Advanced Temperature-Regulated Synthesis of 1,4-Dithiine and Thiophene Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways to access complex heterocyclic scaffolds, particularly those containing sulfur which are pivotal in drug design and electronic materials. Patent CN112442008A introduces a groundbreaking methodology for the preparation of 1,4-dithiine and thiophene compounds by precisely regulating the reaction temperature of elemental sulfur and active internal alkynes. This innovation represents a significant departure from traditional synthetic routes that often rely on harsh conditions and expensive metal catalysts, offering instead a green, economical, and highly selective approach driven by simple base induction. The ability to toggle between six-membered 1,4-dithiine rings and five-membered thiophene rings solely through thermal control provides chemists with unprecedented flexibility in molecular design. Furthermore, the process utilizes elemental sulfur, an abundant and low-cost byproduct of the petroleum refining industry, thereby addressing both economic and environmental concerns associated with sulfur waste accumulation. This report analyzes the technical merits of this patent to demonstrate its value for large-scale manufacturing and supply chain optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,4-dithiine and related sulfur-containing heterocycles has been plagued by significant technical hurdles that impede efficient commercial production. Conventional methods typically necessitate the use of transition metal catalysts, which not only inflate the raw material costs but also introduce complex downstream processing requirements to remove trace metal residues to meet stringent pharmaceutical purity standards. These traditional routes often suffer from harsh reaction conditions, including extreme temperatures or pressures, which can compromise the stability of sensitive functional groups and lead to poor selectivity. Additionally, the structural diversity achievable through older methods is often limited, restricting the chemical space available for medicinal chemists to explore. The reliance on specialized reagents and the generation of hazardous waste streams further exacerbate the environmental footprint of these legacy processes, making them increasingly untenable in a regulatory landscape that demands greener chemistry. Consequently, there is a critical industry need for a synthesis platform that eliminates metal dependency while expanding structural accessibility.
The Novel Approach
The methodology disclosed in CN112442008A offers a transformative solution by leveraging temperature regulation and base induction to drive the cyclization of elemental sulfur and activated alkynes. This metal-free approach operates under mild conditions, utilizing common bases such as potassium hydroxide or triethylamine in polar aprotic solvents like dimethyl sulfoxide. A key advantage of this novel route is its operational simplicity and safety, as it avoids the handling of pyrophoric metal reagents and allows for straightforward workup procedures involving standard aqueous extraction and chromatography. The process demonstrates excellent atom economy by incorporating elemental sulfur directly into the heterocyclic framework, turning an industrial waste product into a high-value chemical intermediate. Moreover, the method exhibits remarkable versatility, accommodating a wide range of substituents including esters, ketones, aryl groups, and trifluoromethyl moieties without compromising yield or selectivity. By simply adjusting the reaction temperature, manufacturers can selectively produce either 1,4-dithiine or thiophene derivatives from the same starting materials, streamlining inventory management and production planning.
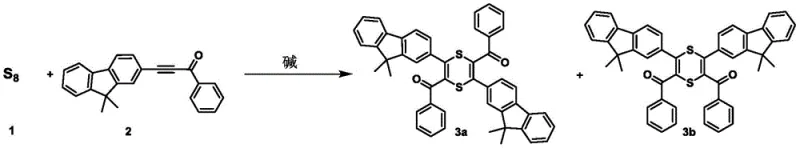
Mechanistic Insights into Temperature-Regulated Cyclization and Conversion
The core innovation of this technology lies in the precise thermodynamic control exerted over the reaction pathway, which dictates the formation of specific heterocyclic architectures. At lower temperatures ranging from 0°C to 60°C, the reaction kinetics favor the formation of the six-membered 1,4-dithiine ring system through a base-induced cyclization mechanism. However, when the temperature is elevated to the range of 61°C to 150°C, the system undergoes a rearrangement or direct cyclization to form the aromatic thiophene ring, which is thermodynamically more stable under these conditions. This temperature-dependent selectivity is a powerful tool for process chemists, allowing for the targeted synthesis of specific isomers without the need for protecting group strategies or additional synthetic steps. The patent further elucidates a unique conversion pathway where pre-formed 1,4-dithiine compounds can be thermally converted into thiophene derivatives, providing a secondary route to access the five-membered heterocycles if needed. This mechanistic flexibility ensures that the production process can be adapted dynamically to meet changing market demands for different sulfur-containing scaffolds.
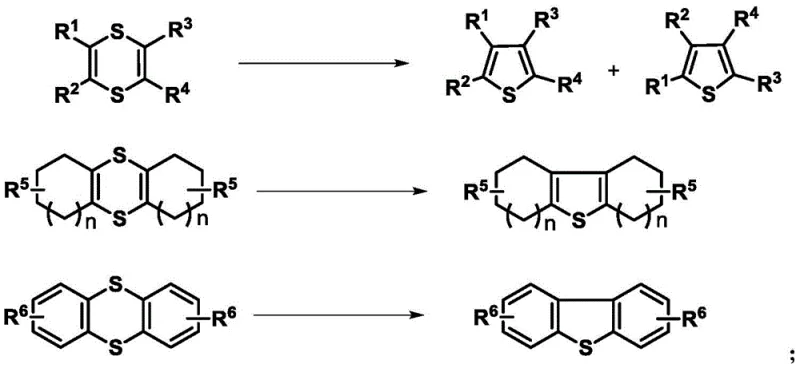
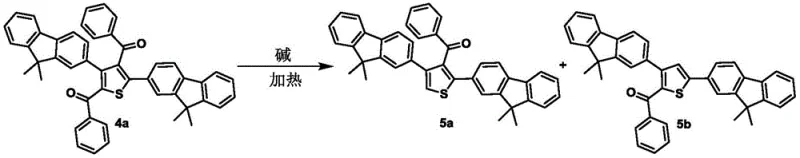
Beyond the ring formation, the patent describes a sophisticated method for modifying the substitution pattern of the resulting thiophene compounds through controlled carbon-carbon bond cleavage. Specifically, benzoyl-substituted thiophenes can undergo selective cleavage of the benzoyl group under high-temperature and alkaline conditions. This reaction enables the efficient removal of specific acyl groups, realizing a green and highly selective way to modify the molecular structure post-cyclization. Such a capability is rare in heterocyclic chemistry, where modifying the core substitution pattern often requires de novo synthesis. The ability to strip benzoyl groups efficiently expands the utility of the synthesized intermediates, allowing for the rapid generation of diverse libraries of thiophene derivatives for biological screening. This level of control over the molecular architecture underscores the robustness of the chemical platform and its potential for generating complex, high-value intermediates for pharmaceutical applications.
How to Synthesize 1,4-Dithiine Efficiently
The synthesis of 1,4-dithiine compounds via this patented method is characterized by its straightforward operational protocol, which is highly amenable to scale-up in a GMP environment. The process begins with the combination of activated alkyne substrates, sublimed elemental sulfur, and a suitable base in a reaction vessel, followed by the addition of a solvent such as DMSO or DMF. The reaction mixture is then heated in an oil bath at a controlled temperature between 0°C and 60°C for a duration of 0.5 to 6 hours, depending on the specific reactivity of the alkyne substrate. Upon completion, the reaction is quenched with saturated sodium chloride solution, and the product is extracted into an organic phase, dried, and purified using standard column chromatography techniques. This streamlined workflow minimizes the number of unit operations required, thereby reducing labor costs and potential points of failure in the manufacturing process. For a detailed breakdown of the specific molar ratios, solvent volumes, and purification parameters, please refer to the standardized synthesis guide below.
- Combine activated alkyne, sublimed elemental sulfur, and a base such as potassium hydroxide in a reaction vessel with a polar aprotic solvent like DMSO.
- Maintain the reaction temperature between 0°C and 60°C for 0.5 to 6 hours to facilitate the formation of the 1,4-dithiine ring structure.
- Quench the reaction with saturated NaCl solution, extract the organic phase with dichloromethane, dry over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this metal-free synthesis technology offers substantial strategic advantages that directly impact the bottom line and operational resilience. The elimination of transition metal catalysts removes a significant cost driver from the bill of materials, as precious metals like palladium or platinum are not only expensive but also subject to volatile market pricing and supply constraints. Furthermore, the absence of heavy metals simplifies the purification process, drastically reducing the consumption of scavenging resins and specialized filtration media that are typically required to meet residual metal specifications in API manufacturing. This simplification translates into shorter cycle times and higher throughput, enhancing the overall efficiency of the production facility. The use of elemental sulfur, a widely available and inexpensive commodity chemical, ensures a stable and secure supply of raw materials, mitigating the risk of shortages that can plague specialized reagent supply chains. Additionally, the mild reaction conditions and simple workup procedures reduce energy consumption and waste generation, aligning with corporate sustainability goals and reducing the costs associated with environmental compliance and waste disposal.
- Cost Reduction in Manufacturing: The replacement of expensive metal catalysts with inexpensive elemental sulfur and common bases results in a significant reduction in raw material costs, while the simplified purification process lowers operational expenditures related to consumables and waste treatment. The high atom economy of the reaction ensures that a greater proportion of the input materials are converted into the desired product, minimizing waste and maximizing yield efficiency. By avoiding the need for complex ligand systems and inert atmosphere handling often required for metal catalysis, the process further reduces infrastructure and maintenance costs. These cumulative savings contribute to a more competitive cost structure for the final sulfur-containing intermediates, providing a distinct market advantage.
- Enhanced Supply Chain Reliability: Utilizing elemental sulfur, a major byproduct of the global petroleum refining industry, ensures a robust and continuous supply of the key sulfur source, insulating the manufacturing process from the supply chain disruptions often associated with specialized fine chemicals. The reliance on common solvents and bases, which are readily available from multiple suppliers, further diversifies the supply base and reduces dependency on single-source vendors. The operational simplicity of the method allows for easier technology transfer between manufacturing sites, facilitating geographic diversification of production capacity to mitigate regional risks. This reliability is crucial for maintaining consistent delivery schedules to downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of hazardous metal reagents make this process inherently safer and easier to scale from laboratory to commercial production volumes without significant engineering challenges. The green nature of the synthesis, characterized by low toxicity reagents and reduced waste generation, simplifies regulatory compliance and permitting processes in jurisdictions with strict environmental regulations. The ability to recycle solvents and the minimal generation of hazardous byproducts contribute to a lower environmental footprint, supporting the company's commitment to sustainable manufacturing practices. This scalability ensures that the technology can meet growing market demand for sulfur heterocycles without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this temperature-regulated synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this method into their existing synthetic workflows. The responses cover aspects ranging from reaction selectivity to the scope of applicable substrates, ensuring a comprehensive overview of the technology's potential.
Q: What are the advantages of using elemental sulfur over metal catalysts for thiophene synthesis?
A: Using elemental sulfur eliminates the need for expensive and toxic transition metal catalysts, significantly reducing raw material costs and simplifying the purification process by removing heavy metal residue concerns.
Q: How does temperature regulation affect the product distribution between 1,4-dithiine and thiophene?
A: Lower temperatures (0-60°C) favor the formation of 1,4-dithiine compounds, while higher temperatures (61-150°C) drive the conversion towards thiophene derivatives, allowing precise control over the final heterocyclic structure.
Q: Can this method be applied to the synthesis of bioactive molecules?
A: Yes, the resulting sulfur-containing heterocycles possess good biological activity and are suitable intermediates for drug synthesis and natural product construction, as highlighted in the patent background.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Dithiine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the temperature-regulated synthesis method described in CN112442008A for producing high-quality sulfur-containing heterocycles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this metal-free process, including precise temperature control and efficient solvent recovery systems. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1,4-dithiine or thiophene intermediate meets the highest standards required for pharmaceutical and electronic applications. Our commitment to quality and consistency makes us the ideal partner for companies looking to leverage this innovative chemistry for their product pipelines.
We invite you to collaborate with us to explore the full commercial potential of this green synthesis technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free route for your target molecules. We encourage you to contact our technical procurement team to request specific COA data for our sulfur heterocycle portfolio and to discuss route feasibility assessments tailored to your development timeline. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain and deep technical expertise that will accelerate your path to market.
