Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Pharmaceutical Applications
Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Pharmaceutical Applications
The landscape of modern pharmaceutical synthesis is constantly evolving, driven by the need for more efficient, safer, and cost-effective routes to complex molecular scaffolds. A significant breakthrough in this domain is detailed in patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. These structures, particularly those combining indolinone and imidazole motifs, are of paramount importance in medicinal chemistry due to their presence in numerous bioactive natural products and drug candidates. The disclosed technology represents a paradigm shift from traditional step-wise constructions to a streamlined, one-pot cascade reaction. By leveraging a palladium-catalyzed carbonylation strategy, this method not only simplifies the synthetic workflow but also eliminates the safety hazards associated with handling toxic carbon monoxide gas directly. For R&D directors and process chemists seeking reliable pharmaceutical intermediate suppliers, understanding the nuances of this catalytic system is crucial for developing next-generation therapeutic agents with improved purity profiles and reduced development timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic frameworks has been fraught with synthetic challenges that hinder rapid drug discovery and commercial manufacturing. Traditional approaches often rely on the direct coupling of two pre-formed heterocyclic substrates, a strategy that frequently suffers from low atom economy and poor regioselectivity. Alternatively, oxidative cyclization reactions involving bis-nucleophiles and activated methyl-substituted heterocycles require harsh oxidizing conditions that can degrade sensitive functional groups, leading to complex impurity profiles that are difficult to purge. Furthermore, conventional carbonylation reactions typically necessitate the use of high-pressure carbon monoxide cylinders, posing severe safety risks and requiring specialized, expensive reactor infrastructure that many contract manufacturing organizations lack. These limitations result in prolonged lead times, inflated production costs, and significant bottlenecks in the supply chain for high-value fine chemical intermediates.
The Novel Approach
In stark contrast, the methodology described in CN115353511A offers a transformative solution by employing a transition metal palladium-catalyzed carbonylation cascade reaction. This innovative approach utilizes cheap and readily available starting materials—specifically trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives—to construct the complex biheterocyclic core in a single operational step. The reaction proceeds under remarkably mild conditions, typically at 30°C, which preserves the integrity of sensitive functional groups and minimizes energy consumption. Crucially, the system generates the necessary carbon monoxide in situ through the decomposition of a formic acid and acetic anhydride mixture, thereby obviating the need for external CO gas sources. This not only enhances laboratory safety but also drastically simplifies the engineering requirements for scale-up, making it an ideal candidate for cost reduction in API manufacturing where safety and efficiency are paramount.
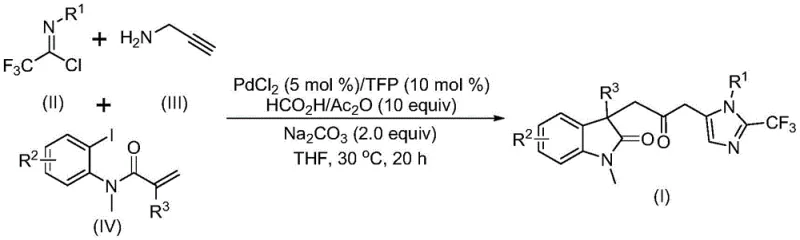
The mechanistic elegance of this transformation lies in its ability to forge multiple chemical bonds simultaneously. The process initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, followed by an intramolecular Heck reaction to generate a divalent alkyl-palladium intermediate. Subsequently, the in situ generated carbon monoxide inserts into the palladium-carbon bond to form an acyl-palladium species. Concurrently, a base-promoted intermolecular reaction between the trifluoroethylimidoyl chloride and propargylamine yields a trifluoroacetamidine intermediate, which undergoes isomerization. The final cyclization is triggered by the activation of this amidine by the acyl-palladium intermediate, closing the ring to afford the target carbonyl-bridged biheterocycle. This intricate dance of catalytic cycles ensures high reaction efficiency and excellent substrate compatibility, allowing for the introduction of diverse substituents such as halogens, alkyl groups, and trifluoromethyl moieties without compromising yield.
From an impurity control perspective, the mild reaction temperature of 30°C plays a pivotal role in suppressing side reactions such as polymerization of the acrylamide or decomposition of the imidoyl chloride. The use of tris(2-furyl)phosphine (TFP) as a ligand further stabilizes the palladium center, preventing the formation of palladium black and ensuring consistent catalytic turnover. For quality assurance teams, this translates to a cleaner crude reaction profile, which significantly reduces the burden on downstream purification processes like column chromatography. The ability to tolerate a wide range of functional groups means that late-stage functionalization strategies become feasible, allowing medicinal chemists to rapidly explore structure-activity relationships (SAR) around the biheterocyclic core. This level of control over the chemical outcome is essential for producing high-purity pharmaceutical intermediates that meet the stringent regulatory standards required for clinical trial materials.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Implementing this synthesis in a practical setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and reproducibility. The patent outlines a robust protocol where palladium chloride (5 mol%) and TFP (10 mol%) serve as the catalytic system, while sodium carbonate acts as the base to facilitate the deprotonation steps. The reaction is typically conducted in tetrahydrofuran (THF), which provides optimal solubility for all components and supports the catalytic cycle effectively. The molar ratio of substrates is critical, with a preferred ratio of trifluoroethylimidoyl chloride to propargylamine to acrylamide being approximately 1:2:1.5, ensuring that the more volatile or reactive amines are present in slight excess to drive the reaction to completion. Detailed standardized synthesis steps follow below.
- Combine palladium chloride (5 mol%), TFP ligand (10 mol%), sodium carbonate (2.0 equiv), and the formic acid/acetic anhydride CO source in THF solvent.
- Add trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide substrate to the reaction mixture under inert atmosphere.
- Stir the reaction at 30°C for 12-20 hours, then filter and purify the crude product via silica gel column chromatography to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary advantage lies in the substantial reduction of raw material costs, as the key building blocks—propargylamine and acrylamide derivatives—are commodity chemicals available in bulk quantities from multiple global suppliers. Unlike traditional methods that might require custom-synthesized, expensive heterocyclic precursors, this multi-component approach leverages simple, off-the-shelf reagents. Furthermore, the elimination of high-pressure carbon monoxide equipment removes a significant capital expenditure barrier, allowing for production in standard glass-lined or stainless steel reactors without the need for specialized autoclaves. This flexibility enhances supply chain resilience by enabling manufacturing across a broader network of qualified facilities, reducing the risk of single-source bottlenecks.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the convergence of low-cost inputs and simplified processing. By avoiding the use of toxic CO gas, the facility saves on the costs associated with gas handling safety systems, monitoring equipment, and specialized training for operators. Additionally, the mild reaction conditions (30°C) significantly lower energy consumption compared to high-temperature reflux protocols often seen in heterocycle synthesis. The high atom economy of the multi-component reaction means less waste generation, which in turn reduces the costs associated with waste disposal and environmental compliance. These factors collectively contribute to a leaner manufacturing cost structure, allowing for more competitive pricing of the final pharmaceutical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply continuity is a critical concern for any organization managing complex drug pipelines. This synthesis method mitigates supply risk by relying on widely available starting materials rather than niche, proprietary reagents that may have long lead times or limited supplier bases. The robustness of the reaction conditions also implies a lower sensitivity to minor variations in raw material quality, reducing the frequency of batch failures and reworks. Moreover, the scalability demonstrated in the patent, extending successfully to gram-scale and beyond, suggests that the process can be smoothly transferred from pilot plant to commercial production volumes. This scalability ensures that as demand for the final drug product grows, the supply of the intermediate can be ramped up rapidly to meet market needs without extensive process re-optimization.
- Scalability and Environmental Compliance: In an era of increasing regulatory scrutiny regarding environmental impact, the green chemistry attributes of this method are a significant asset. The in situ generation of carbon monoxide from formic acid and acetic anhydride avoids the release of fugitive emissions associated with gas cylinder usage. The use of THF as a solvent, while requiring proper recovery systems, is well-established in the industry with mature recycling technologies available. The high efficiency of the reaction minimizes the volume of solvent required per kilogram of product, further reducing the environmental footprint. For companies aiming to meet sustainability goals, adopting this pathway demonstrates a commitment to safer, cleaner chemical manufacturing practices, which can be a differentiator in tenders and partnerships with environmentally conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on reaction scope and operational parameters. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the critical reaction conditions for this carbonylation cascade?
A: The reaction operates optimally at mild temperatures around 30°C using THF as the solvent. It requires a specific PdCl2/TFP catalytic system and a formic acid/acetic anhydride mixture to generate carbon monoxide in situ, avoiding the need for toxic high-pressure CO gas.
Q: Can this method tolerate diverse functional groups on the substrates?
A: Yes, the protocol demonstrates excellent substrate compatibility. It successfully accommodates various substituents on the aryl rings, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens, trifluoromethyl, and nitro groups, yielding diverse biheterocyclic scaffolds.
Q: Is this process suitable for large-scale industrial production?
A: The patent explicitly highlights the scalability of the method, noting successful expansion to gram-scale reactions. The use of cheap, commercially available starting materials and the avoidance of hazardous gaseous CO make it highly viable for commercial scale-up in pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN115353511A for accelerating drug development programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the bench to the plant. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with precision, supported by rigorous QC labs that enforce stringent purity specifications to guarantee the quality of every batch. We understand that in the competitive landscape of pharmaceutical intermediates, consistency and reliability are non-negotiable, and our team is dedicated to delivering products that meet the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your specific pipeline needs. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your project, identifying opportunities to optimize your supply chain and reduce overall manufacturing expenses. Please contact our technical procurement team today to request specific COA data for related compounds and to discuss route feasibility assessments. Let us help you navigate the complexities of modern chemical synthesis and secure a reliable supply of high-quality intermediates for your future success.
