Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Manufacturing
The continuous evolution of organic synthesis methodologies is critical for the pharmaceutical industry to access novel chemical space efficiently. Patent CN113045503B, published in late 2022, introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, a privileged scaffold found in numerous bioactive molecules. This technology addresses long-standing challenges in heterocyclic chemistry by utilizing a transition metal palladium-catalyzed carbonylation cascade reaction. The significance of this development cannot be overstated, as quinazolinone derivatives are central to drugs exhibiting antifungal, antiviral, and anticancer properties, such as the sedative Methaqualone and the bioactive alkaloid Rutaecarpine shown in the structural overview below.
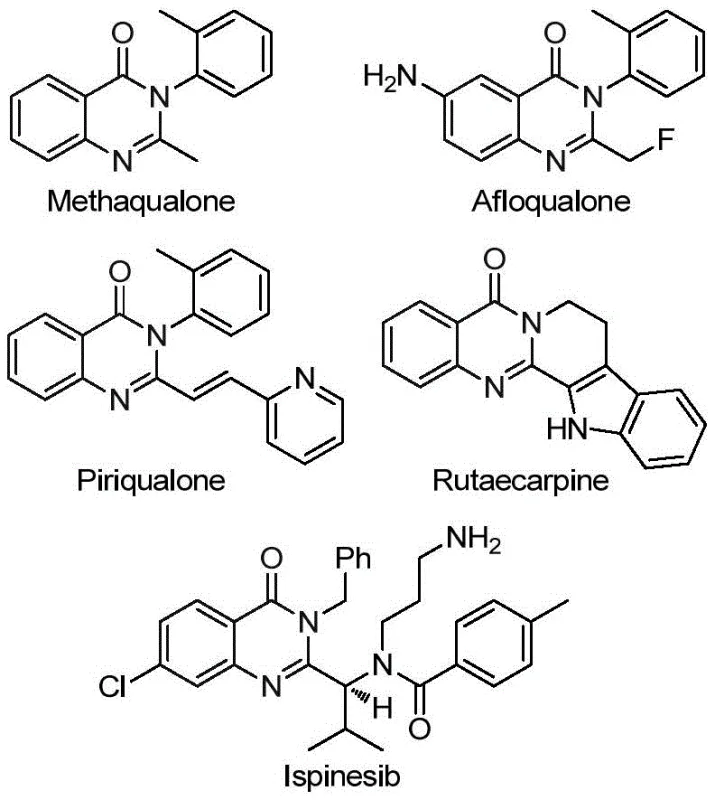
By leveraging cheap and readily available starting materials like trifluoroethylimidoyl chloride and various amines, this invention offers a robust pathway for industrial application. The method is not merely a laboratory curiosity but a scalable process designed to operate under relatively mild conditions compared to traditional routes. For R&D directors and process chemists, this represents a viable strategy to introduce trifluoromethyl groups, which are known to enhance metabolic stability and lipophilicity, into complex molecular architectures without compromising yield or purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl-substituted quinazolinones has been fraught with significant operational and economic hurdles that hinder large-scale production. Conventional literature methods often rely on the cyclization of anthranilamides with ethyl trifluoroacetate or trifluoroacetic anhydride, which frequently require harsh reaction conditions and expensive, moisture-sensitive reagents. Other approaches involve the use of unstable trifluoroacetamides or isatoic anhydrides, which suffer from narrow substrate scopes and low atom economy. Furthermore, methods promoted by coupling reagents like T3P can be prohibitively costly for kilogram-scale manufacturing due to the generation of stoichiometric waste and the difficulty in removing phosphorus-containing byproducts. These limitations collectively result in higher production costs, extended lead times, and increased environmental burden, making them less attractive for modern green chemistry initiatives.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a palladium-catalyzed carbonylation strategy that fundamentally reshapes the synthetic landscape for these heterocycles. By employing trifluoroethylimidoyl chloride and amines as primary building blocks, the method bypasses the need for pre-activated carboxylic acid derivatives. The core innovation lies in the use of TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide surrogate, which releases CO safely in situ upon heating. This eliminates the safety risks associated with handling high-pressure CO gas cylinders in a plant setting. As illustrated in the general reaction scheme below, the transformation proceeds efficiently in 1,4-dioxane at 110°C, delivering the target quinazolinones with excellent yields and broad functional group compatibility.
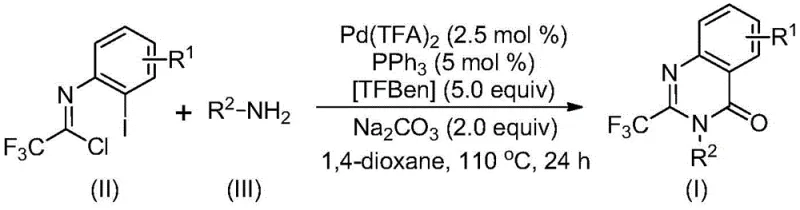
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is essential for process optimization and troubleshooting during scale-up. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond coupling between the imidoyl chloride and the amine, generating a trifluoroacetamidine derivative intermediate. Subsequently, the palladium catalyst, generated from Pd(TFA)2 and PPh3, inserts into the carbon-iodine bond of the aromatic ring to form a divalent palladium species. This step is crucial as it activates the aryl halide for the subsequent carbonylation event. The thermal decomposition of TFBen releases carbon monoxide directly into the reaction medium, which then inserts into the carbon-palladium bond to form an acyl-palladium intermediate.
Following CO insertion, the presence of base facilitates the formation of a palladium-nitrogen bond, leading to the construction of a seven-membered ring palladium intermediate. This cyclization step is the key determinant of the quinazolinone core structure. Finally, reductive elimination occurs to release the final 2-trifluoromethyl-substituted quinazolinone product and regenerate the active palladium catalyst for the next cycle. This elegant cascade minimizes the number of isolation steps required, thereby reducing material loss and solvent consumption. For quality control teams, understanding this mechanism helps in identifying potential side products, such as those arising from incomplete CO insertion or competitive hydrolysis, ensuring that stringent purity specifications are met consistently.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The practical execution of this synthesis is designed to be straightforward, requiring standard laboratory or pilot plant equipment without the need for specialized high-pressure reactors. The protocol involves charging a reactor with the palladium catalyst system, the solid CO source, and the organic substrates in an aprotic solvent. Maintaining the temperature at 110°C for a duration of 16 to 30 hours ensures complete conversion of the starting materials. Detailed standardized operating procedures regarding reagent addition rates, stirring speeds, and specific workup protocols are critical for reproducibility.
- Combine palladium trifluoroacetate, triphenylphosphine, TFBen, sodium carbonate, trifluoroethylimidoyl chloride, and amine in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 110°C and stir for 16 to 30 hours to allow the carbonylation cascade to proceed to completion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl-substituted quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative advantages that directly impact the bottom line and operational resilience. The shift towards using commercially available and inexpensive starting materials, such as simple amines and trifluoroethylimidoyl chlorides, drastically reduces the raw material cost basis compared to traditional routes relying on exotic reagents. Furthermore, the use of a solid CO surrogate like TFBen simplifies logistics and storage requirements, removing the regulatory and safety burdens associated with compressed toxic gases. This translates to a more agile supply chain capable of responding quickly to demand fluctuations without being bottlenecked by hazardous material handling protocols.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive coupling reagents and the use of a low-loading palladium catalyst system. By avoiding stoichiometric amounts of activators like T3P or anhydrides, the waste disposal costs are significantly lowered, contributing to substantial overall cost savings. Additionally, the high reaction efficiency and yields reported in the patent examples mean that less raw material is required to produce the same amount of product, further enhancing the cost-effectiveness of the manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is vital for maintaining uninterrupted supply to downstream API manufacturers. The broad substrate scope implies that a single manufacturing platform can be adapted to produce a variety of quinazolinone derivatives, reducing the need for multiple dedicated production lines. This flexibility allows for better inventory management and reduces the risk of stockouts for critical pharmaceutical intermediates, ensuring a steady flow of materials for drug development pipelines.
- Scalability and Environmental Compliance: The method has been successfully demonstrated on a gram scale in the patent, indicating strong potential for kilogram and tonne-scale production. The use of 1,4-dioxane as a solvent, while requiring careful recovery, is a well-established industrial solvent with existing recycling infrastructure. Moreover, the atom economy of the carbonylation cascade is superior to stepwise condensation methods, resulting in less chemical waste generation. This aligns with increasingly strict environmental regulations and corporate sustainability goals, making the process more attractive for long-term investment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is key to successful technology transfer and process validation.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a safe and effective carbon monoxide surrogate, releasing CO in situ under heating conditions. This eliminates the need for handling hazardous high-pressure CO gas, significantly improving operational safety and simplifying the equipment requirements for commercial scale-up.
Q: What is the substrate scope for the amine component in this reaction?
A: The method demonstrates excellent functional group tolerance, accommodating a wide range of amines including alkyl amines (n-Bu, t-Bu), benzyl amines, cycloalkyl amines, and substituted aryl amines. This versatility allows for the rapid generation of diverse quinazolinone libraries for drug discovery without needing extensive protocol optimization.
Q: Can this method be applied to the synthesis of complex drug molecules?
A: Yes, the patent explicitly demonstrates the successful application of this methodology in the efficient total synthesis of Rutaecarpine, a bioactive natural product. The process achieved a high overall yield through a streamlined three-step sequence, proving its viability for constructing complex heterocyclic scaffolds found in pharmaceuticals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN113045503B for accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory results are translated into reliable industrial reality. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite global partners to collaborate with us to leverage this cutting-edge technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on the foundation of the most efficient and cost-effective chemistry available.
