Advanced Palladium-Catalyzed Strategy for Scalable Production of Bioactive Pyrrol-2-one Derivatives
Introduction to Next-Generation Lactam Synthesis
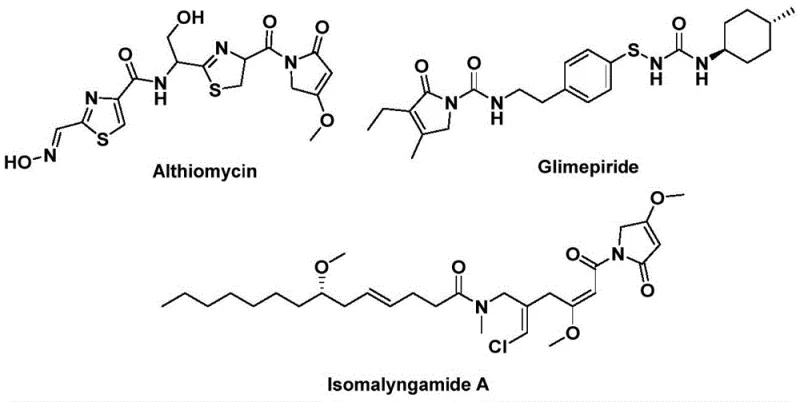
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a privileged scaffold in medicinal chemistry, serving as the core backbone for numerous bioactive natural products and pharmaceutical agents. As highlighted in recent intellectual property developments, specifically patent CN112694430B, innovative synthetic methodologies are emerging to access these valuable heterocycles more efficiently. This patent discloses a robust preparation method that leverages palladium catalysis to construct the pyrrol-2-one ring system in a single operational step. The significance of this chemical architecture cannot be overstated, as it is integral to molecules exhibiting potent antibacterial, hypoglycemic, and anticancer activities. For instance, derivatives of this class are found in compounds like Althiomycin and Glimepiride, underscoring the critical need for reliable supply chains capable of delivering high-purity intermediates. By adopting this advanced catalytic protocol, manufacturers can overcome historical bottlenecks associated with lactam synthesis, paving the way for more agile and cost-effective drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of five-membered lactam rings has often relied on multi-step sequences involving the activation of carboxylic acids or the use of harsh cyclization conditions that limit functional group tolerance. Classical approaches may require pre-functionalized starting materials that are expensive or difficult to source, thereby inflating the overall cost of goods sold (COGS) for the final active pharmaceutical ingredient. Furthermore, many existing protocols suffer from poor atom economy, generating significant waste streams that complicate downstream purification and environmental compliance. The reliance on stoichiometric reagents rather than catalytic systems in older methods also contributes to higher material costs and increased burden on waste management infrastructure. These inefficiencies create substantial friction in the supply chain, leading to longer lead times and reduced flexibility for process chemists attempting to optimize routes for commercial scale-up.
The Novel Approach
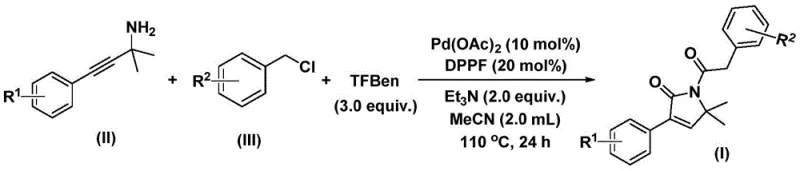
In stark contrast, the methodology described in patent CN112694430B introduces a streamlined, palladium-catalyzed bis-carbonylation strategy that fundamentally reshapes the synthetic landscape for these intermediates. This novel approach utilizes readily available benzyl chlorides and propargyl amines as starting materials, which are commodity chemicals accessible from a wide network of global suppliers. By employing a solid carbon monoxide surrogate instead of toxic CO gas, the process enhances operational safety while maintaining high reaction efficiency. The transformation proceeds under relatively mild thermal conditions, typically between 100-120°C, and demonstrates exceptional compatibility with diverse functional groups including halogens and alkoxy substituents. This one-step convergence not only drastically reduces the number of unit operations but also minimizes solvent consumption and waste generation, aligning perfectly with modern green chemistry principles and sustainability goals for the fine chemical industry.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
The catalytic cycle underpinning this transformation is a sophisticated example of transition metal-mediated carbonylative cyclization, offering deep insights for process optimization. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium intermediate. Subsequently, carbon monoxide, which is released in situ from the phenol 1,3,5-tricarboxylate surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes coordination and insertion with the alkyne moiety of the propargyl amine, setting the stage for ring closure. A second molecule of carbon monoxide is inserted into the newly formed organopalladium bond, expanding the metallacycle before final reductive elimination releases the 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active catalyst. Understanding this mechanistic pathway is crucial for R&D teams aiming to fine-tune ligand environments or adjust stoichiometry to maximize turnover numbers and minimize catalyst loading in large-scale reactors.
Control over impurity profiles is another critical aspect where this mechanism offers distinct advantages over non-catalytic alternatives. The specificity of the palladium insertion and the sequential nature of the carbonyl insertions help to suppress side reactions such as homocoupling of the benzyl chloride or polymerization of the alkyne. The use of DPPF as a bidentate ligand provides a stable coordination environment that favors the desired cyclization pathway over competing decomposition routes. Furthermore, the choice of triethylamine as a base ensures efficient neutralization of the hydrochloric acid byproduct without promoting unwanted base-catalyzed degradation of the sensitive lactam ring. For quality control professionals, this means a cleaner crude reaction profile, which simplifies the purification process and ensures that the final API intermediate meets stringent regulatory specifications for residual metals and organic impurities. The robustness of this catalytic system allows for consistent batch-to-batch reproducibility, a key metric for validating commercial manufacturing processes.
How to Synthesize 1,5-dihydro-2H-pyrrol-2-one Efficiently
To implement this high-efficiency synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and thermal parameters as outlined in the patent literature. The process involves charging a reaction vessel with palladium acetate, the DPPF ligand, the CO surrogate, and the amine and chloride substrates in acetonitrile solvent. Maintaining the temperature at 110°C for approximately 24 hours is critical to driving the reaction to full conversion while minimizing thermal degradation. Detailed standard operating procedures regarding reagent addition order, inert atmosphere maintenance, and workup protocols are essential for achieving the reported high yields.
- Combine palladium acetate, DPPF ligand, carbon monoxide surrogate (TFBen), triethylamine, propargyl amine, and benzyl chloride in acetonitrile solvent within a reaction vessel.
- Heat the reaction mixture to a temperature range of 100-120°C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 1,5-dihydro-2H-pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents a compelling value proposition centered on cost stability and sourcing flexibility. The reliance on benzyl chlorides and propargyl amines as feedstocks means that raw material availability is not constrained by niche suppliers, as these are produced globally in massive volumes for various chemical sectors. This commoditization of starting materials insulates the supply chain from the volatility often seen with specialized, custom-synthesized building blocks. Moreover, the elimination of hazardous gaseous carbon monoxide from the process inventory reduces the need for specialized high-pressure equipment and extensive safety monitoring systems, leading to significant capital expenditure savings during facility setup. The simplified one-pot nature of the reaction also translates to reduced labor costs and shorter cycle times, allowing manufacturing sites to increase throughput without expanding physical footprint.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high atom economy and the use of inexpensive, bench-stable reagents. By avoiding multi-step sequences, manufacturers save on solvent usage, energy consumption for heating and cooling between steps, and the labor associated with isolating intermediate compounds. The high yields reported in the patent data, often exceeding 80% for a wide range of substrates, ensure that raw material utilization is maximized, directly lowering the variable cost per kilogram of the final product. Additionally, the ability to use a solid CO surrogate eliminates the logistical costs and safety premiums associated with transporting and storing compressed carbon monoxide gas cylinders.
- Enhanced Supply Chain Reliability: Sourcing resilience is significantly improved because the key inputs are not proprietary or single-source dependent. Benzyl chlorides with various substituents (such as chloro, fluoro, or methyl groups) are standard catalog items available from multiple tier-1 chemical distributors worldwide. This redundancy in the supply base mitigates the risk of production stoppages due to vendor shortages or geopolitical disruptions. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, reducing the rejection rate of incoming raw materials and smoothing out inventory management workflows for procurement teams.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this methodology offers a cleaner profile that facilitates easier permitting and regulatory compliance. The use of acetonitrile, a common and recyclable solvent, simplifies waste stream management compared to processes requiring exotic or highly toxic solvents. The absence of heavy metal waste streams typical of stoichiometric oxidants or reductants further reduces the burden on wastewater treatment facilities. As the industry moves towards stricter carbon footprint reporting, the high efficiency and reduced step count of this process contribute to a lower overall greenhouse gas emission profile per unit of product, aligning with corporate sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers evaluating this route for their own portfolios. Understanding these nuances helps in assessing the feasibility of technology transfer and the potential for process optimization.
Q: What are the primary advantages of this Pd-catalyzed method over traditional lactam synthesis?
A: This novel approach utilizes a one-step bis-carbonylation strategy that eliminates the need for multi-step sequences often required in conventional lactam formation. It features mild reaction conditions, high atom economy, and excellent functional group tolerance, allowing for the direct assembly of complex scaffolds from readily available benzyl chlorides and propargyl amines.
Q: Which substrates are compatible with this synthetic route?
A: The methodology demonstrates broad substrate compatibility, accommodating various substituents on both the benzyl chloride and propargyl amine components. Electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens (F, Cl, Br) and trifluoromethyl groups, are well-tolerated, yielding products with high efficiency.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly amenable to scale-up due to its operational simplicity and the use of commercially available, cost-effective starting materials. The reaction employs standard organic solvents like acetonitrile and avoids hazardous high-pressure carbon monoxide gas by using a solid CO surrogate, significantly enhancing safety and feasibility for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-dihydro-2H-pyrrol-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering high-purity 1,5-dihydro-2H-pyrrol-2-one derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to adapt the patented Pd-catalyzed bis-carbonylation protocol allows us to offer a competitive advantage in terms of both speed to market and cost efficiency for our global partners.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this methodology for your supply chain. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to initiate a dialogue regarding route feasibility assessments for your custom synthesis requirements, ensuring your projects remain on the cutting edge of chemical innovation.
