Advanced Industrial Synthesis of Agomelatine: Technical Breakthroughs and Commercial Scalability
The pharmaceutical landscape for central nervous system treatments continues to evolve, driven by the demand for more efficient and cost-effective manufacturing processes for key active ingredients. A pivotal development in this sector is detailed in patent CN101643434B, which discloses a novel industrial method for the synthesis of Agomelatine, also known chemically as N-[2-(7-methoxy-1-naphthyl)ethyl]acetamide. This compound is highly valued for its dual pharmacological profile as a melatonergic agonist and a 5-HT2C antagonist, making it critical for treating major depression and sleep disorders. The significance of this patent lies not merely in the molecule itself, but in the strategic re-engineering of its synthetic pathway to overcome historical bottlenecks associated with yield, purity, and scalability. By shifting the synthetic starting point to more accessible precursors, this technology offers a robust framework for cost reduction in pharmaceutical intermediates manufacturing, addressing the critical needs of R&D directors and procurement managers alike who seek reliable supply chains for complex CNS agents.
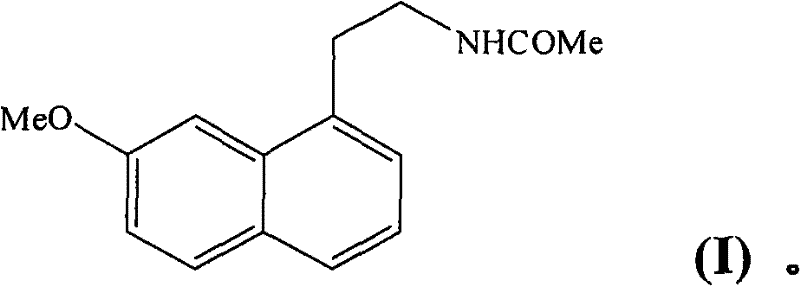
Historically, the industrial production of Agomelatine has faced significant challenges regarding step count and overall efficiency. Conventional methods, such as those described in earlier patents like EP 0 447 285, often relied on starting materials like 7-methoxy-1-tetralone. While functional, these legacy routes typically involved cumbersome eight-step sequences that resulted in average yields of less than 30 percent. Such low efficiency translates directly into higher waste generation, increased solvent consumption, and elevated production costs, creating friction in the supply chain. Furthermore, the necessity of performing aromatization steps on a tetralone core introduces additional complexity and potential safety hazards at scale. For a reliable pharmaceutical intermediates supplier, adhering to such inefficient pathways limits the ability to offer competitive pricing and guarantees of continuous supply, which are paramount for downstream drug manufacturers managing tight inventory schedules.
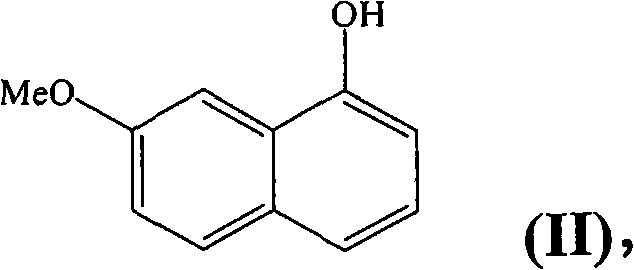
In stark contrast, the novel approach outlined in the present invention fundamentally alters the synthetic logic by utilizing 7-methoxy-1-naphthol as the primary starting material. This strategic shift offers immediate advantages because the naphthalene ring system is already present in the starting substrate, effectively bypassing the need for any aromatization reactions. This elimination of a chemically demanding step not only streamlines the process flow but also significantly enhances the safety profile of the manufacturing operation. The new route allows for the direct functionalization of the naphthalene core, converting the hydroxy group into a suitable leaving group such as a triflate or halide. This activation sets the stage for a highly efficient palladium-catalyzed coupling reaction. By adopting this methodology, manufacturers can achieve high-purity pharmaceutical intermediates with a drastically simplified workflow, ensuring that the final active ingredient meets stringent quality specifications without the need for laborious purification protocols that often plague older synthetic routes.
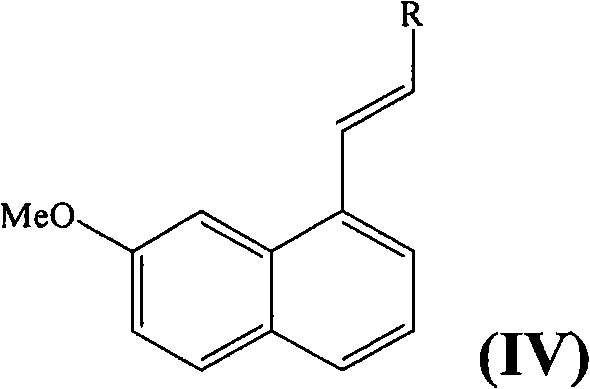
The mechanistic elegance of this synthesis is centered around the palladium-catalyzed condensation reaction, which serves as the cornerstone for constructing the carbon skeleton of Agomelatine. In this critical step, the activated naphthyl derivative reacts with a vinyl compound, such as N-vinylphthalimide or acrylamide, in the presence of a palladium catalyst like tetrakis(triphenylphosphine)palladium. The choice of catalyst and reaction conditions, typically involving reflux in toluene, is meticulously optimized to ensure complete control over regioselectivity. This precision is vital because any deviation could lead to the formation of structural isomers that are difficult to separate and could compromise the safety profile of the final drug. Following the coupling, the resulting vinyl intermediate undergoes catalytic hydrogenation, preferably using palladium on carbon, to saturate the double bond. This sequence demonstrates a sophisticated understanding of organometallic chemistry applied to industrial scale-up, ensuring that the commercial scale-up of complex pharmaceutical intermediates proceeds with minimal formation of unwanted by-products.
Following the construction of the carbon framework, the process moves towards the final functional group manipulations required to generate the active amine and subsequently the acetamide. The saturated intermediate is subjected to hydrolysis or treatment with a binary reducing agent and acid system, such as sodium borohydride and acetic acid, to reveal the primary amine functionality. This step is crucial as it converts the protected nitrogen species into the free amine necessary for the final acetylation. The use of mild yet effective reducing conditions ensures that the sensitive naphthalene ring system remains intact while efficiently cleaving the protecting groups. Finally, the free amine is treated with sodium acetate and acetic anhydride to yield the target Agomelatine. The entire sequence is designed to be telescoped where possible, minimizing isolation steps and solvent swaps. This attention to detail in the reaction design underscores the commitment to producing high-purity OLED material grade chemicals, although in this context, it specifically applies to pharmaceutical grades where impurity profiles are strictly regulated by global health authorities.
How to Synthesize Agomelatine Efficiently
The synthesis of Agomelatine via this novel pathway represents a significant leap forward in process chemistry, offering a clear roadmap for industrial implementation. The procedure begins with the activation of 7-methoxy-1-naphthol, followed by a palladium-catalyzed coupling with a vinyl derivative to establish the ethyl side chain precursor. Subsequent hydrogenation and deprotection steps unlock the amine functionality, which is finally acetylated to yield the target molecule. This streamlined approach not only improves overall yield but also enhances the reproducibility of the crystalline form of the product, a critical quality attribute for bioavailability. For technical teams looking to implement this route, the detailed standardized synthesis steps are provided below to ensure consistent results across different production batches.
- Convert 7-methoxy-1-naphthol into a reactive leaving group such as a triflate or halide to enable subsequent coupling.
- Perform a palladium-catalyzed condensation with a vinyl derivative like N-vinylphthalimide to form the carbon-carbon bond.
- Execute catalytic hydrogenation followed by hydrolysis and acetylation to yield the final Agomelatine product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for procurement managers and supply chain heads who are constantly balancing cost pressures with the need for supply security. The primary driver of value here is the substitution of expensive and multi-step precursors with 7-methoxy-1-naphthol, a commodity chemical that is simpler and significantly less costly to source in bulk quantities. This fundamental change in the bill of materials directly impacts the bottom line, allowing for substantial cost savings that can be passed down the supply chain or reinvested into further R&D. Moreover, the elimination of the aromatization step removes a major bottleneck that often causes delays and variability in production schedules. By simplifying the process flow, manufacturers can achieve faster turnaround times and more predictable output, which is essential for maintaining reducing lead time for high-purity pharmaceutical intermediates in a competitive global market.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the synthetic route. By removing the need for aromatization and reducing the total number of unit operations, the consumption of energy, solvents, and reagents is significantly lowered. Additionally, the use of cheaper starting materials reduces the raw material cost base, while the high selectivity of the palladium-catalyzed steps minimizes the loss of valuable intermediates to side reactions. This cumulative effect results in a much more lean manufacturing process, enabling the production of Agomelatine at a fraction of the cost associated with legacy methods, thereby enhancing the overall margin profile for the final drug product.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the robustness of the underlying chemistry. This new method enhances reliability by utilizing widely available starting materials that are less susceptible to market volatility compared to specialized tetralone derivatives. Furthermore, the process operates under conditions that are easier to control and scale, reducing the risk of batch failures due to sensitive reaction parameters. The ability to consistently produce the correct crystalline form without extensive recrystallization steps means that inventory can be moved faster through quality control, ensuring that customers receive their orders on time and without the risk of specification deviations that could halt their own production lines.
- Scalability and Environmental Compliance: As regulatory scrutiny on environmental impact intensifies, the greener profile of this synthesis becomes a key competitive advantage. The reduction in step count inherently leads to a lower E-factor, meaning less waste is generated per kilogram of product. Avoiding harsh aromatization conditions also reduces the burden on waste treatment facilities and lowers the risk of emitting hazardous volatiles. This alignment with green chemistry principles facilitates smoother regulatory approvals and permits for expansion, allowing manufacturers to scale up production from pilot plants to multi-ton commercial facilities with greater ease and confidence, ensuring long-term supply continuity for global pharmaceutical partners.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its potential for integration into their supply networks. The following questions address common inquiries regarding the process capabilities, quality attributes, and operational feasibility. These answers are derived directly from the technical disclosures within the patent documentation, providing a transparent view of what this technology can deliver in a real-world manufacturing environment. Whether you are concerned about impurity profiles or the scalability of the palladium steps, the following insights clarify the operational strengths of this novel approach.
Q: What is the primary advantage of using 7-methoxy-1-naphthol over 7-methoxy-1-tetralone?
A: Using 7-methoxy-1-naphthol eliminates the need for an aromatization step, which is historically problematic and energy-intensive in industrial settings, thereby simplifying the process and reducing costs.
Q: How does this process ensure high regioselectivity during the coupling reaction?
A: The process utilizes specific palladium catalysts, such as tetrakis(triphenylphosphine)palladium, under controlled reflux conditions in toluene, which allows for complete control over regioselectivity during the coupling with formula (III) compounds.
Q: Is the crystalline form of the produced Agomelatine consistent with pharmacopeial standards?
A: Yes, the compound obtained via this method reproducibly exhibits the specific crystalline form properties described in patent EP1564202, ensuring it meets the rigorous physical specifications required for pharmaceutical active ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Agomelatine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and a proven track record in process optimization. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101643434B are fully realized in practical manufacturing scenarios. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Agomelatine meets the highest international standards. Our commitment to excellence extends beyond mere compliance; we actively work with our clients to refine processes for maximum efficiency and sustainability, positioning us as a trusted ally in the complex landscape of pharmaceutical intermediate production.
We invite global pharmaceutical companies and contract manufacturers to engage with us to explore how this advanced synthesis route can benefit their specific projects. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis tailored to your volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can build a resilient and cost-effective supply chain that supports the delivery of life-saving medications to patients worldwide, ensuring that the promise of innovative chemistry translates into tangible commercial success.
