Unlocking Scalable Production of High-Purity Thiophene Intermediates Through Temperature-Controlled Metal-Free Synthesis Technology
Patent CN112442008B introduces a groundbreaking methodology for synthesizing sulfur-containing heterocyclic compounds through precise temperature regulation of elemental sulfur and active internal alkynes without any transition metal catalysts. This innovation leverages alkali induction to establish a green pathway for producing both 1,4-dithiine and thiophene derivatives under remarkably mild conditions ranging from 0°C to 150°C with exceptional selectivity across diverse substrate classes including aryl alkynes and fluorinated derivatives. The resulting compounds exhibit significant biological activity making them highly valuable for pharmaceutical applications including drug discovery and natural product synthesis where high purity standards are non-negotiable requirements. This patent represents a strategic advancement in sustainable chemistry that directly addresses critical industry challenges in cost efficiency while maintaining stringent quality specifications essential for pharmaceutical intermediates manufacturing operations worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing sulfur-containing heterocycles typically rely on transition metal catalysts such as palladium or copper complexes which introduce substantial challenges in pharmaceutical manufacturing environments requiring extensive purification steps to remove toxic metal residues that could compromise final product purity specifications demanded by regulatory authorities worldwide. These methods often necessitate stringent anhydrous conditions elevated temperatures exceeding 200°C and complex multi-step procedures that significantly increase production costs while generating hazardous waste streams requiring specialized disposal protocols that conflict with modern sustainability initiatives adopted by leading pharmaceutical companies globally.
The Novel Approach
The patented methodology overcomes these limitations through an elegant metal-free system utilizing elemental sulfur and activated alkynes under alkali induction at precisely controlled temperatures between 0°C and 150°C enabling selective formation of either dithiine compounds at lower temperatures or direct thiophene derivatives at elevated temperatures without requiring any transition metal catalysts whatsoever thus eliminating all associated purification costs environmental concerns and regulatory hurdles related to heavy metal contamination in final products destined for pharmaceutical applications where even trace amounts could trigger batch rejection during quality control inspections.
Mechanistic Insights into Alkali-Mediated Metal-Free Conversion
The fundamental mechanism involves alkali-induced activation of elemental sulfur followed by nucleophilic attack on activated alkyne substrates where at lower temperatures between 0°C and 60°C this process forms stable six-membered dithiine ring structures through stepwise addition mechanisms involving sequential sulfur atom incorporation across triple bonds under mild conditions with excellent stereoselectivity due to conformational constraints imposed by developing ring systems that prevent unwanted side reactions commonly observed in traditional catalytic approaches requiring harsher reaction environments that degrade sensitive functional groups present in complex pharmaceutical intermediates.
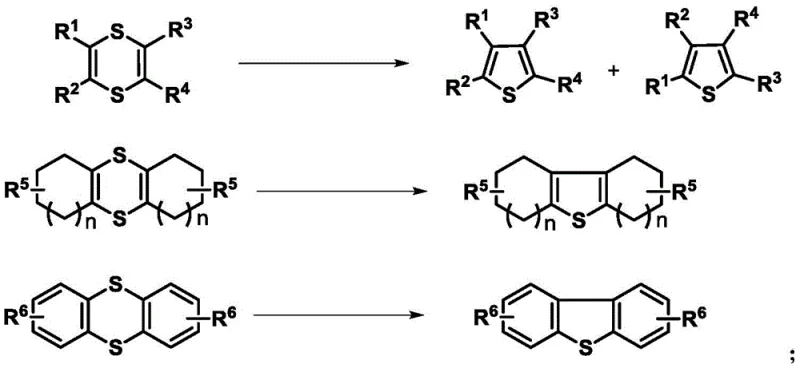
Impurity control is achieved through precise temperature modulation which governs reaction pathway selectivity where elevated temperatures between 61°C and 150°C trigger spontaneous thermal rearrangement of initially formed dithiine intermediates into more stable five-membered thiophene ring systems via concerted ring contraction mechanisms that maintain high regioselectivity without requiring additional reagents or catalysts thus minimizing byproduct formation through well-defined mechanistic pathways that avoid radical intermediates responsible for polymerization or decomposition issues plaguing conventional synthetic routes used in pharmaceutical intermediate manufacturing operations worldwide.
How to Synthesize Thiophene Derivatives Efficiently
This innovative synthesis route represents a significant advancement in producing complex sulfur-containing heterocycles for pharmaceutical applications where purity requirements exceed industry standards by enabling precise control over product formation through simple temperature modulation without requiring expensive catalysts or specialized equipment thus providing substantial process optimization opportunities while maintaining high product quality essential for regulatory compliance in global pharmaceutical markets where even minor impurities can trigger costly batch rejections during quality assurance inspections conducted by major multinational drug manufacturers seeking reliable supply partners capable of meeting stringent specifications consistently across all production scales from laboratory validation through full commercial implementation.
- Combine activated alkyne substrate at 0.1-2 mol/L concentration with elemental sulfur (molar ratio 1: 2-6) and alkali base (molar ratio 0.5-3) in organic solvents like DMSO under inert atmosphere.
- Precisely control reaction temperature between 61°C and 150°C using oil bath systems while monitoring progress via TLC until completion within typical timeframe of 0.5 to 6 hours.
- Execute standard workup procedure by adding saturated NaCl solution followed by dichloromethane extraction; dry organic phase with anhydrous magnesium sulfate before column chromatography purification using optimized solvent mixtures.
Commercial Advantages for Procurement and Supply Chain Teams
The patented temperature-regulated synthesis methodology delivers substantial operational benefits that directly address critical pain points in pharmaceutical supply chain management by eliminating transition metal catalysts from production processes thereby removing significant cost drivers associated with catalyst procurement handling and removal while simultaneously enhancing product purity profiles essential for regulatory compliance across global markets where quality standards continue to tighten requiring manufacturers to demonstrate complete traceability from raw materials through final product delivery without any contamination risks that could jeopardize client relationships with major pharmaceutical companies demanding absolute reliability in their supply networks.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts represents a major cost-saving opportunity through reduced capital expenditure requirements since standard manufacturing equipment can be utilized without modifications while avoiding extensive chromatography needed to remove metal residues below regulatory limits thus significantly lowering operational expenses associated with solvent consumption waste disposal and specialized personnel training required for handling air-sensitive catalysts that often lead to batch failures during scale-up operations.
- Enhanced Supply Chain Reliability: Reliance on readily available starting materials including elemental sulfur as an abundant petroleum refining byproduct ensures consistent raw material availability regardless of geopolitical supply chain disruptions while robust reaction conditions tolerate minor variations in starting material quality maintaining high product consistency that reduces batch rejection rates commonly experienced with traditional synthetic routes requiring highly purified precursors thus enabling faster response times to changing market demands without compromising quality standards required by global pharmaceutical clients.
- Scalability and Environmental Compliance: Compatibility with standard manufacturing equipment operating under ambient pressure conditions facilitates seamless scale-up from pilot plant to full commercial production without substantial capital investment while absence of toxic metals significantly reduces environmental impact simplifying waste treatment procedures to meet increasingly stringent regulatory requirements thus supporting corporate sustainability initiatives through higher atom economy compared to conventional methods requiring multiple protection/deprotection steps that generate excessive waste streams during pharmaceutical intermediate manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations regarding implementation of this innovative synthesis methodology in commercial pharmaceutical manufacturing environments based on detailed analysis of patent specifications experimental data provided by inventors demonstrating practical applicability across diverse production scenarios where quality consistency reliability and regulatory compliance represent non-negotiable requirements for successful adoption by major multinational pharmaceutical companies seeking advanced synthetic solutions for complex intermediate production needs.
Q: How does this metal-free synthesis improve environmental compliance compared to conventional methods?
A: The elimination of transition metal catalysts removes hazardous waste streams associated with metal removal processes while utilizing elemental sulfur as an abundant industrial byproduct. This significantly reduces environmental impact through simplified waste treatment procedures and lower energy consumption during manufacturing operations.
Q: What are the scalability advantages of temperature-regulated synthesis for commercial production?
A: The process operates under standard atmospheric pressure using common laboratory equipment that scales directly to commercial reactors without requiring specialized infrastructure. Temperature control provides precise pathway selection between dithiine and thiophene products without additional reagents or catalysts.
Q: How does the elimination of metal catalysts impact cost reduction in pharmaceutical intermediate manufacturing?
A: Removing expensive transition metals eliminates both catalyst procurement costs and extensive purification steps needed to remove metal residues below regulatory limits. This streamlines production while reducing solvent consumption and waste generation throughout the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiophene Derivatives Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation ensuring consistent delivery of high-quality intermediates meeting global regulatory standards required by leading pharmaceutical manufacturers worldwide where even minor deviations can trigger costly batch rejections during quality assurance inspections conducted by major multinational drug companies demanding absolute reliability in their supply networks across all production scales from laboratory validation through full commercial implementation without any compromise on quality or consistency.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis route can optimize your specific supply chain requirements please contact us for detailed COA data and route feasibility assessments tailored to your pharmaceutical intermediate needs ensuring seamless integration into your existing manufacturing processes while maximizing cost efficiency without compromising quality standards essential for regulatory compliance in global markets.
