Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrol-2-One Scaffolds for Commercial API Production
Introduction to Novel Pyrrolone Synthesis Technology
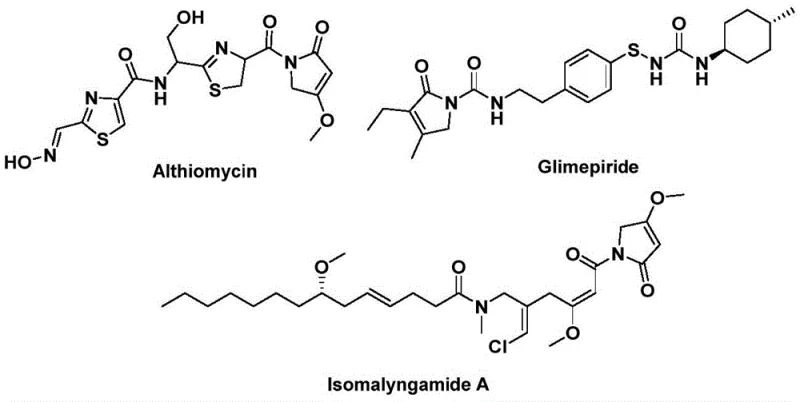
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a privileged scaffold in medicinal chemistry, serving as the core backbone for numerous bioactive natural products and therapeutic agents. As illustrated in the provided chemical structures, prominent examples include Althiomycin, known for its significant antibacterial properties, Glimepiride, a widely prescribed hypoglycemic agent for diabetes management, and Isomalyngamide A, which exhibits potential anticancer activity. The strategic importance of this heterocyclic system drives continuous demand for efficient synthetic methodologies capable of delivering high-purity intermediates for pharmaceutical development. Patent CN112694430B discloses a groundbreaking preparation method that addresses historical challenges in constructing this ring system, utilizing a palladium-catalyzed bis-carbonylation strategy.
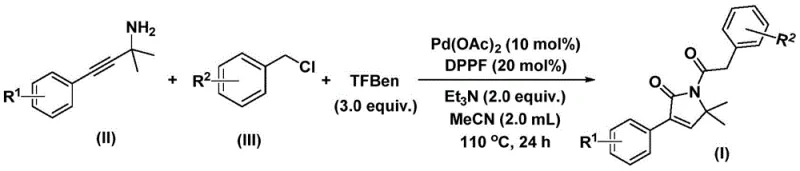
This innovative protocol leverages propargyl amines and benzyl chlorides as readily accessible starting materials, reacting them in the presence of a palladium catalyst and a solid carbon monoxide substitute. Unlike traditional methods that often rely on dangerous gaseous CO under high pressure, this technique employs phenyl 1,3,5-tricarboxylate as a safe and manageable CO source. The reaction proceeds efficiently in organic solvents such as acetonitrile at moderate temperatures between 100°C and 120°C. This technological advancement not only streamlines the synthetic route but also enhances the safety profile of the manufacturing process, making it highly attractive for industrial scale-up and the production of reliable pharmaceutical intermediate supplier networks seeking robust chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one skeleton has been fraught with synthetic complexities and safety hazards. Conventional carbonylation reactions typically necessitate the use of carbon monoxide gas, which is extremely toxic and requires specialized high-pressure equipment to handle safely, imposing significant capital expenditure and operational risks on manufacturing facilities. Furthermore, traditional routes often involve multi-step sequences with poor atom economy, leading to substantial waste generation and increased purification burdens. Many existing protocols suffer from limited substrate scope, failing to tolerate sensitive functional groups or requiring harsh reaction conditions that degrade complex molecules. These inefficiencies result in higher production costs and longer lead times, creating bottlenecks in the supply chain for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast, the method described in patent CN112694430B offers a transformative one-pot solution that circumvents these obstacles through the use of a solid CO surrogate. By utilizing phenyl 1,3,5-tricarboxylate, the process eliminates the need for gas cylinders and high-pressure reactors, allowing the reaction to be conducted in standard glassware or stainless steel vessels under atmospheric or slightly elevated pressure. The reaction demonstrates exceptional versatility, accommodating a wide array of substituents on both the amine and chloride components, including electron-donating and electron-withdrawing groups. This broad compatibility ensures that diverse analogues can be synthesized without redesigning the entire process, facilitating rapid library generation for drug discovery. The simplicity of the workup, involving filtration and standard column chromatography, further underscores the practical utility of this method for commercial applications.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The mechanistic pathway of this transformation involves a sophisticated catalytic cycle driven by palladium species, which orchestrates the sequential insertion of carbon monoxide and cyclization events. Initially, the palladium catalyst undergoes oxidative insertion into the carbon-chlorine bond of the benzyl chloride substrate to generate a benzylpalladium intermediate. Subsequently, carbon monoxide, released in situ from the thermal decomposition of the phenyl 1,3,5-tricarboxylate surrogate, inserts into the palladium-carbon bond to form an acylpalladium species. This acyl intermediate then reacts with the propargyl amine component, triggering a cyclization event that constructs the five-membered nitrogen-containing ring. A second molecule of carbon monoxide is inserted into the resulting organopalladium species, expanding the coordination sphere before final reductive elimination releases the target 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active catalyst.
Understanding this mechanism is crucial for R&D directors focused on purity and impurity profiles. The controlled release of CO from the solid surrogate helps maintain a steady, low concentration of carbon monoxide in the reaction medium, which minimizes side reactions such as homocoupling of the benzyl chloride or over-carbonylation. The choice of ligand, specifically 1,1'-bis(diphenylphosphino)ferrocene (DPPF), plays a pivotal role in stabilizing the palladium center and promoting the specific regioselectivity required for the formation of the desired pyrrolone ring rather than alternative isomers. This precise control over the catalytic cycle ensures high selectivity and reduces the formation of difficult-to-remove byproducts, thereby simplifying downstream purification and ensuring the delivery of high-purity pharmaceutical intermediates suitable for stringent regulatory standards.
How to Synthesize 1,5-Dihydro-2H-Pyrrol-2-One Efficiently
The execution of this synthesis requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and efficiency. The protocol specifies the use of palladium acetate as the precatalyst and triethylamine as the base, dissolved in acetonitrile, which has been identified as the optimal solvent for solubilizing all components and facilitating high conversion rates. The reaction temperature is maintained between 100°C and 120°C for a period of 24 to 48 hours, ensuring complete consumption of the starting materials. Detailed standardized operating procedures regarding specific molar ratios, addition sequences, and purification techniques are essential for reproducibility.
- Charge a reaction vessel with palladium acetate catalyst, DPPF ligand, triethylamine base, phenyl 1,3,5-tricarboxylate (CO source), propargyl amine substrate, and benzyl chloride derivative in acetonitrile solvent.
- Heat the reaction mixture to a temperature range of 100-120°C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 1,5-dihydro-2H-pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible operational improvements and risk mitigation. The shift from gaseous CO to a solid surrogate fundamentally alters the safety landscape of the production facility, removing the requirement for specialized high-pressure infrastructure and reducing insurance and compliance costs associated with toxic gas handling. This simplification allows for greater flexibility in manufacturing site selection and reduces the barrier to entry for producing these valuable intermediates. Additionally, the use of commercially available starting materials like benzyl chlorides and propargyl amines ensures a stable and diversified supply base, minimizing the risk of raw material shortages that could disrupt production schedules.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the associated safety measures leads to significant capital expenditure savings. Furthermore, the one-pot nature of the reaction reduces solvent usage, energy consumption, and labor hours compared to multi-step alternatives. The high reaction efficiency and yields observed across various substrates mean less raw material is wasted, directly improving the cost of goods sold. By avoiding expensive and hazardous reagents, the overall process economics are optimized, allowing for more competitive pricing strategies in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions and the wide availability of the requisite reagents contribute to a more resilient supply chain. The method's tolerance for various functional groups means that supply disruptions for specific substituted starting materials can often be mitigated by switching to alternative analogues without revalidating the entire process. The simplified workup procedure accelerates the turnaround time from reaction completion to finished product, enabling faster response to customer demands and reducing inventory holding times.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of gas-liquid mass transfer limitations typical of traditional carbonylations. The use of a solid CO source ensures consistent reaction kinetics regardless of scale. Moreover, the reduced generation of hazardous waste and the avoidance of toxic gases align with increasingly stringent environmental regulations, facilitating smoother permitting processes and enhancing the company's sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this technology for large-scale manufacturing.
Q: What is the primary advantage of using phenyl 1,3,5-tricarboxylate in this synthesis?
A: Phenyl 1,3,5-tricarboxylate acts as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous high-pressure CO gas, thereby significantly enhancing operational safety and simplifying equipment requirements for scale-up.
Q: What is the substrate compatibility of this palladium-catalyzed method?
A: The method demonstrates excellent functional group tolerance, successfully accommodating various substituents such as halogens (F, Cl, Br), alkyl groups (methyl, tert-butyl), alkoxy groups (methoxy), and electron-withdrawing groups like trifluoromethyl and cyano on both the propargyl amine and benzyl chloride components.
Q: How does this method compare to traditional carbonylation techniques in terms of yield?
A: This novel approach achieves high reaction efficiency with isolated yields often exceeding 75% and reaching up to 92% for optimized substrates, which is competitive with or superior to many conventional multi-step syntheses that suffer from lower overall throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrol-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and commercialization. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet rigorous quality standards, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities. Our facility is equipped to handle complex catalytic processes, including the palladium-catalyzed reactions described herein, with a focus on safety, consistency, and regulatory compliance.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your project requirements. We encourage you to contact us for specific COA data and route feasibility assessments to evaluate the suitability of our manufacturing capabilities for your next-generation therapeutic programs. Together, we can drive efficiency and innovation in the production of vital bioactive molecules.
