Advanced Mo-Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharmaceutical Manufacturing
Advanced Mo-Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharmaceutical Manufacturing
The landscape of modern pharmaceutical synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign pathways to access complex heterocyclic scaffolds. A pivotal development in this domain is detailed in Chinese Patent CN113307778A, which discloses a robust preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These nitrogen-containing heterocycles are not merely academic curiosities; they serve as critical structural motifs in a vast array of bioactive molecules, including well-known therapeutics such as Sitagliptin, Deferasirox, and Maraviroc. The introduction of a trifluoromethyl group into these frameworks is particularly strategic, as it significantly enhances key physicochemical properties such as metabolic stability, lipophilicity, and bioavailability, thereby optimizing the drug-like characteristics of the final active pharmaceutical ingredient.
This patented methodology represents a significant leap forward by utilizing a dual catalytic system involving molybdenum hexacarbonyl and cuprous acetate to facilitate the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. Unlike traditional routes that often suffer from harsh conditions or limited substrate scope, this approach operates under relatively mild thermal conditions and employs commercially available starting materials. For R&D directors and process chemists, the ability to access these valuable intermediates through a streamlined, one-pot procedure offers a compelling advantage in accelerating lead optimization and process development timelines. The versatility of this chemistry allows for the facile incorporation of various functional groups, making it an indispensable tool for the construction of diverse chemical libraries aimed at discovering next-generation therapeutics.
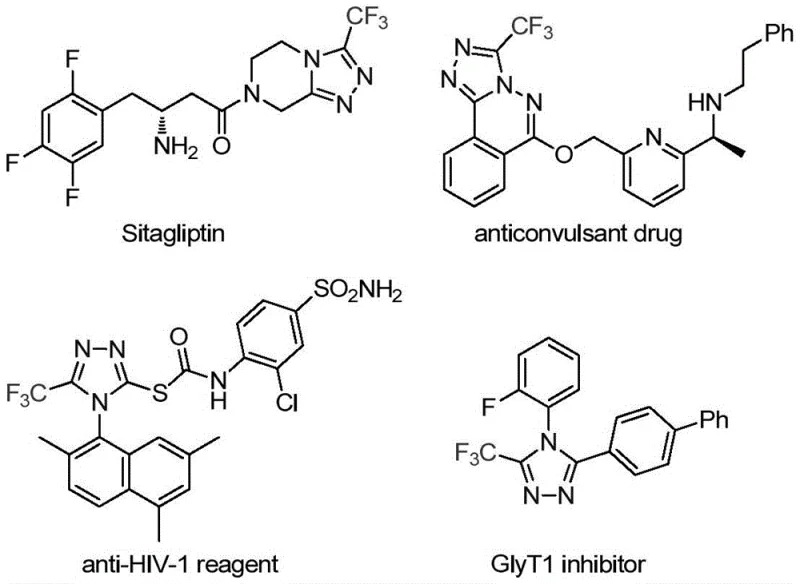
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazole rings has relied on synthetic strategies that present significant operational and economic challenges for industrial scale-up. Traditional literature reports predominantly describe the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinone precursors. These classical pathways often necessitate the use of highly reactive and potentially hazardous hydrazine derivatives, which pose safety risks and complicate waste management protocols in a manufacturing setting. Furthermore, alternative multi-component reactions involving diazonium salts and trifluorodiazoethane, while effective in some contexts, frequently require stringent control over reaction parameters and can exhibit poor atom economy. The reliance on such specialized or unstable reagents invariably drives up the cost of goods sold (COGS) and introduces supply chain vulnerabilities, as the availability of high-purity diazo compounds can be inconsistent. Additionally, many of these legacy methods struggle to selectively produce 3,4-disubstituted 1,2,4-triazoles, often resulting in complex mixtures of regioisomers that are difficult and expensive to separate, thereby reducing the overall process efficiency and yield.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach outlined in patent CN113307778A leverages a sophisticated yet practical transition metal co-catalysis strategy to achieve high-efficiency synthesis. By employing trifluoroethylimidoyl chloride and functionalized isonitriles (specifically phosphazide-type isonitriles like Ph3P=N-NC) as the primary building blocks, this method bypasses the need for unstable hydrazines or diazo species entirely. The reaction proceeds through a cooperative catalytic cycle where molybdenum hexacarbonyl activates the isonitrile species while cuprous acetate promotes the subsequent [3+2] cycloaddition with the imidoyl chloride. This synergistic interaction allows the reaction to proceed smoothly at moderate temperatures ranging from 70°C to 90°C, significantly reducing energy consumption compared to high-temperature pyrolysis methods. Moreover, the use of common organic solvents like tetrahydrofuran (THF) and inexpensive bases like triethylamine ensures that the process remains economically viable and operationally simple. The result is a highly convergent synthesis that delivers the target 3-trifluoromethyl-1,2,4-triazole core with excellent regioselectivity and minimal byproduct formation, streamlining the downstream purification process.
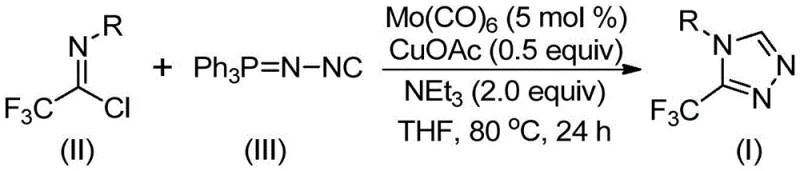
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
Understanding the mechanistic underpinnings of this transformation is crucial for process chemists aiming to optimize reaction parameters and troubleshoot potential scale-up issues. The proposed mechanism initiates with the coordination of molybdenum hexacarbonyl to the functionalized isonitrile, forming a reactive metal-isocyanide complex that lowers the activation energy for the subsequent bond-forming events. Simultaneously, the cuprous acetate acts as a Lewis acid promoter, facilitating the nucleophilic attack of the activated isonitrile species onto the electrophilic carbon of the trifluoroethylimidoyl chloride. This cooperative activation triggers a formal [3+2] cycloaddition, constructing the five-membered triazole ring intermediate with high fidelity. A critical feature of this mechanism is the subsequent elimination of triphenylphosphine oxide, which serves as the driving force for the aromatization of the triazole ring, yielding the stable final product. The presence of molecular sieves in the reaction mixture plays a vital role in scavenging trace water, which could otherwise hydrolyze the sensitive imidoyl chloride starting material or deactivate the metal catalysts, thereby ensuring consistent high yields across different substrate classes.
From an impurity control perspective, this catalytic system offers distinct advantages over non-catalytic thermal cyclizations. The specific electronic tuning provided by the Mo/Cu combination ensures that the cycloaddition occurs with high regioselectivity, effectively suppressing the formation of unwanted 1,2,3-triazole isomers or open-chain adducts that often plague uncatalyzed variants. The mild basicity of triethylamine is sufficient to neutralize the HCl byproduct generated during the reaction without promoting base-sensitive side reactions such as ester hydrolysis or elimination, which is particularly important when dealing with substrates containing delicate functional groups like esters or halides. Furthermore, the catalytic nature of the metal species means that they are used in sub-stoichiometric amounts (e.g., 5 mol% for Mo and 0.5 equiv for Cu), minimizing the residual metal burden in the final API intermediate. This simplifies the heavy metal clearance strategy, a critical regulatory requirement for pharmaceutical manufacturing, as the low loading levels make it easier to meet strict ICH guidelines for residual metals through standard workup procedures like filtration and chromatography.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The execution of this synthesis protocol is designed to be straightforward and adaptable to various laboratory and pilot plant settings. The general procedure involves charging a reaction vessel with the requisite amounts of molybdenum hexacarbonyl, cuprous acetate, triethylamine, and molecular sieves under an inert atmosphere to prevent oxidation of the catalysts. To this mixture, the trifluoroethylimidoyl chloride and the functionalized isonitrile are added in an appropriate organic solvent, typically THF, which provides the optimal balance of solubility and reactivity for these polar intermediates. The reaction mixture is then heated to the specified temperature range and monitored until conversion is complete, after which a simple filtration step removes the solid catalyst residues and molecular sieves. The detailed standardized synthesis steps, including precise stoichiometric ratios and specific workup instructions for various derivatives, are provided in the guide below.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent like THF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete cycloaddition.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthetic route translates into tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the substantial cost reduction in pharmaceutical intermediate manufacturing achieved through the utilization of commodity-grade starting materials. Both trifluoroethylimidoyl chlorides and the phosphazide-type isonitriles are derived from widely available bulk chemicals, eliminating the dependency on exotic or custom-synthesized reagents that often carry exorbitant price tags and long lead times. By shifting the supply base to these common precursors, manufacturers can leverage existing global supply chains to secure raw materials at competitive prices, thereby insulating the production budget from the volatility associated with niche chemical markets. Furthermore, the high atom economy of this cycloaddition reaction minimizes waste generation, which directly correlates to lower disposal costs and a reduced environmental footprint, aligning with increasingly stringent green chemistry mandates.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as hydrazines or diazo compounds results in significant operational savings. Since the catalysts (Mo and Cu salts) are used in catalytic quantities rather than stoichiometric amounts, the direct material cost per kilogram of product is drastically lowered. Additionally, the mild reaction conditions (70-90°C) reduce the energy load required for heating compared to high-temperature processes, contributing to lower utility costs. The simplicity of the workup, which avoids complex extraction sequences or cryogenic conditions, further reduces labor and processing time, enhancing the overall throughput of the manufacturing facility.
- Enhanced Supply Chain Reliability: Relying on commercially available starting materials significantly mitigates the risk of supply disruptions. Unlike specialized reagents that may have single-source suppliers or long synthesis lead times, the precursors for this method are produced by multiple vendors globally, ensuring a robust and redundant supply network. This diversity in sourcing options empowers procurement teams to negotiate better terms and maintain continuous production schedules even in the face of regional logistical challenges. The scalability of the reaction, demonstrated from gram to multi-gram levels in the patent data, suggests that the process can be readily transferred to kiloliter-scale reactors without fundamental changes to the chemistry, ensuring a smooth transition from R&D to commercial production.
- Scalability and Environmental Compliance: The use of THF as a solvent and the absence of highly toxic byproducts simplify the waste management profile of the process. The solid byproducts, primarily consisting of metal residues and molecular sieves, can be easily separated by filtration, while the liquid waste streams are amenable to standard distillation and recovery protocols. This streamlined waste profile facilitates compliance with environmental regulations and reduces the burden on effluent treatment plants. Moreover, the robustness of the catalytic system implies that the reaction is less sensitive to minor fluctuations in mixing or temperature, a critical factor for maintaining consistent quality during large-scale batch production, thus reducing the rate of batch failures and reprocessing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method, based on the experimental data and disclosures within the patent documentation. These insights are intended to assist technical teams in evaluating the feasibility of this route for their specific project needs. Understanding the nuances of catalyst loading, substrate compatibility, and purification requirements is essential for successful technology transfer and process validation.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies using THF as the preferred solvent at temperatures between 70°C and 90°C for 18 to 30 hours, utilizing a dual catalyst system of Mo(CO)6 and CuOAc.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates broad substrate tolerance, successfully accommodating substituents such as methyl, methoxy, fluoro, chloro, and nitro groups on the aromatic ring without significant yield loss.
Q: Is the purification process complex for large-scale production?
A: The post-treatment is straightforward, involving simple filtration followed by standard silica gel column chromatography, which is a scalable and industry-standard purification technique.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of life-saving medications. Our team of expert process chemists has thoroughly analyzed the methodology described in patent CN113307778A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to your supply chain. We are committed to delivering high-purity 3-trifluoromethyl-1,2,4-triazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel analogs or reliable tonnage supply of established intermediates, our infrastructure is designed to support your growth from early-stage discovery through to full-scale commercialization.
We invite you to engage with our technical procurement team to discuss how this advanced Mo/Cu co-catalyzed technology can optimize your specific project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume needs, ensuring that you achieve the best possible value without compromising on quality. We encourage you to contact us today to request specific COA data for our available catalog compounds or to initiate a dialogue regarding route feasibility assessments for your proprietary targets. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing and securing a competitive edge in the global pharmaceutical market.
