tolerance allows for the modular synthesis of complex libraries, enabling rapid structure-activity relationship (SAR) studies without the need for extensive re-optimization of reaction conditions for each new analog.
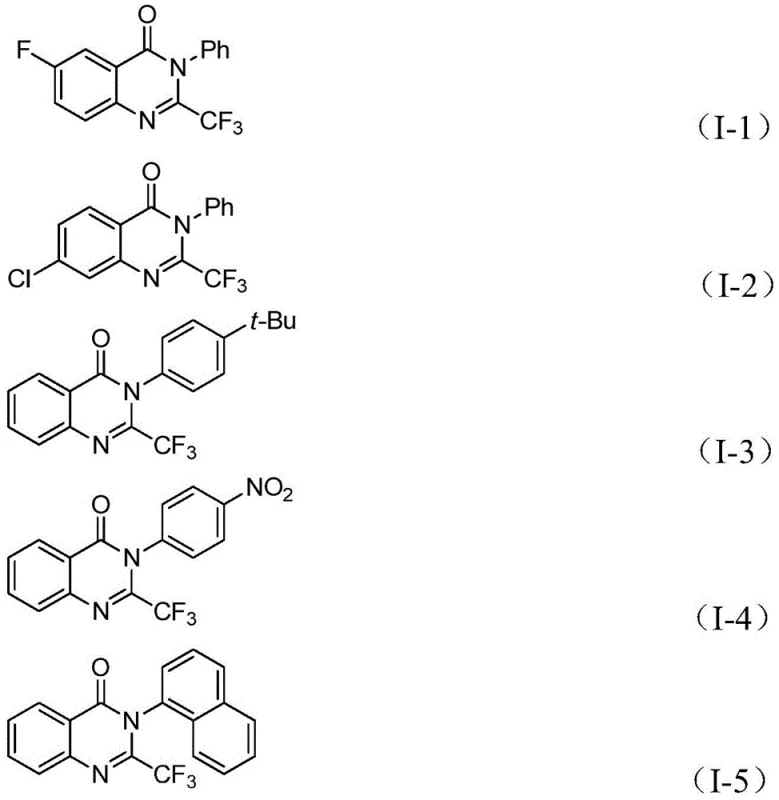
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible framework for generating these valuable intermediates. The process involves charging a reaction vessel with the palladium catalyst, specifically bis(triphenylphosphine)palladium(II) dichloride, along with the bidentate ligand 1,3-bis(diphenylphosphino)propane (dppp). To this mixture, the solid carbon monoxide source TFBen, the base potassium tert-butoxide, the o-iodoaniline substrate, and the trifluoroacetimidoyl chloride are added in an organic solvent such as THF. The detailed standardized synthesis steps below outline the precise molar ratios and operational parameters required to achieve optimal conversion and yield, ensuring consistency across different batches and scales.
- Combine palladium catalyst, dppp ligand, potassium tert-butoxide, TFBen (solid CO source), trifluoroacetimidoyl chloride, and o-iodoaniline in an organic solvent like THF.
- Heat the reaction mixture to 90°C and maintain stirring for 16 to 30 hours to allow the carbonylation tandem reaction to proceed to completion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl quinazolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits that extend beyond mere chemical efficiency. The shift from hazardous gaseous reagents to stable solid surrogates fundamentally alters the risk profile of the manufacturing process, leading to substantial cost savings in terms of safety infrastructure and regulatory compliance. By utilizing commercially available and inexpensive starting materials like o-iodoaniline derivatives and trifluoroacetimidoyl chlorides, the raw material costs are significantly reduced compared to specialized pre-activated substrates required by older methods. Furthermore, the high yield and purity achieved minimize the need for extensive downstream purification, reducing solvent consumption and waste disposal costs. This streamlined process enhances the overall economic viability of producing these critical pharmaceutical intermediates on a commercial scale.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas removes the necessity for specialized high-pressure reactors and complex gas handling systems, which are capital-intensive and require rigorous maintenance. Instead, the use of a solid CO surrogate allows the reaction to proceed in standard glass-lined or stainless steel reactors under atmospheric or low pressure. This simplification of equipment requirements translates directly into lower capital expenditure (CAPEX) and operational expenditure (OPEX). Additionally, the high catalytic efficiency means that lower loadings of the precious palladium catalyst can be utilized while maintaining high turnover numbers, further driving down the cost per kilogram of the final product. The avoidance of expensive activating agents and the use of cheap, bulk-available solvents like THF contribute to a leaner and more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals ensures a stable and resilient supply chain. Starting materials such as substituted o-iodoanilines and trifluoroacetimidoyl chlorides are produced by multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that variations in raw material quality have minimal impact on the final outcome, ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of pharmaceutical clients. The simplified post-treatment process, involving basic filtration and chromatography, reduces the turnaround time from reaction completion to finished goods, allowing for faster response to market demands.
- Scalability and Environmental Compliance: Scaling up chemical processes often introduces new challenges, but this methodology is inherently designed for scalability. The use of a solid CO source eliminates the mass transfer limitations associated with gas-liquid reactions, ensuring uniform reaction kinetics even in large-volume reactors. This facilitates a smoother transition from pilot plant to full-scale commercial production without the need for significant process re-engineering. From an environmental standpoint, the method aligns with green chemistry principles by reducing the use of hazardous substances and minimizing waste generation. The high atom economy and the ability to recover and recycle the palladium catalyst further enhance the sustainability profile of the process, helping manufacturers meet increasingly stringent environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2-trifluoromethyl quinazolinone derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals seeking to implement this technology.
Q: What are the safety advantages of this synthesis method compared to traditional carbonylation?
A: This method utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide surrogate, effectively eliminating the need for handling toxic and hazardous carbon monoxide gas, thereby significantly enhancing operational safety in industrial settings.
Q: What is the substrate scope for this palladium-catalyzed reaction?
A: The protocol demonstrates excellent compatibility with various substituents on both the o-iodoaniline and the imidoyl chloride components, including halogens (F, Cl, Br), alkyl groups, and nitro groups, allowing for the synthesis of diverse derivatives.
Q: Why is the introduction of a trifluoromethyl group beneficial in drug design?
A: Incorporating a trifluoromethyl group significantly improves the physicochemical properties of the parent molecule, such as enhancing metabolic stability, lipophilicity, and bioavailability, which are critical parameters for developing effective pharmaceutical agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel analogs or large-scale supply of established intermediates, our flexible manufacturing capabilities allow us to adapt quickly to your evolving requirements.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can accelerate your timeline and reduce your overall manufacturing costs. Let us be your partner in turning complex chemical challenges into commercial successes.
