Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Intermediates
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. Quinazolinones are privileged structures found in numerous natural products and drug candidates, exhibiting a wide spectrum of biological activities including anti-cancer, anticonvulsant, anti-inflammatory, antifungal, and antimalarial properties. The strategic introduction of a trifluoromethyl group into these heterocyclic systems further enhances their pharmacokinetic profiles by improving electronegativity, metabolic stability, and lipophilicity. This patent presents a transformative approach that leverages inexpensive iron catalysis to achieve these valuable transformations, addressing long-standing challenges in synthetic efficiency and economic viability for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives bearing trifluoromethyl functionalities has relied heavily on cyclization reactions involving specific synthons such as anthranilamide, anthranilic acid, or isatoic anhydride reacting with trifluoroacetic anhydride or ethyl trifluoroacetate. While these traditional pathways have served the industry for years, they are increasingly recognized for their significant operational drawbacks. These conventional methods often necessitate severe reaction conditions that can compromise sensitive functional groups, leading to complex impurity profiles that are difficult to separate. Furthermore, the starting materials required, particularly specialized trifluoromethyl synthons, are frequently expensive and not readily available in bulk quantities, creating supply chain bottlenecks. The narrow substrate scope of these older techniques limits the structural diversity accessible to medicinal chemists, often resulting in low yields that are economically unsustainable for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN111675662B introduces a highly efficient and versatile synthetic route utilizing readily available trifluoroethylimidoyl chloride and isatin as primary starting materials. This innovative strategy employs a cheap metal iron catalyst, specifically ferric chloride, to drive a series of cyclization reactions that construct the quinazolinone core with high precision. The reaction design allows for exceptional functional group tolerance, enabling the incorporation of various substituents such as alkyl, halogen, and methoxy groups at different positions on the aromatic rings. As illustrated in the general reaction scheme below, this method streamlines the synthesis process, converting simple precursors directly into the desired 2-trifluoromethyl substituted quinazolinone structure with remarkable efficiency.
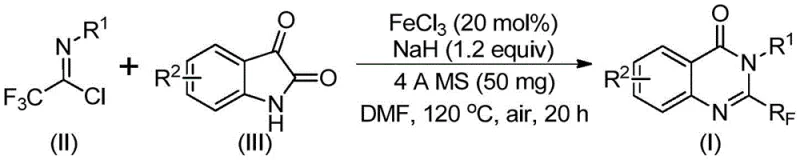
The scope of this novel approach is further demonstrated by the successful synthesis of specific derivatives, such as compounds (I-1) through (I-5), which showcase the method's ability to handle diverse substitution patterns including methyl, fluoro, bromo, and chloro groups. This flexibility is crucial for drug discovery programs where rapid analog generation is required to optimize biological activity. By shifting away from expensive precious metals and harsh reagents, this new methodology not only simplifies the synthetic workflow but also opens up new possibilities for designing and synthesizing quinazolinone compounds tailored to specific therapeutic needs, thereby enhancing the overall practicability and commercial potential of these valuable chemical entities.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the iron catalyst and the base promoter. The reaction initiates with an alkali-promoted formation of carbon-nitrogen bonds between the trifluoroethylimidoyl chloride and the isatin substrate. This initial step generates a trifluoroacetamidine intermediate, which serves as the precursor for the subsequent ring-closing events. The presence of sodium hydride is critical in deprotonating the isatin nitrogen, thereby increasing its nucleophilicity and facilitating the attack on the imidoyl chloride. Following this bond formation, the system undergoes an iron-catalyzed decarbonylation and cyclization sequence. The ferric chloride acts as a Lewis acid, coordinating with the carbonyl oxygen and activating the molecule for the loss of carbon monoxide, which drives the thermodynamic equilibrium towards the formation of the stable quinazolinone ring system. This isomerization process is highly selective, ensuring that the trifluoromethyl group is positioned correctly at the 2-position of the quinazolinone ring.
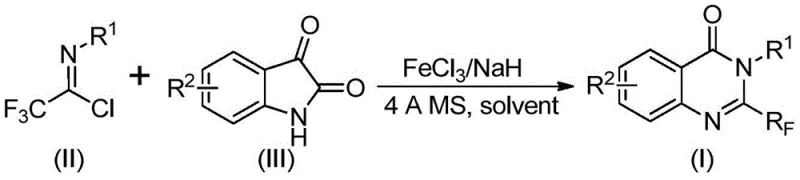
From an impurity control perspective, the use of 4A molecular sieves plays a pivotal role in maintaining reaction integrity. These sieves effectively scavenge trace amounts of water that could otherwise hydrolyze the sensitive imidoyl chloride starting material or interfere with the sodium hydride base, leading to side reactions and reduced yields. The choice of dimethylformamide (DMF) as the solvent is also mechanistically significant, as its high polarity and aprotic nature facilitate the dissolution of ionic intermediates and stabilize the transition states involved in the cyclization. The two-stage temperature profile, starting at 40°C to allow for initial adduct formation and then ramping to 120°C to drive the decarbonylation and cyclization, ensures that each step proceeds with optimal kinetics. This careful orchestration of reaction parameters minimizes the formation of by-products, resulting in a cleaner crude reaction mixture that simplifies downstream purification and ensures the delivery of high-purity OLED material or pharmaceutical intermediates.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized protocol to maximize yield and purity. The process begins with the precise weighing and mixing of ferric chloride, sodium hydride, and activated 4A molecular sieves in a dry reaction vessel under an inert atmosphere to prevent moisture ingress. The trifluoroethylimidoyl chloride and isatin substrates are then introduced into the organic solvent, typically DMF, and the mixture is stirred uniformly to ensure homogeneous contact between all reagents. The reaction is allowed to proceed at a moderate temperature of 40°C for approximately 8 to 10 hours to facilitate the initial coupling, followed by heating to 120°C for an extended period of 18 to 20 hours to complete the cyclization. Upon completion, the reaction mixture is filtered to remove solid residues, and the filtrate is processed through standard workup procedures involving silica gel mixing and column chromatography purification to isolate the target compound. For a detailed, step-by-step breakdown of the standardized synthesis procedure, please refer to the guide below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C and react for an additional 18-20 hours.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed synthesis route offers substantial strategic benefits that extend far beyond simple chemical transformation. The shift from precious metal catalysts to ubiquitous iron salts represents a fundamental change in the cost structure of manufacturing these intermediates. By eliminating the need for expensive palladium, rhodium, or iridium complexes, manufacturers can significantly reduce the raw material costs associated with catalysis. Furthermore, the removal of heavy metal residues from the final product becomes trivial, as iron is generally regarded as safe and is easily removed or tolerated in many downstream processes, thereby saving the considerable time and expense associated with rigorous heavy metal scavenging steps often required in pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the utilization of commodity chemicals rather than specialized reagents. Trifluoroethylimidoyl chloride and isatin are commercially available in large quantities at competitive prices, ensuring a stable and predictable cost base for production. The catalyst, ferric chloride, is one of the most inexpensive Lewis acids available on the global market, contributing to a drastic simplification of the bill of materials. Additionally, the high conversion rates and good yields observed across a wide range of substrates mean that less raw material is wasted, further driving down the cost per kilogram of the final active pharmaceutical ingredient or intermediate. This efficiency translates directly into improved margins and the ability to offer more competitive pricing to downstream partners without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the widespread availability of the key reagents used in this synthesis. Unlike proprietary or niche catalysts that may be sourced from a single supplier with long lead times, ferric chloride and sodium hydride are produced by multiple chemical manufacturers worldwide, mitigating the risk of supply disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, allowing for greater flexibility in sourcing. This reliability ensures consistent production schedules and reduces the likelihood of delays caused by material shortages, which is critical for maintaining the continuity of supply for high-purity pharmaceutical intermediates required by global drug developers.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns well with modern green chemistry principles. The use of iron, a non-toxic and abundant metal, reduces the environmental footprint compared to processes relying on toxic heavy metals. The reaction operates in a single pot with a straightforward workup, minimizing the generation of hazardous waste streams and reducing the solvent load required for purification. The simplicity of the protocol, involving basic filtration and chromatography, makes it highly amenable to scale-up from gram-scale laboratory experiments to multi-ton commercial production. This ease of scaling ensures that the technology can meet growing market demands efficiently while adhering to increasingly stringent environmental regulations regarding waste disposal and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of this route for their specific projects and for procurement teams assessing the long-term viability of the supply chain. The information provided here aims to clarify the operational parameters and the strategic advantages of adopting this iron-catalyzed approach for the production of quinazolinone derivatives.
Q: What are the advantages of using FeCl3 over traditional catalysts?
A: Ferric chloride is significantly cheaper and more abundant than precious metal catalysts, reducing overall production costs while maintaining high efficiency and functional group tolerance.
Q: Can this method accommodate diverse substrate substitutions?
A: Yes, the method demonstrates excellent functional group tolerance, allowing for various substituents such as alkyl, halogen, and methoxy groups on both the aryl and isatin rings.
Q: Is this process suitable for large-scale manufacturing?
A: The protocol uses readily available raw materials and simple post-treatment steps like filtration and chromatography, making it highly amenable to gram-level and industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the technology disclosed in patent CN111675662B and is fully prepared to leverage this iron-catalyzed methodology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and reliable. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone we deliver meets the highest standards of quality required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to discuss your project further,索取 specific COA data for our existing inventory, or request detailed route feasibility assessments for your custom synthesis needs. Let us help you accelerate your drug development timeline with our reliable supply of high-quality chemical intermediates.
