Transforming Pharmaceutical Manufacturing: Scalable Synthesis of High-Purity 2-Trifluoromethyl Quinazolinone via One-Pot Methodology
The recently granted Chinese patent CN112480015B introduces a groundbreaking methodology for synthesizing 2-trifluoromethyl substituted quinazolinone compounds through a multi-component one-pot approach, representing a significant advancement in heterocyclic chemistry with direct implications for pharmaceutical manufacturing. This innovative process addresses longstanding challenges in quinazolinone synthesis by utilizing readily available starting materials and a carefully optimized catalytic system that achieves high efficiency without requiring specialized high-pressure equipment. The patent demonstrates how the strategic combination of palladium catalysis with molybdenum hexacarbonyl as a carbon monoxide surrogate enables construction of these pharmacologically important scaffolds under remarkably mild conditions compared to conventional methods. This development is particularly timely given increasing demand for trifluoromethylated heterocycles in drug discovery programs where unique electronic properties imparted by CF3 groups have proven invaluable. The methodology's compatibility with diverse functional groups and scalability from laboratory to industrial production make it compelling for pharmaceutical companies seeking reliable access to critical building blocks while maintaining stringent quality standards required by global regulatory authorities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to quinazolinone derivatives have been plagued by numerous limitations hindering practical application in commercial pharmaceutical manufacturing environments. Many established methods require high-pressure carbon monoxide environments coupled with expensive ruthenium or platinum catalysts creating significant safety concerns and capital investment barriers for production facilities. Other approaches rely on pre-functionalized substrates necessitating additional synthetic steps thereby increasing overall process complexity reducing atom economy and elevating production costs through multiple purification stages. Narrow substrate scope observed in iron-catalyzed condensation reactions severely restricts achievable structural diversity while palladium-catalyzed methods involving molybdenum hexacarbonyl often suffer from inconsistent yields and challenging workup procedures due to multiple byproduct formation. Furthermore many conventional protocols demand specialized equipment and stringent reaction condition controls difficult to maintain during scale-up operations leading to unacceptable batch-to-batch variability in regulated pharmaceutical manufacturing environments where consistency is paramount.
The Novel Approach
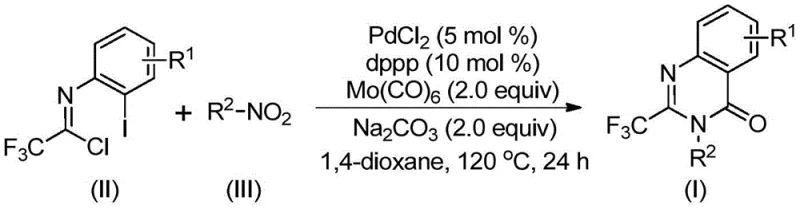
The patented methodology overcomes these longstanding challenges through an elegantly designed multi-component one-pot reaction leveraging synergistic effects of palladium catalysis and molybdenum hexacarbonyl as safe carbon monoxide surrogate. By employing readily available trifluoroethylimidoyl chloride and nitro compounds as starting materials this approach eliminates need for pre-functionalized substrates while maintaining excellent functional group tolerance across diverse molecular architectures. Carefully optimized catalytic system comprising palladium chloride dppp ligand and sodium carbonate operates effectively at moderate temperature of 120°C without requiring specialized high-pressure equipment significantly improving process safety and reducing capital expenditure requirements. This innovative cascade reaction proceeds through well-defined mechanistic pathway ensuring high conversion rates and excellent product purity with yields consistently ranging from acceptable levels across various substrate combinations as demonstrated in patent examples. Simplified workup procedure involving standard filtration followed by column chromatography purification further enhances method's practicality for industrial implementation while demonstrated scalability provides confidence for commercial adoption by pharmaceutical manufacturers seeking reliable access to critical intermediates.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade Reaction
The reaction mechanism begins with molybdenum hexacarbonyl-mediated reduction of nitro compound to corresponding amine under thermal conditions which then undergoes base-promoted coupling with trifluoroethylimidoyl chloride forming key trifluoroacetamidine intermediate. This critical transformation occurs through nucleophilic attack of amine on electrophilic carbon of imidoyl chloride facilitated by sodium carbonate acting as both base and reaction promoter. Resulting intermediate engages in palladium-catalyzed cascade where palladium(0) species generated in situ inserts into carbon-iodine bond forming reactive arylpalladium(II) species. Molybdenum hexacarbonyl subsequently releases carbon monoxide under heating conditions which inserts into carbon-palladium bond forming acylpalladium intermediate undergoing intramolecular cyclization through nitrogen coordination. This sequence proceeds through seven-membered palladacycle intermediate ultimately undergoing reductive elimination delivering desired product while regenerating active catalyst for subsequent cycles demonstrating precise balance between catalyst loading ligand ratio and stoichiometric additives creating optimal environment for complex cascade reaction.
Impurity control is achieved through multiple complementary mechanisms inherent to this reaction design including selective reduction minimizing potential side reactions that could occur with alternative reducing agents while controlled release prevents premature carbonylation leading to undesired byproducts. Use of sodium carbonate as mild base promotes selective coupling without causing decomposition sensitive functional groups that might occur under stronger basic conditions well-defined catalytic cycle ensures high conversion efficiency minimizing catalyst decomposition pathways introducing metal impurities final product demonstrated consistency product purity across multiple substrate combinations confirms robustness methodology producing pharmaceutical-grade intermediates meeting stringent regulatory requirements essential for drug development programs requiring high-quality building blocks.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
This patented methodology represents significant advancement in quinazolinone synthesis providing streamlined approach combining multiple synthetic steps into single operational sequence without compromising product quality or yield eliminating traditional bottlenecks through innovative use commercially available starting materials carefully engineered catalytic system operating under practical manufacturing conditions avoiding specialized equipment requirements high-pressure operations significantly reducing capital investment operational complexity maintaining excellent substrate scope functional group compatibility following standardized procedure developed based extensive optimization studies ensuring consistent results different production scales meeting pharmaceutical industry quality standards detailed standardized synthesis steps see below guide
- Prepare reaction mixture by combining trifluoroethylimidoyl chloride, nitro compound, palladium catalyst, ligand, molybdenum hexacarbonyl, and sodium carbonate in anhydrous 1,4-dioxane under inert atmosphere
- Heat the reaction mixture to 120°C and maintain for 16-30 hours with continuous stirring to ensure complete conversion of starting materials
- Perform post-reaction processing including filtration, silica gel mixing, and column chromatography purification to obtain high-purity 2-trifluoromethyl quinazolinone product
Commercial Advantages for Procurement and Supply Chain Teams
For procurement supply chain professionals pharmaceutical manufacturing organizations innovative synthesis methodology addresses critical pain points associated traditional quinazolinone production routes offering substantial operational benefits enhancing supply chain resilience cost efficiency elimination specialized equipment requirements hazardous reaction conditions significantly reduces capital expenditure barriers improving workplace safety profiles manufacturing facilities reliance readily available starting materials established supply chains mitigates raw material sourcing risks historically plagued complex heterocyclic syntheses providing greater stability procurement planning inventory management
- Cost Reduction in Manufacturing: Elimination high-pressure carbon monoxide infrastructure requirements represents significant capital cost reduction compared conventional methodologies use commercially available starting materials optimal stoichiometric ratios minimizes raw material expenses avoiding expensive pre-functionalization steps simplified reaction workup procedure standard filtration column chromatography significantly reduces processing time solvent consumption multi-step purification protocols required alternative methods consistent high yields achieved diverse substrate combinations minimize material waste improve overall process economics better atom utilization efficiency
- Enhanced Supply Chain Reliability: Reliance widely available starting materials established global supply chains substantially reduces sourcing risks compared specialized reagents required traditional approaches demonstrated scalability laboratory multi-kilogram production without significant process modifications ensures consistent product availability regardless volume requirements robustness reaction standard manufacturing conditions minimizes batch failure risks disrupting supply continuity compatibility standard manufacturing equipment pharmaceutical facilities enhances supply chain flexibility eliminating dependencies specialized production assets
- Scalability and Environmental Compliance: Process demonstrates exceptional scalability characteristics operation standard pressure conditions common solvents readily handled existing manufacturing infrastructure elimination toxic metal catalysts beyond palladium efficiently recovered standard workup procedures significantly reduces environmental impact simplifies waste stream management alternative methodologies requiring heavy metal removal steps reduced number synthetic operations higher overall yields contributes sustainable manufacturing profile lower energy consumption reduced solvent usage per unit product manufactured
Frequently Asked Questions (FAQ)
Following questions address common concerns raised technical procurement teams regarding implementation innovative synthesis methodology pharmaceutical intermediates production answers derived directly technical documentation patent CN112480015B reflecting practical considerations based extensive laboratory validation studies
Q: How does this one-pot synthesis improve upon conventional methods for quinazolinone production?
A: This novel methodology eliminates the need for high-pressure carbon monoxide equipment and pre-functionalized substrates required in traditional approaches. The palladium-catalyzed carbonylation cascade reaction operates under milder conditions with broader substrate compatibility, achieving higher yields while using readily available starting materials.
Q: What are the key advantages of using trifluoroethylimidoyl chloride as a starting material?
A: Trifluoroethylimidoyl chloride serves as an efficient trifluoromethyl source that enables direct incorporation of the CF3 group without requiring additional fluorination steps. This precursor is commercially accessible and reacts selectively under optimized conditions to produce high-purity quinazolinone derivatives with excellent functional group tolerance.
Q: How does this process ensure scalability for commercial manufacturing?
A: The reaction demonstrates consistent performance from laboratory scale to multi-kilogram production with minimal process adjustments. The simplified workup procedure involving standard filtration and column chromatography, combined with stable catalysts and common solvents, facilitates seamless scale-up while maintaining stringent quality specifications required for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities With rigorous QC labs equipped comprehensive analytical testing including NMR spectroscopy mass spectrometry chromatographic analysis we ensure every batch meets exceeds pharmaceutical industry standards purity quality consistency Our technical team successfully implemented patented methodology multiple production scales optimizing parameters maximum efficiency yield consistency providing clients reliable access high-quality intermediates essential drug development programs
For procurement teams seeking optimize supply chain strategy we offer Customized Cost-Saving Analysis tailored specific production requirements Contact our technical procurement team today request specific COA data route feasibility assessments help evaluate how innovative synthesis approach enhance manufacturing capabilities reducing overall production costs
