Advanced Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly quinazolinone scaffolds which serve as critical backbones for numerous bioactive molecules. As highlighted in recent intellectual property developments, specifically patent CN111675662B, there has been a significant breakthrough in the efficient preparation of 2-trifluoromethyl substituted quinazolinone compounds. These structures are not merely academic curiosities; they are foundational elements in the design of next-generation therapeutics exhibiting anti-cancer, anticonvulsant, and anti-inflammatory properties. The introduction of the trifluoromethyl group specifically enhances metabolic stability and lipophilicity, making these intermediates highly coveted by R&D teams globally. This report analyzes the technical merits of this iron-catalyzed approach and its implications for supply chain optimization.
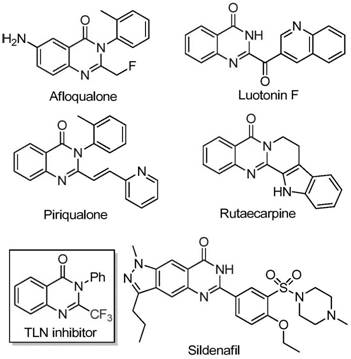
Historically, the construction of trifluoromethyl-substituted quinazolinones has been plagued by significant synthetic bottlenecks that hindered widespread adoption in process chemistry. Conventional methods typically rely on the cyclization of synthons bearing the trifluoromethyl group with substrates like anthranilamide or isatoic anhydride. While chemically feasible, these traditional pathways often suffer from severe reaction conditions that require stringent anhydrous environments or expensive activating agents. Furthermore, the reliance on specialized trifluoromethyl synthons such as trifluoroacetic anhydride drives up the raw material costs substantially, creating a barrier for cost-sensitive manufacturing projects. The narrow substrate scope of these older methods also limits the ability of medicinal chemists to explore diverse chemical space efficiently, often resulting in low yields when electronic or steric variations are introduced to the aromatic rings.
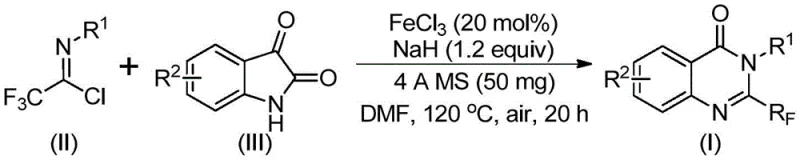
In stark contrast, the novel approach detailed in the referenced patent utilizes a remarkably simple yet effective strategy centered around inexpensive iron catalysis. By employing readily available trifluoroethylimidoyl chloride and isatin as starting raw materials, this method bypasses the need for costly pre-functionalized building blocks. The reaction proceeds through a series of cyclization steps catalyzed by cheap metal iron, specifically ferric chloride, which is abundant and economically viable for large-scale operations. This shift in reagent selection represents a paradigm change in how these valuable intermediates can be accessed, moving away from precious metal dependency towards base-metal catalysis. The operational simplicity allows for gram-level expansion with the clear possibility for industrial scale application, addressing the critical need for scalable processes in the fine chemical sector.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic chemistry applied to process development. During the reaction, an alkali-promoted carbon-nitrogen bond formation occurs initially between the trifluoroethylimidoyl chloride and the isatin substrate. This intermediate step generates a trifluoroacetamidine compound which serves as the precursor for the subsequent ring closure. Following this, the iron catalyst facilitates a decarbonylation and cyclization sequence that isomerizes the intermediate into the final stable 2-trifluoromethyl-substituted quinazolinone structure. The use of sodium hydride as a base ensures the deprotonation necessary for nucleophilic attack, while the 4A molecular sieves play a crucial role in scavenging moisture that could otherwise deactivate the sensitive imidoyl chloride species.
Understanding the impurity profile is paramount for R&D directors focused on regulatory compliance and downstream processing. The high functional group tolerance observed in this system suggests that side reactions such as hydrolysis of the imidoyl chloride are effectively minimized under the optimized conditions. The reaction conditions, typically involving a temperature ramp from 40°C to 120°C in DMF solvent, allow for controlled kinetics that favor the desired cyclization over competing decomposition pathways. The robustness of the iron catalyst ensures that even with substrates bearing electron-withdrawing or electron-donating groups, the reaction proceeds with high conversion rates. This mechanistic reliability translates directly into cleaner crude reaction mixtures, reducing the burden on purification teams and ensuring that the final active pharmaceutical ingredient precursors meet stringent quality specifications.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and safety. The protocol dictates a precise stoichiometric balance where the molar ratio of ferric chloride to sodium hydride is maintained around 0.2 to 1.2 relative to the substrate. Operators must ensure the thorough mixing of trifluoroethylimidoyl chloride and isatin in an aprotic organic solvent, with DMF being the preferred medium due to its superior solubilizing power for these polar intermediates. The detailed standardized synthesis steps involve a two-stage heating process to manage the exothermic nature of the initial bond formation followed by the energy-intensive cyclization step. For a comprehensive guide on the exact operational parameters and safety precautions, please refer to the structured protocol below.
- Combine ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent such as DMF.
- Initiate the reaction at 40°C for 8-10 hours, then increase temperature to 120°C and maintain for 18-20 hours under air atmosphere.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the transition to this iron-catalyzed methodology offers substantial strategic benefits that extend beyond mere chemical yield. The primary driver for cost reduction in pharmaceutical intermediates manufacturing lies in the replacement of expensive reagents with commodity chemicals. By eliminating the need for precious metal catalysts and specialized trifluoromethylating agents, the direct material cost of goods sold is significantly lowered. This economic efficiency is compounded by the use of DMF, a common industrial solvent that is easily sourced and recycled, further enhancing the overall process economics. For procurement managers, this means a more stable pricing model for these critical building blocks, insulated from the volatility often associated with rare earth or precious metal markets.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts like palladium or rhodium removes the necessity for expensive heavy metal removal steps downstream. This simplification of the purification train reduces both the consumption of silica gel and the time required for chromatographic separation. Consequently, the overall production cost is drastically simplified, allowing for substantial cost savings that can be passed down the supply chain. The use of ferric chloride, a ubiquitous industrial chemical, ensures that catalyst sourcing is never a bottleneck, providing a predictable and low-cost input for continuous manufacturing campaigns.
- Enhanced Supply Chain Reliability: The starting materials, specifically isatin and various aromatic amines used to generate the imidoyl chloride, are widely available from multiple global suppliers. This multi-sourcing capability mitigates the risk of supply disruption, a critical concern for supply chain heads managing just-in-time inventory. The robustness of the reaction conditions, which tolerate air and moisture better than many organometallic processes, reduces the requirement for specialized inert atmosphere equipment. This flexibility allows for production in a wider range of facilities, increasing the overall resilience of the supply network against regional disruptions or logistical delays.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, having been validated from milligram to gram scales with consistent results, indicating a smooth path to commercial scale-up of complex pharmaceutical intermediates. The waste profile is favorable as the primary byproducts are inorganic salts that are easier to treat than organic heavy metal waste. This aligns with increasingly stringent environmental regulations, reducing the liability and cost associated with waste disposal. The ability to run the reaction at moderate temperatures without extreme pressure requirements further lowers the energy footprint of the manufacturing process, supporting sustainability goals.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific pipelines, we have compiled answers to common inquiries regarding the practical implementation of this patent. The following insights are derived directly from the experimental data and beneficial effects described in the intellectual property documentation. These responses address concerns related to catalyst loading, substrate versatility, and purification strategies, providing a clear picture of what to expect during technology transfer. Understanding these nuances is essential for assessing the feasibility of integrating this route into existing production schedules.
Q: What are the primary advantages of using FeCl3 in this quinazolinone synthesis?
A: The use of ferric chloride offers significant cost advantages over precious metal catalysts while maintaining high reaction efficiency and broad substrate tolerance.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the protocol utilizes readily available starting materials and simple post-treatment procedures like filtration and chromatography, facilitating scalable manufacturing.
Q: What is the typical purity profile of the resulting intermediates?
A: The method demonstrates excellent functional group tolerance and yields high-purity products, often exceeding 90% yield in optimized examples without complex purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
As the demand for fluorinated heterocycles continues to surge in the development of novel therapeutics, having a manufacturing partner with deep technical expertise is indispensable. NINGBO INNO PHARMCHEM stands ready to support your projects with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 2-trifluoromethyl quinazolinone meets the exacting standards required for clinical and commercial applications. We understand the critical nature of these intermediates in your value chain and are committed to delivering consistency and quality.
We invite you to engage with our technical team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this iron-catalyzed route for your specific targets. Please contact our technical procurement team today to索取 specific COA data and route feasibility assessments. Let us collaborate to optimize your supply chain and accelerate the delivery of life-saving medicines to the market.
