Revolutionizing Pharmaceutical Intermediates: Scalable Synthesis of Carbonyl-Bridged Biheterocyclic Compounds with Enhanced Purity and Process Efficiency
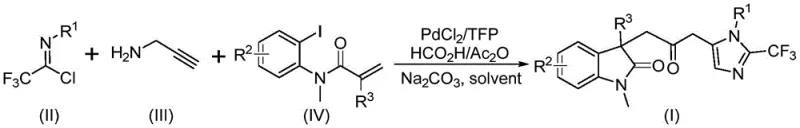
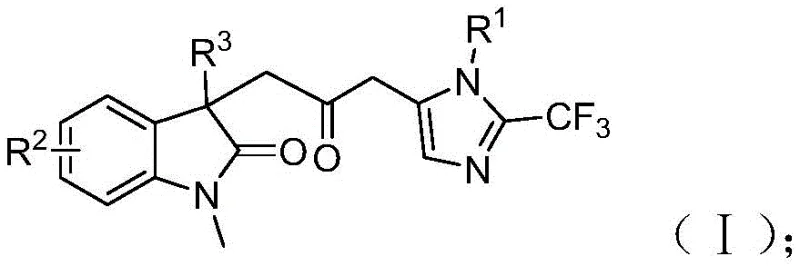
The patent CN115353511A introduces a groundbreaking multi-component synthesis methodology for carbonyl-bridged biheterocyclic compounds, representing a significant advancement in pharmaceutical intermediate manufacturing. This innovative approach addresses critical limitations in traditional carbonylation techniques by eliminating the requirement for toxic carbon monoxide gas while maintaining high reaction efficiency and substrate versatility. The process leverages readily available starting materials including trifluoroethylimidoyl chloride, propargylamine, and acrylamide under palladium catalysis to construct complex molecular architectures essential for bioactive compound development. By operating at mild conditions of 30°C with simple post-processing protocols, this method provides pharmaceutical manufacturers with a scalable pathway to produce high-value intermediates that are otherwise challenging to access through conventional routes. The patent demonstrates successful gram-scale implementation, establishing a robust foundation for commercial adoption in active pharmaceutical ingredient synthesis where structural complexity and purity are paramount considerations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing carbonyl-bridged biheterocyclic compounds face significant challenges including the mandatory use of toxic carbon monoxide gas under high-pressure conditions, which necessitates specialized equipment and stringent safety protocols that increase operational complexity and capital expenditure. Conventional methods often suffer from narrow substrate scope with poor functional group tolerance, limiting their applicability for diverse pharmaceutical intermediate production where structural variations are essential for structure-activity relationship studies. The requirement for pre-formed carbonyl sources frequently results in additional synthetic steps that reduce overall yield and increase impurity profiles, creating substantial hurdles for regulatory compliance in pharmaceutical manufacturing. Furthermore, many existing protocols demonstrate inconsistent performance when scaled beyond laboratory quantities due to exothermic reaction profiles and sensitivity to trace impurities, making commercial implementation economically unviable for most contract manufacturing organizations serving the pharmaceutical industry.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed cascade reaction that utilizes formic acid/acetic anhydride as a safe carbon monoxide surrogate, eliminating hazardous gas handling while maintaining high catalytic efficiency at ambient pressure. This innovation enables the direct construction of complex biheterocyclic frameworks through a one-pot multi-component reaction that simultaneously forms multiple chemical bonds with exceptional atom economy. The process demonstrates remarkable substrate flexibility with diverse R-group substitutions across all three components, allowing pharmaceutical manufacturers to rapidly access structurally varied intermediates without modifying the core reaction protocol. Operating under mild conditions of 30°C in standard tetrahydrofuran solvent with straightforward purification via column chromatography, this method provides exceptional scalability from laboratory to commercial production while maintaining consistent product quality and purity profiles required for pharmaceutical applications.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The reaction mechanism begins with oxidative addition of zero-valent palladium into the carbon-iodine bond of the acrylamide substrate, followed by intramolecular Heck-type cyclization that generates a key alkyl palladium intermediate. This intermediate undergoes carbonylation through carbon monoxide released in situ from the formic acid/acetic anhydride system, forming an acyl palladium species that serves as the critical electrophilic component for subsequent transformations. Concurrently, base-promoted coupling between trifluoroethylimidoyl chloride and propargylamine forms a trifluoroacetamidine intermediate that undergoes isomerization to generate the nucleophilic partner required for the final cyclization step. The acyl palladium species activates this intermediate through coordination, facilitating intramolecular cyclization that constructs the final biheterocyclic framework with precise regiocontrol. This cascade process demonstrates exceptional chemoselectivity by avoiding common side reactions such as homocoupling or β-hydride elimination that typically plague transition metal-catalyzed systems, resulting in cleaner reaction profiles and simplified purification requirements.
Impurity control is achieved through multiple mechanistic features inherent to this cascade process, including the precise stoichiometric control of reactive intermediates that minimizes dimerization or oligomerization side products commonly observed in similar systems. The mild reaction temperature of 30°C prevents thermal decomposition pathways that could generate colored impurities or degradation products problematic for pharmaceutical applications. The use of sodium carbonate as a mild base avoids strong alkaline conditions that might promote hydrolysis of sensitive functional groups present in the substrates or products. The catalytic system demonstrates excellent selectivity for the desired cyclization pathway over potential competing reactions such as direct carbonylation or alternative cyclization modes, resulting in consistently high purity profiles across diverse substrate combinations as evidenced by the comprehensive analytical data provided in the patent examples. This inherent selectivity significantly reduces the burden on downstream purification processes while ensuring stringent quality standards required for pharmaceutical intermediates.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
This innovative synthesis pathway represents a significant advancement in pharmaceutical intermediate manufacturing by providing a streamlined approach to complex molecular architectures previously requiring multiple synthetic steps. The methodology eliminates hazardous reagents while maintaining high efficiency and product quality through carefully optimized reaction parameters that balance reactivity with selectivity. The process demonstrates exceptional robustness across diverse substrate combinations, making it suitable for producing various structural analogs required during drug discovery and development phases. Detailed standardized synthesis procedures have been developed based on the patent methodology to ensure consistent results across different production scales, with specific protocols tailored to accommodate variations in starting material specifications while maintaining product quality standards. The following step-by-step guide provides essential operational parameters for successful implementation in pharmaceutical manufacturing environments.
- Prepare reaction mixture with palladium chloride catalyst, trifuryl phosphine ligand, and sodium carbonate base in tetrahydrofuran solvent under inert atmosphere
- Introduce trifluoroethylimidoyl chloride, propargylamine, and acrylamide substrates at controlled stoichiometric ratios with precise temperature management
- Conduct reaction at 30°C for 12-20 hours with continuous monitoring, followed by standard filtration and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial value to procurement and supply chain decision-makers by addressing critical pain points in pharmaceutical intermediate sourcing through innovative process design that enhances both economic and operational performance. The elimination of specialized gas handling requirements significantly reduces capital investment barriers while improving facility flexibility, allowing manufacturers to allocate resources toward value-added activities rather than safety infrastructure. The use of commercially available starting materials with established supply chains provides procurement teams with greater sourcing options and reduced dependency on single-source suppliers, enhancing overall supply chain resilience in volatile market conditions. These advantages collectively contribute to more predictable production timelines and improved resource allocation strategies that directly support business continuity planning for pharmaceutical manufacturers.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas handling requirements removes substantial infrastructure costs associated with specialized containment systems and safety protocols, while the use of inexpensive palladium chloride catalyst instead of more expensive alternatives provides significant material cost savings without compromising performance. The simplified one-pot reaction sequence reduces processing time and eliminates intermediate isolation steps that typically generate waste streams requiring costly disposal procedures, resulting in substantial operational cost reductions across the manufacturing value chain.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials with established global supply networks, this methodology minimizes vulnerability to single-source dependencies that often disrupt pharmaceutical manufacturing operations. The robust reaction profile tolerates minor variations in raw material quality without affecting final product specifications, providing procurement teams with greater flexibility in supplier selection while maintaining consistent output quality. This inherent process robustness translates directly to more reliable delivery schedules and reduced risk of production interruptions that could impact critical drug supply chains.
- Scalability and Environmental Compliance: The process demonstrates seamless scalability from laboratory to commercial production without requiring significant re-engineering, as evidenced by successful gram-scale implementation documented in the patent examples. The elimination of hazardous reagents and reduction in waste generation through atom-economical design aligns with increasingly stringent environmental regulations while reducing disposal costs. The simplified purification protocol using standard column chromatography techniques ensures consistent product quality at scale without requiring specialized equipment that could create bottlenecks in commercial manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations for implementing this patented methodology in pharmaceutical manufacturing operations, based on detailed analysis of the patent specifications and experimental data provided in the original disclosure. These insights reflect practical implementation considerations derived from the technical documentation while addressing common concerns raised by procurement, R&D, and supply chain stakeholders during technology evaluation processes. Each response is grounded in the specific experimental evidence and process parameters documented in the patent literature to ensure accuracy and relevance for decision-makers evaluating this technology for commercial adoption.
Q: How does this method eliminate the need for toxic carbon monoxide gas while maintaining high reaction efficiency?
A: The process utilizes formic acid/acetic anhydride as a safe CO surrogate that generates carbon monoxide in situ under controlled conditions, eliminating handling risks while maintaining catalytic efficiency through optimized palladium-trifuryl phosphine system.
Q: What substrate flexibility does this methodology offer for pharmaceutical intermediate production?
A: The system demonstrates exceptional functional group tolerance with diverse R-group substitutions on all three components, enabling synthesis of structurally varied biheterocyclic compounds with trifluoromethyl groups while maintaining high yields across multiple substrate combinations.
Q: How does this process ensure supply chain reliability for pharmaceutical manufacturers?
A: By utilizing commercially available starting materials with simplified purification protocols and eliminating specialized gas handling requirements, the method provides consistent production capability from laboratory scale to commercial manufacturing without complex infrastructure dependencies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required for pharmaceutical applications through state-of-the-art QC labs equipped with advanced analytical instrumentation. As a CDMO specialist with deep expertise in complex heterocyclic chemistry, we have successfully implemented similar palladium-catalyzed methodologies across multiple client projects, demonstrating our capability to translate innovative academic discoveries into robust commercial manufacturing processes. Our integrated approach combines cutting-edge process chemistry development with rigorous quality systems to ensure consistent delivery of high-purity intermediates that meet global regulatory requirements while optimizing cost structures through continuous process improvement initiatives.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis methodology can enhance your specific supply chain requirements. Contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs, enabling informed decision-making about integrating this advanced technology into your manufacturing portfolio.
