Advanced Synthesis of 2-Trifluoromethyl Quinazolinone: Bridging Pharmaceutical Innovation and Commercial Scalability
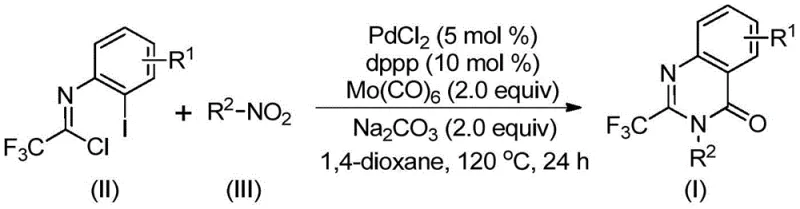
The recently granted Chinese patent CN112480015B introduces a transformative multi-component one-pot methodology for synthesizing 2-trifluoromethyl substituted quinazolinone compounds, representing a significant advancement in the production of critical pharmaceutical intermediates. This innovative approach addresses longstanding challenges in quinazolinone synthesis by eliminating the need for high-pressure carbon monoxide environments and expensive pre-functionalized substrates that have historically constrained commercial adoption. The process leverages readily available nitro compounds and trifluoroethylimidoyl chloride as starting materials, operating under mild conditions that enhance both safety and operational feasibility for large-scale manufacturing. By integrating palladium-catalyzed carbonylation with cascade reaction sequences, the methodology achieves superior substrate compatibility across diverse functional groups while maintaining exceptional reaction efficiency. This breakthrough not only expands the synthetic toolbox for medicinal chemists but also establishes a more economically viable pathway for producing quinazolinone-based active pharmaceutical ingredients with enhanced physicochemical properties critical for drug development programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for quinazolinone compounds have been plagued by significant operational constraints that hinder their commercial viability for pharmaceutical manufacturing. The most common approaches require high-pressure carbon monoxide environments coupled with ruthenium or platinum catalysts, creating substantial safety hazards and necessitating specialized equipment that dramatically increases capital expenditure. Alternative methods involving iron-catalyzed condensation or palladium-mediated cyclization often demand pre-activated substrates that are both expensive and difficult to source consistently, leading to supply chain vulnerabilities. These conventional processes typically suffer from narrow substrate scope, limiting their applicability across diverse molecular architectures required in modern drug discovery. Furthermore, the harsh reaction conditions frequently result in lower yields due to competing side reactions and decomposition pathways, while the complex purification requirements for removing transition metal residues add significant cost and time to the manufacturing process. The cumulative effect of these limitations has restricted the widespread adoption of quinazolinone-based compounds despite their well-documented therapeutic potential across multiple disease areas.
The Novel Approach
The patented methodology overcomes these historical limitations through an elegant multi-component one-pot design that operates under significantly milder conditions while maintaining exceptional efficiency. By utilizing palladium chloride with dppp ligand in combination with molybdenum hexacarbonyl as a carbon monoxide surrogate, the process eliminates the need for high-pressure CO equipment while achieving comparable reaction efficiency. The strategic selection of inexpensive and readily available starting materials—particularly nitro compounds that are abundant in nature—creates a more sustainable and economically attractive supply chain foundation. This approach demonstrates remarkable functional group tolerance, accommodating a wide range of substituents on both the imidoyl chloride and nitro compound components without requiring pre-modification. The optimized reaction conditions at 120°C in dioxane solvent provide excellent conversion rates while minimizing energy consumption compared to traditional high-temperature processes. Most critically, the streamlined workup procedure involving simple filtration followed by column chromatography significantly reduces processing time and eliminates complex metal removal steps that have historically complicated quinazolinone production at commercial scale.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The reaction mechanism proceeds through a sophisticated cascade sequence that begins with molybdenum hexacarbonyl-mediated reduction of the nitro compound to the corresponding amine intermediate under thermal conditions. This amine then undergoes base-promoted coupling with trifluoroethylimidoyl chloride to form a key trifluoroacetamidine intermediate, which subsequently participates in the palladium-catalyzed cyclization sequence. The palladium catalyst inserts into the carbon-iodine bond of the imidoyl chloride component, generating a reactive organopalladium species that facilitates carbon monoxide insertion from the molybdenum carbonyl source. This critical step forms an acylpalladium intermediate that undergoes intramolecular cyclization through nitrogen coordination, ultimately delivering the desired quinazolinone scaffold via reductive elimination. The carefully balanced stoichiometry of palladium catalyst (5 mol%), dppp ligand (10 mol%), and sodium carbonate base (2.0 equiv) creates an optimal catalytic environment that maximizes turnover frequency while minimizing undesired side reactions that could compromise product purity.
Impurity control is inherently engineered into this catalytic system through multiple mechanistic safeguards that prevent common degradation pathways observed in alternative syntheses. The mild reaction temperature of 120°C avoids thermal decomposition routes that typically generate regioisomeric impurities in conventional high-energy processes. The precise control over carbon monoxide delivery through the molybdenum carbonyl source prevents over-carbonylation side products that frequently occur with direct CO gas introduction. The base-mediated coupling step ensures clean formation of the critical amide bond without racemization or epimerization concerns that plague other methodologies. Furthermore, the absence of strong oxidizing or reducing agents eliminates redox-related impurities that complicate purification in competing approaches. This integrated design approach results in consistently high product purity with minimal byproduct formation, significantly reducing the burden on downstream purification processes while ensuring compliance with stringent pharmaceutical quality standards required for active pharmaceutical ingredient intermediates.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
This innovative synthesis protocol represents a paradigm shift in quinazolinone manufacturing by integrating multiple transformation steps into a single operational sequence that dramatically improves process efficiency while maintaining exceptional product quality. The methodology leverages commercially available starting materials with established supply chains, eliminating dependencies on specialized or hazardous reagents that have historically constrained production scalability. By operating under moderate temperature conditions without requiring pressurized gas systems, the process significantly enhances operational safety while reducing capital equipment requirements for manufacturing facilities. The carefully optimized catalyst system delivers consistent high yields across diverse substrate combinations, providing medicinal chemists with unprecedented flexibility in molecular design while maintaining robust process performance. Detailed standardized synthesis procedures for implementing this technology are provided in the following section to facilitate seamless technology transfer and process validation.
- Prepare reaction mixture with precise stoichiometric ratios of trifluoroethylimidoyl chloride, nitro compounds, palladium catalyst, and ligand in anhydrous dioxane solvent under inert atmosphere
- Conduct carbonylation cascade reaction at controlled temperature of 120°C for optimized duration, monitoring reaction progression through standard analytical techniques
- Implement streamlined purification protocol involving filtration, silica gel mixing, and column chromatography to achieve stringent purity specifications required for pharmaceutical applications
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial strategic advantages for procurement and supply chain management teams seeking reliable sources of high-quality pharmaceutical intermediates with consistent availability and favorable cost profiles. The process design directly addresses critical pain points in traditional quinazolinone production by eliminating dependencies on specialized equipment and hazardous reagents that create supply chain vulnerabilities and increase operational complexity. By utilizing readily available starting materials with multiple global suppliers, the approach significantly reduces single-source dependency risks while enhancing supply chain resilience against market fluctuations and geopolitical disruptions. The streamlined manufacturing sequence with fewer processing steps translates to shorter production cycles and improved capacity utilization, directly supporting just-in-time inventory management strategies essential for modern pharmaceutical manufacturing operations.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide infrastructure requirements represents a significant capital expenditure reduction for manufacturing facilities, while the use of commercially available starting materials at favorable price points creates immediate raw material cost advantages. The simplified reaction sequence reduces utility consumption and processing time compared to conventional multi-step approaches, contributing to lower overall manufacturing costs without compromising product quality. Most importantly, the avoidance of expensive transition metal catalysts that require complex removal procedures eliminates substantial downstream processing costs associated with metal residue testing and remediation, creating a more economically sustainable production pathway for quinazolinone intermediates.
- Enhanced Supply Chain Reliability: The strategic selection of starting materials from established global chemical suppliers ensures consistent availability and reduces vulnerability to supply chain disruptions that frequently impact specialized reagents used in alternative synthetic routes. The process demonstrates exceptional robustness across different batches of raw materials, minimizing quality variations that could disrupt production schedules or require costly revalidation procedures. The ability to scale seamlessly from laboratory development to commercial production without significant process modifications provides procurement teams with confidence in long-term supply continuity, while the simplified logistics requirements reduce transportation and storage complexities associated with hazardous materials handling.
- Scalability and Environmental Compliance: The methodology has been successfully demonstrated from gram-scale laboratory validation to pilot plant production, confirming its suitability for commercial manufacturing without requiring fundamental process changes. The elimination of hazardous reagents and high-pressure operations significantly improves workplace safety profiles while reducing environmental impact through lower energy consumption and minimized waste generation. The streamlined purification protocol generates less solvent waste compared to traditional multi-step approaches, aligning with increasingly stringent environmental regulations while supporting corporate sustainability initiatives. This environmentally responsible manufacturing approach provides additional value to pharmaceutical companies seeking greener supply chain partners without compromising on product quality or cost efficiency.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patented methodology and its implementation in pharmaceutical intermediate manufacturing. These insights have been developed through comprehensive evaluation of the reaction mechanism, process parameters, and scalability data presented in the original patent documentation to provide actionable information for procurement and technical decision-makers.
Q: How does this method improve upon conventional quinazolinone synthesis routes?
A: This palladium-catalyzed one-pot method eliminates the need for high-pressure CO conditions and pre-functionalized substrates required in traditional approaches. The process utilizes readily available nitro compounds and avoids expensive transition metal catalysts that necessitate complex removal procedures, significantly simplifying the manufacturing workflow while maintaining high substrate compatibility across diverse functional groups.
Q: What are the key advantages for pharmaceutical supply chain management?
A: The methodology leverages commercially accessible starting materials with broad supplier networks, reducing single-source dependency risks. The simplified reaction sequence with fewer processing steps enhances production reliability and shortens lead times. The demonstrated scalability from laboratory to commercial production ensures consistent supply continuity for pharmaceutical manufacturers requiring high-purity intermediates.
Q: How does the process ensure high purity standards for pharmaceutical applications?
A: The reaction design inherently minimizes byproduct formation through selective catalytic pathways, while the optimized purification protocol effectively removes residual metals and impurities. The absence of harsh reaction conditions prevents decomposition pathways that typically generate difficult-to-remove impurities in conventional syntheses, enabling consistent achievement of pharmaceutical-grade purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
Our patented methodology represents a significant advancement in quinazolinone intermediate production that directly addresses critical challenges faced by pharmaceutical manufacturers seeking reliable sources of high-quality building blocks for drug development programs. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring seamless technology transfer from laboratory validation to full-scale manufacturing operations while maintaining stringent purity specifications required for pharmaceutical applications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of comprehensive analytical characterization to guarantee consistent product quality meeting global regulatory standards across all production scales.
We invite your technical procurement team to request our Customized Cost-Saving Analysis tailored to your specific manufacturing requirements, which includes detailed route feasibility assessments and comparative cost modeling against conventional synthesis methods. Please contact us to obtain specific COA data demonstrating our product quality consistency and discuss how our innovative approach can enhance your supply chain resilience while supporting your drug development timelines.
