Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles: A Cost-Effective Route for Pharmaceutical Intermediates
Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles: A Cost-Effective Route for Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and economically viable pathways for constructing nitrogen-containing heterocycles, particularly those bearing fluorinated motifs that enhance metabolic stability and bioavailability. Patent CN114920707B discloses a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, a privileged scaffold found in numerous bioactive molecules ranging from anticonvulsants to Factor IXa inhibitors. This technology represents a paradigm shift in heterocyclic synthesis by leveraging the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source. For R&D directors and procurement strategists, this innovation offers a compelling value proposition: the ability to generate high-purity pharmaceutical intermediates through a simplified, metal-free protocol that drastically reduces raw material complexity and operational overhead.
The significance of this chemical architecture cannot be overstated, as trifluoromethyl-substituted triazoles serve as critical building blocks for next-generation therapeutics. As illustrated in the structural diversity of bioactive targets, the incorporation of the CF3 group significantly modulates the physicochemical properties of the parent molecule, often leading to improved pharmacokinetics. 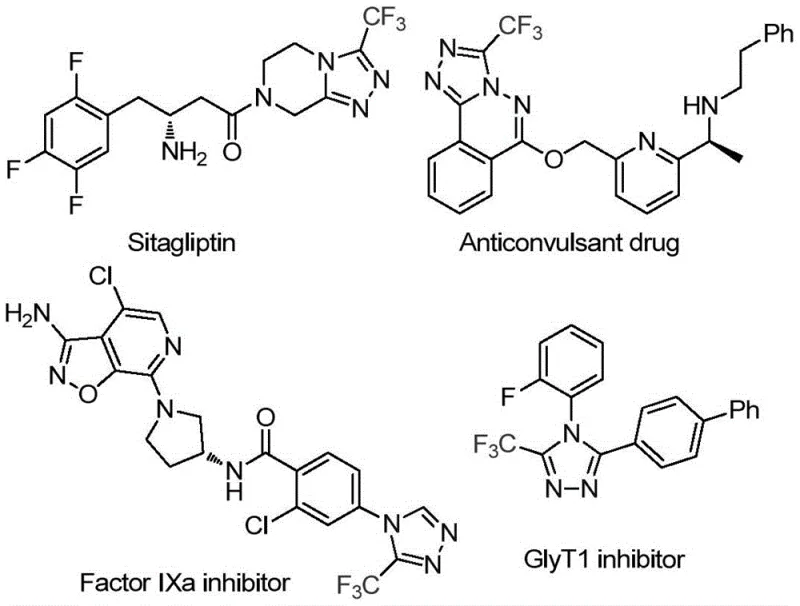
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to 1,2,4-triazoles often suffer from significant inefficiencies that burden both the laboratory and the production floor. Classical methodologies frequently rely on the condensation of hydrazides with nitriles or amidines, which can require harsh reaction conditions, elevated temperatures, and prolonged reaction times to drive the equilibrium toward the desired product. Furthermore, many existing protocols necessitate the use of expensive transition metal catalysts or stoichiometric amounts of hazardous dehydrating agents, introducing heavy metal impurities that are notoriously difficult and costly to remove to meet stringent regulatory standards for active pharmaceutical ingredients (APIs). The requirement for strictly anhydrous and oxygen-free environments in many of these legacy processes further escalates capital expenditure (CAPEX) and operational expenditure (OPEX), as it demands specialized equipment and rigorous safety protocols that slow down throughput and increase the risk of batch failure.
The Novel Approach
In stark contrast, the methodology described in patent CN114920707B introduces an elegant solution by utilizing molecular iodine as a promoter and DMF as a dual-function reagent. This approach eliminates the need for exotic carbon sources or complex catalytic systems, relying instead on the intrinsic reactivity of the solvent itself. The reaction operates under aerobic conditions, meaning that the exclusion of moisture and oxygen is unnecessary, thereby simplifying the engineering controls required for scale-up. By transforming a common commodity chemical into a reactive synthon, this process achieves high atom economy and reduces the overall E-factor of the synthesis. The versatility of this method is demonstrated by its compatibility with a broad spectrum of substrates, allowing for the efficient generation of diverse derivatives without the need for extensive re-optimization of reaction parameters for each new analog.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The core of this technological advancement lies in the unique activation of DMF by molecular iodine, which facilitates a tandem cyclization sequence. Mechanistically, the reaction can proceed via two potential pathways depending on which fragment of the DMF molecule participates as the carbon source. In one pathway, the formyl group of DMF undergoes condensation with the trifluoroethyliminohydrazide to form a hydrazone intermediate, which subsequently cyclizes intramolecularly with the elimination of dimethylamine to yield the triazole ring. Alternatively, the N-methyl group of DMF can be activated by iodine to form an amine salt, which then engages in nucleophilic addition followed by elimination and oxidative aromatization. 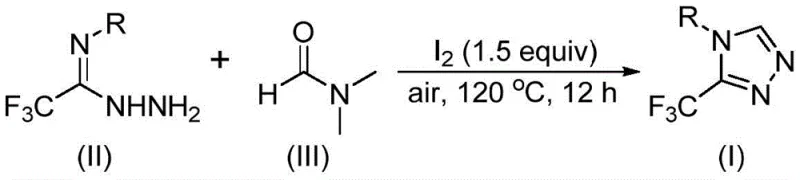
This mechanistic flexibility ensures high conversion rates across various electronic environments of the substrate. The oxidative aromatization step is particularly crucial, as it drives the reaction to completion and establishes the aromatic stability of the final 1,2,4-triazole system. From an impurity control perspective, the use of iodine as a mild oxidant avoids the formation of over-oxidized byproducts often seen with stronger oxidizing agents. The reaction conditions, typically maintained between 110°C and 130°C, provide sufficient thermal energy to overcome activation barriers while remaining within the safe operating limits of standard glass-lined or stainless steel reactors used in fine chemical manufacturing. This balance of reactivity and selectivity is key to achieving the high purity profiles required for downstream pharmaceutical applications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The operational simplicity of this synthesis makes it highly attractive for process chemistry teams looking to streamline their workflow. The procedure involves mixing the trifluoroethyliminohydrazide precursor with molecular iodine in DMF, followed by heating under air. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to ensure reproducibility and safety during implementation.
- Combine molecular iodine, trifluoroethyliminohydrazide, and DMF solvent in a reaction vessel.
- Heat the mixture to 110-130°C under air atmosphere for 10-15 hours to facilitate cyclization.
- Perform post-treatment including filtration, washing, and column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates directly into tangible strategic advantages regarding cost structure and supply reliability. The elimination of precious metal catalysts and the reliance on commodity-grade solvents as reactants fundamentally alters the cost basis of the intermediate. By removing the dependency on scarce or volatile raw materials, manufacturers can secure more stable pricing contracts and mitigate the risks associated with supply chain disruptions. The robustness of the reaction conditions further enhances supply continuity, as the process is less susceptible to variations in environmental controls or minor deviations in reagent quality.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the dual role of DMF, which acts as both the solvent and the carbon source. This consolidation of functions means that facilities do not need to procure, store, and handle separate carbon donor reagents, which are often expensive and hazardous. Additionally, the absence of transition metal catalysts removes the costly and time-consuming step of heavy metal scavenging and removal, which is a mandatory requirement for API production. The simplified purification process, often requiring only filtration and standard chromatography, reduces solvent consumption and waste disposal costs, leading to substantial overall cost savings in the manufacturing of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including trifluoroethyliminohydrazide derivatives and molecular iodine, are commercially available from multiple global suppliers, ensuring a competitive and resilient supply base. Unlike processes that rely on custom-synthesized reagents with long lead times, the commoditized nature of the inputs allows for just-in-time inventory management and rapid response to demand fluctuations. The tolerance of the reaction to air and moisture further reduces the logistical burden, as reagents do not require specialized shipping or storage conditions, thereby minimizing the risk of degradation during transit and ensuring consistent quality upon arrival at the production site.
- Scalability and Environmental Compliance: From a scale-up perspective, the reaction's operation under air atmosphere and moderate temperatures makes it inherently safer and easier to translate from kilogram to ton-scale production. The process avoids the use of pyrophoric reagents or high-pressure equipment, reducing the safety infrastructure requirements and insurance costs associated with large-scale chemical manufacturing. Furthermore, the high atom efficiency and reduced waste generation align with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. This sustainability profile not only mitigates regulatory risk but also enhances the brand value of the final product in markets that prioritize eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (C1 synthon), eliminating the need for additional expensive carbon donors and simplifying the workup process significantly.
Q: Does this process require inert gas protection?
A: No, the reaction proceeds efficiently under an air atmosphere, which removes the operational complexity and cost associated with maintaining strict anhydrous or anaerobic conditions.
Q: What is the substrate scope for the R group?
A: The method tolerates a wide range of substituents on the aryl ring, including alkyl, alkoxy, alkylthio, halogens, and trifluoromethyl groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iodine-promoted synthesis route for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the exacting standards required for drug development and commercial manufacturing.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective technology for their pipeline projects. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in process optimization can accelerate your time-to-market while optimizing your overall cost of goods sold.
