Advanced Metal-Free Synthesis of 1,4-Dithiine and Thiophene Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing sulfur-containing heterocycles, which serve as critical scaffolds in bioactive molecules and electronic materials. Patent CN112442008B introduces a groundbreaking temperature-regulated synthesis method that utilizes elemental sulfur and active internal alkynes to produce 1,4-dithiine and thiophene derivatives without the need for transition metal catalysts. This innovation addresses significant environmental and economic pain points by leveraging abundant industrial sulfur by-products and simplifying purification workflows through alkali induction. The ability to toggle between 1,4-dithiine and thiophene structures solely through thermal control offers unprecedented flexibility for process chemists aiming to optimize impurity profiles and reaction efficiency. 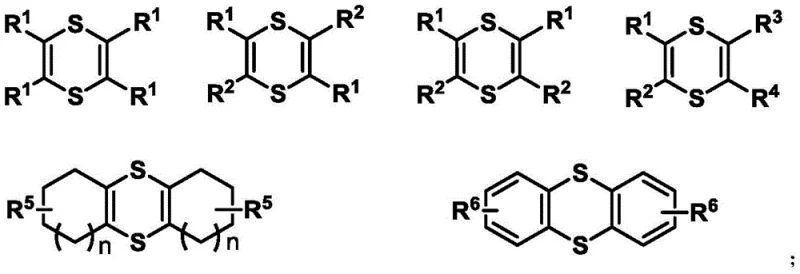
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for accessing complex sulfur-containing heterocycles often rely heavily on transition metal catalysis, which introduces substantial downstream processing burdens and cost inefficiencies. The presence of metal residues necessitates rigorous purification steps, such as scavenging or recrystallization, to meet stringent regulatory limits for pharmaceutical intermediates, thereby increasing production lead times and operational expenses. Furthermore, many conventional methods suffer from harsh reaction conditions, limited substrate scope, and poor atom economy, resulting in significant waste generation and reduced overall yields. These factors collectively hinder the commercial viability of producing high-purity sulfur heterocycles at an industrial scale, creating a bottleneck for supply chains dependent on these critical building blocks.
The Novel Approach
In stark contrast, the methodology disclosed in CN112442008B employs a metal-free, base-induced strategy that significantly streamlines the synthetic pathway while enhancing environmental sustainability. By utilizing elemental sulfur as a readily available and cost-effective reagent, this process eliminates the dependency on precious metal catalysts, thereby removing the associated costs of catalyst procurement and heavy metal removal. The reaction operates under mild conditions with excellent selectivity, allowing for the direct formation of target compounds with minimal by-product formation. This approach not only reduces the complexity of the workup procedure but also aligns with green chemistry principles, making it an attractive option for manufacturers seeking to reduce their carbon footprint and operational overhead.
Mechanistic Insights into Temperature-Regulated Cyclization
The core innovation of this technology lies in the precise thermal regulation that dictates the reaction trajectory between 1,4-dithiine and thiophene formation. At lower temperatures ranging from 0°C to 60°C, the reaction favors the cyclization of activated alkynes with elemental sulfur to form 1,4-dithiine compounds, which possess a unique non-aromatic boat-shaped structure. 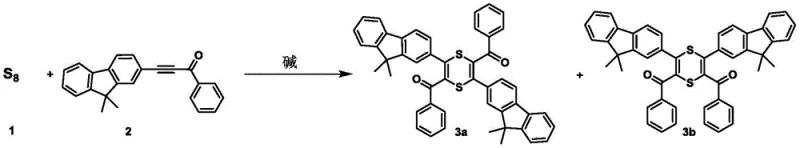 Upon increasing the temperature to the 61°C to 150°C range, the system undergoes a thermodynamic transformation where the 1,4-dithiine intermediate converts into the more stable aromatic thiophene ring system. This temperature-dependent selectivity provides chemists with a powerful tool to access diverse chemical spaces from the same starting materials simply by adjusting the thermal parameters of the reaction vessel.
Upon increasing the temperature to the 61°C to 150°C range, the system undergoes a thermodynamic transformation where the 1,4-dithiine intermediate converts into the more stable aromatic thiophene ring system. This temperature-dependent selectivity provides chemists with a powerful tool to access diverse chemical spaces from the same starting materials simply by adjusting the thermal parameters of the reaction vessel.
Furthermore, the process demonstrates remarkable versatility in handling various functional groups, including esters, ketones, and aryl substituents, without compromising yield or purity. The mechanism involves an alkali-induced activation of the alkyne, facilitating nucleophilic attack on elemental sulfur followed by cyclization. 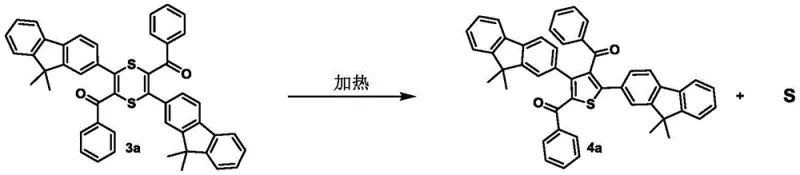 This pathway avoids the formation of complex organometallic intermediates, thereby reducing the risk of side reactions and ensuring a cleaner reaction profile. The ability to subsequently cleave carbon-carbon bonds under alkaline conditions further expands the utility of these compounds, allowing for the modular construction of specialized thiophene derivatives required in advanced material science and drug discovery applications.
This pathway avoids the formation of complex organometallic intermediates, thereby reducing the risk of side reactions and ensuring a cleaner reaction profile. The ability to subsequently cleave carbon-carbon bonds under alkaline conditions further expands the utility of these compounds, allowing for the modular construction of specialized thiophene derivatives required in advanced material science and drug discovery applications.
How to Synthesize 1,4-Dithiine Efficiently
To implement this synthesis effectively, operators must adhere to strict stoichiometric ratios and solvent selections as outlined in the patent examples. The process typically involves dissolving activated alkynes and elemental sulfur in polar aprotic solvents like DMSO or DMF, followed by the addition of a base such as potassium hydroxide or triethylamine. Detailed standardized synthesis steps see the guide below.
- Mix activated alkyne, sublimed sulfur, and an organic base such as potassium hydroxide or triethylamine in a polar aprotic solvent like DMSO.
- Control reaction temperature precisely: maintain 0-60°C for 1,4-dithiine formation or increase to 61-150°C for direct thiophene synthesis.
- Quench with saturated NaCl, extract with dichloromethane, dry over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route presents compelling opportunities for cost optimization and risk mitigation. The elimination of transition metal catalysts removes a significant variable cost component and simplifies the supply chain by reducing reliance on volatile precious metal markets. Additionally, the use of elemental sulfur, an abundant petroleum refining by-product, ensures a stable and low-cost raw material base that is less susceptible to geopolitical supply disruptions compared to specialized organometallic reagents.
- Cost Reduction in Manufacturing: The absence of expensive metal catalysts directly translates to lower raw material costs and eliminates the need for specialized metal scavenging resins or extensive purification protocols. This streamlined workflow reduces solvent consumption and energy usage during the workup phase, leading to substantial operational savings. By simplifying the production process, manufacturers can achieve higher throughput rates with existing infrastructure, effectively lowering the cost per kilogram of the final high-purity intermediate without compromising quality standards.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals like elemental sulfur and common organic bases enhances supply chain resilience by diversifying the vendor base and reducing lead times for raw material acquisition. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is less sensitive to minor fluctuations in reagent quality compared to sensitive catalytic systems. This reliability ensures consistent delivery timelines for downstream clients, minimizing the risk of production stoppages due to material shortages or quality failures in the upstream supply network.
- Scalability and Environmental Compliance: The green nature of this synthesis, characterized by high atom economy and the absence of toxic heavy metals, facilitates easier regulatory approval and environmental compliance across different jurisdictions. Scaling this process from laboratory to commercial production is straightforward due to the simplicity of the reaction setup and the use of standard industrial solvents. This scalability supports the growing demand for sulfur-containing heterocycles in the pharmaceutical and electronic sectors while maintaining a sustainable manufacturing profile that aligns with corporate ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations for potential partners.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process CN112442008B utilizes a metal-free approach induced by organic or inorganic bases, eliminating the need for costly palladium or copper catalysts and subsequent heavy metal removal steps.
Q: How is selectivity between 1,4-dithiine and thiophene controlled?
A: Selectivity is strictly governed by temperature regulation; lower temperatures (0-60°C) favor 1,4-dithiine formation, while higher temperatures (61-150°C) drive the conversion to thiophene compounds.
Q: Is elemental sulfur a viable raw material for large-scale production?
A: Yes, elemental sulfur is an abundant industrial by-product from petroleum refining, making it a highly economical and sustainable feedstock for the commercial scale-up of these sulfur-containing heterocycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Dithiine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative, metal-free synthetic routes like CN112442008B to enhance product portfolios and operational efficiency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries are translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1,4-dithiine or thiophene intermediate meets the exacting standards required by global pharmaceutical and electronic material clients.
We invite you to collaborate with us to leverage this technology for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain objectives and drive value for your organization.
