Advanced One-Pot Carbonylation Strategy for Scalable Quinazolinone Pharmaceutical Intermediates
Introduction to Next-Generation Quinazolinone Synthesis
The quinazolinone scaffold represents a cornerstone structure in modern medicinal chemistry, underpinning the efficacy of numerous blockbuster drugs ranging from antifungal agents to potent anticancer therapeutics. As illustrated in the structural diversity of known bioactive molecules such as Methaqualone and Afloqualone, the incorporation of fluorine-containing groups, particularly the trifluoromethyl moiety, dramatically enhances metabolic stability and lipophilicity. 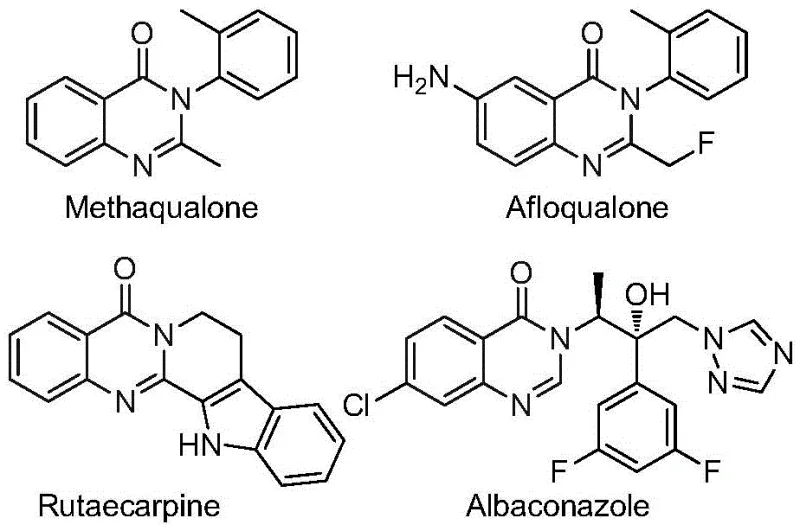 Recent advancements documented in patent CN112480015B introduce a transformative multi-component one-pot methodology that addresses long-standing synthetic bottlenecks. This innovative approach leverages a palladium-catalyzed carbonylation cascade to construct 2-trifluoromethyl substituted quinazolinones directly from inexpensive nitro compounds and trifluoroethylimidoyl chlorides. For R&D directors and procurement strategists, this development signals a pivotal shift towards more economical and operationally simpler manufacturing protocols for high-value pharmaceutical intermediates.
Recent advancements documented in patent CN112480015B introduce a transformative multi-component one-pot methodology that addresses long-standing synthetic bottlenecks. This innovative approach leverages a palladium-catalyzed carbonylation cascade to construct 2-trifluoromethyl substituted quinazolinones directly from inexpensive nitro compounds and trifluoroethylimidoyl chlorides. For R&D directors and procurement strategists, this development signals a pivotal shift towards more economical and operationally simpler manufacturing protocols for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied on methodologies that impose significant logistical and safety burdens on industrial production. Traditional routes often necessitate the use of high-pressure carbon monoxide gas, requiring specialized autoclaves and rigorous safety protocols that inflate capital expenditure. Furthermore, many established protocols depend on pre-activated substrates such as 2-bromoformylanilines or acid anhydrides, which are not only costly to procure but also generate substantial stoichiometric waste. The reliance on noble metals like ruthenium or platinum in earlier iterations further exacerbates cost issues, while narrow substrate scopes limit the ability to rapidly generate diverse analogue libraries for structure-activity relationship studies. These factors collectively hinder the efficient scale-up required for commercial API manufacturing.
The Novel Approach
The methodology disclosed in the referenced patent circumvents these challenges by employing a clever tandem reaction sequence that utilizes solid molybdenum hexacarbonyl as a safe and convenient carbon monoxide surrogate. 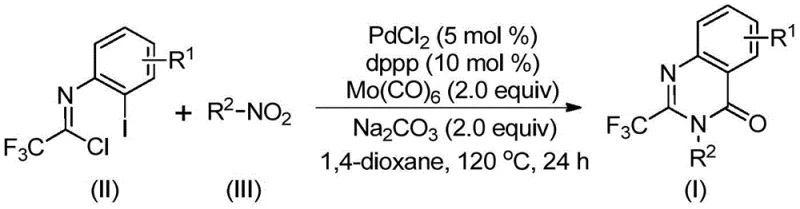 By reacting readily available nitro compounds with trifluoroethylimidoyl chlorides in the presence of a PdCl2/dppp catalyst system, the process achieves ring closure under relatively mild thermal conditions without external CO gas. This one-pot strategy eliminates the need for isolating sensitive amine intermediates, thereby streamlining the workflow and reducing solvent consumption. The compatibility with a wide range of functional groups on both the nitroarene and the imidoyl chloride components ensures that this platform technology can be adapted for the synthesis of complex drug candidates with minimal process re-engineering.
By reacting readily available nitro compounds with trifluoroethylimidoyl chlorides in the presence of a PdCl2/dppp catalyst system, the process achieves ring closure under relatively mild thermal conditions without external CO gas. This one-pot strategy eliminates the need for isolating sensitive amine intermediates, thereby streamlining the workflow and reducing solvent consumption. The compatibility with a wide range of functional groups on both the nitroarene and the imidoyl chloride components ensures that this platform technology can be adapted for the synthesis of complex drug candidates with minimal process re-engineering.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The success of this transformation hinges on a sophisticated catalytic cycle that orchestrates reduction, coupling, and cyclization within a single vessel. The mechanism likely initiates with the reduction of the nitro group to an amine by Mo(CO)6, followed immediately by a base-promoted nucleophilic attack on the imidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the aromatic ring, generating a reactive organopalladium species. The carbon monoxide released in situ from the molybdenum complex then inserts into the carbon-palladium bond to form an acyl-palladium intermediate. This critical acylation step sets the stage for intramolecular cyclization, where the nitrogen atom attacks the carbonyl carbon, eventually leading to reductive elimination that releases the final 2-trifluoromethyl quinazolinone product.  Understanding this mechanistic pathway is vital for process chemists aiming to optimize reaction parameters such as temperature and ligand loading to maximize turnover numbers.
Understanding this mechanistic pathway is vital for process chemists aiming to optimize reaction parameters such as temperature and ligand loading to maximize turnover numbers.
From an impurity control perspective, the concerted nature of this cascade reaction offers distinct advantages over stepwise syntheses. By avoiding the isolation of the free amine intermediate, the process minimizes exposure to air and moisture, which are common sources of oxidation byproducts and degradation. The use of sodium carbonate as a mild base helps to neutralize the hydrochloric acid byproduct generated during the amidine formation without promoting hydrolysis of the sensitive imidoyl chloride starting material. Furthermore, the specific choice of the dppp ligand stabilizes the palladium center, preventing the formation of palladium black and ensuring consistent catalytic activity throughout the extended reaction time of 16 to 30 hours. This robustness translates directly to a cleaner reaction profile, reducing the burden on downstream purification units and improving overall mass balance.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis requires precise control over reagent stoichiometry and reaction conditions to ensure high conversion and selectivity. The protocol dictates the use of an aprotic solvent such as 1,4-dioxane, which effectively dissolves both the organic substrates and the inorganic base while maintaining thermal stability at the required 120°C operating temperature. The molar ratio of the trifluoroethylimidoyl chloride to the nitro compound is typically maintained at roughly 1:1.2, ensuring that the nitro compound is in slight excess to drive the initial reduction and coupling steps to completion. Adhering to these standardized parameters allows for the reproducible generation of high-purity intermediates suitable for subsequent biological evaluation or formulation.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow the carbonylation cascade and cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and operational benefits that extend beyond simple yield improvements. The primary driver for cost reduction lies in the substitution of expensive, pre-functionalized starting materials with commodity-grade nitro compounds and imidoyl chlorides. These feedstocks are produced on a massive global scale, ensuring a stable supply base that is less susceptible to the price volatility often seen with specialized fine chemicals. Additionally, the elimination of high-pressure carbon monoxide gas removes the need for specialized gas handling infrastructure and safety audits, significantly lowering the barrier to entry for contract manufacturing organizations looking to offer this chemistry.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of earth-abundant additives and the avoidance of stoichiometric activating agents. By utilizing Mo(CO)6 as a solid CO source, the process avoids the logistical costs and safety premiums associated with transporting and storing compressed toxic gases. Furthermore, the high atom economy of the cascade reaction means that fewer raw materials are wasted as byproducts, leading to a more sustainable and cost-efficient production cycle that aligns with green chemistry principles.
- Enhanced Supply Chain Reliability: Reliance on widely available nitroarenes mitigates the risk of supply disruptions that can occur when depending on single-source suppliers for exotic reagents. The robustness of the reaction conditions, which tolerate a variety of substituents including halogens and alkyl groups, means that the same production line can be easily switched between different analogues without extensive cleaning or re-validation. This flexibility allows manufacturers to respond rapidly to changing demand patterns in the pharmaceutical market, ensuring continuous availability of critical intermediates.
- Scalability and Environmental Compliance: The scalability of this method is evidenced by its successful demonstration on gram scales with straightforward workup procedures involving filtration and chromatography. The absence of heavy metal waste streams typical of other transition metal-catalyzed processes simplifies wastewater treatment and regulatory compliance. As production volumes increase from pilot plant to commercial tonnage, the simplified downstream processing will result in substantial reductions in solvent usage and energy consumption, contributing to a lower overall environmental footprint for the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic methodology. These insights are derived directly from the experimental data and mechanistic understanding presented in the patent literature, providing a reliable foundation for process development teams evaluating this technology for adoption. Engaging with these technical details early in the project lifecycle can help identify potential scale-up challenges and optimize the process for maximum efficiency.
Q: What are the key advantages of using nitro compounds over traditional amines in this synthesis?
A: Nitro compounds are significantly cheaper and more readily available than pre-activated amines or acid anhydrides. This method eliminates the need for harsh high-pressure carbon monoxide gas, utilizing solid Mo(CO)6 instead, which greatly enhances operational safety and simplifies the supply chain for raw materials.
Q: How does this method improve impurity control compared to conventional routes?
A: The one-pot cascade mechanism minimizes the isolation of unstable intermediates, reducing the formation of side products associated with multi-step workups. The specific use of the PdCl2/dppp catalyst system ensures high regioselectivity during the cyclization step, resulting in a cleaner crude profile that simplifies downstream purification.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the protocol uses standard organic solvents like dioxane and operates at atmospheric pressure regarding CO gas, making it highly scalable. The robustness of the catalyst system allows for broad substrate tolerance, meaning diverse analogues can be produced without re-optimizing the entire process for each new derivative.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into viable manufacturing processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite you to collaborate with us to leverage this cutting-edge carbonylation technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce time-to-market for your valuable therapeutic candidates.
