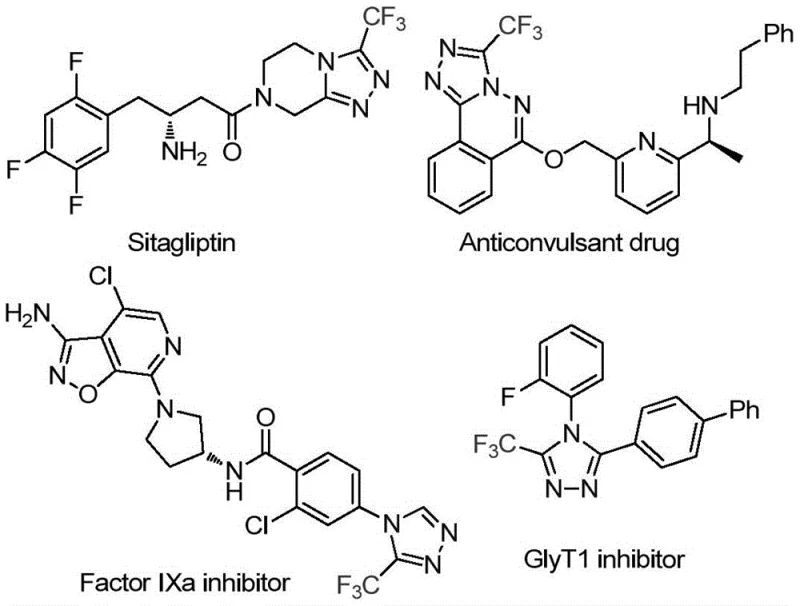
Revolutionizing Triazole Synthesis: Scalable, Cost-Efficient Production of High-Purity Trifluoromethyl Triazoles for Pharmaceutical Applications
The recently granted Chinese patent CN114920707B introduces a groundbreaking methodology for synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in the production of critical pharmaceutical intermediates. This innovative approach leverages dimethylformamide (DMF) as both solvent and carbon source under ambient atmospheric conditions, eliminating traditional constraints that have hindered scalable manufacturing of these biologically active heterocycles. The process demonstrates remarkable operational simplicity while maintaining high efficiency across diverse substrate variations, addressing long-standing challenges in triazole chemistry that have limited commercial adoption in drug development pipelines. By utilizing molecular iodine as a catalyst at moderate temperatures (120°C), this method achieves consistent conversion without requiring specialized equipment or handling protocols, thereby opening new pathways for cost-effective production of fluorinated building blocks essential to modern therapeutics. The patent's emphasis on practical implementation rather than theoretical novelty positions it as a transformative solution for pharmaceutical manufacturers seeking reliable access to these high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to trifluoromethyl-substituted triazoles have been plagued by significant operational constraints that compromise both feasibility and economic viability for commercial production. Most established methods require strictly anhydrous and anaerobic conditions to prevent catalyst deactivation or unwanted side reactions, necessitating expensive glovebox systems and continuous inert gas purging that substantially increase capital expenditure and operational complexity. These processes often employ transition metal catalysts that introduce costly purification challenges due to residual metal contamination, particularly problematic for pharmaceutical applications where stringent purity specifications must be met. Furthermore, conventional approaches typically suffer from narrow substrate scope with limited functional group tolerance, restricting their applicability across diverse molecular architectures required in drug discovery programs. The multi-step nature of many existing syntheses also contributes to lower overall yields and higher waste generation, creating sustainability challenges that conflict with modern green chemistry principles increasingly mandated by regulatory bodies worldwide.
The Novel Approach
The patented methodology overcomes these limitations through an elegantly simple yet highly effective strategy that leverages DMF's dual functionality as both reaction medium and carbon source under ambient atmospheric conditions. By employing molecular iodine as a catalyst at moderate temperatures (120°C), the process achieves efficient cyclization without requiring specialized equipment or handling protocols that have traditionally constrained triazole synthesis. This approach demonstrates exceptional substrate versatility across various aryl substitutions including halogens, alkyl groups, and trifluoromethyl moieties, enabling access to diverse product variants from a single streamlined process. The elimination of moisture-sensitive reagents and transition metal catalysts significantly simplifies downstream purification while ensuring compliance with pharmaceutical quality standards. Most notably, the reaction operates effectively in air atmosphere without yield compromise, representing a paradigm shift from conventional methods that demanded rigorous exclusion of oxygen and moisture to prevent decomposition or side reactions.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism reveals a sophisticated dual pathway where DMF's methyl and formyl groups both serve as carbon sources through distinct cyclization routes. When the formyl group participates, it undergoes condensation with trifluoroethyliminohydrazide to form a hydrazone intermediate that subsequently undergoes intramolecular cyclization with elimination of dimethylamine to yield the target triazole product. Alternatively, when the N-methyl group acts as carbon source, DMF first reacts with iodine to generate an amine salt that eliminates hydrogen iodide before engaging in nucleophilic addition with the hydrazide substrate. This pathway proceeds through an azadiene intermediate followed by intramolecular nucleophilic addition and oxidative aromatization to produce the final triazole structure. The iodine catalyst plays a crucial role in activating both reaction pathways by facilitating key bond cleavage events while maintaining compatibility with air exposure—a significant departure from conventional metal-catalyzed systems that require inert atmospheres.
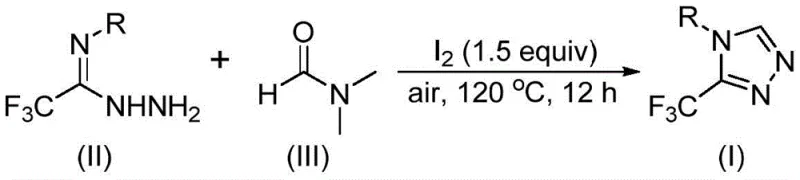
Impurity control is inherently achieved through the reaction's selective mechanism that minimizes competing pathways typically responsible for byproduct formation in triazole syntheses. The absence of transition metals eliminates concerns about residual metal contamination that would require additional purification steps to meet pharmaceutical quality standards. The moderate reaction temperature (120°C) prevents thermal decomposition pathways while ensuring sufficient energy for cyclization completion within the specified timeframe. Substrate scope studies demonstrate consistent performance across various functional groups including halogens (F, Cl), alkyl groups (Me, Et), and electron-withdrawing substituents (CF₃), with no observed interference from these moieties during the cyclization process. This inherent selectivity significantly reduces impurity profiles compared to conventional methods that often require extensive optimization for each new substrate variant.
How to Synthesize Trifluoromethyl Triazoles Efficiently
This innovative synthesis represents a significant advancement in manufacturing efficiency for fluorinated heterocyclic compounds essential to modern pharmaceutical development. The process eliminates multiple pain points associated with traditional triazole production methods through its unique combination of operational simplicity and chemical elegance. By utilizing readily available starting materials under ambient conditions, it removes barriers that have historically limited commercial scale-up of these valuable intermediates. The following standardized procedure provides a reliable framework for consistent production of high-purity trifluoromethyl triazoles while maintaining the flexibility needed to accommodate diverse structural requirements across various therapeutic applications. Detailed implementation guidelines are provided below to ensure successful technology transfer from laboratory to manufacturing environment.
- Combine iodine catalyst (1.5 equivalents), trifluoroethyliminohydrazide substrate, and DMF solvent in a reaction vessel under ambient air conditions
- Heat the mixture to 120°C and maintain for 12 hours with continuous stirring to ensure complete cyclization
- Perform standard post-treatment including filtration, silica gel mixing, and column chromatography purification to isolate the target triazole compound
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points in the acquisition of complex pharmaceutical intermediates. The elimination of specialized infrastructure requirements significantly reduces capital investment barriers while enhancing operational flexibility across manufacturing sites worldwide. By leveraging commercially available materials under standard processing conditions, the process creates new opportunities for reliable sourcing that aligns with modern supply chain resilience strategies essential in today's volatile global market environment. These advantages translate directly into improved business continuity metrics while maintaining the high quality standards required for pharmaceutical applications.
- Cost Reduction in Manufacturing: The elimination of anhydrous/anaerobic processing requirements removes substantial infrastructure costs associated with inert gas systems and controlled atmosphere environments. By utilizing DMF as both solvent and reactant, the process achieves significant material efficiency while avoiding expensive transition metal catalysts that require complex removal protocols. This streamlined approach substantially reduces both capital expenditure and operational costs throughout the manufacturing lifecycle without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials including molecular iodine and standard DMF solvent ensures consistent access to raw materials regardless of regional supply constraints. The air-stable reaction conditions eliminate sensitivity to environmental fluctuations during transportation and storage, providing greater flexibility in logistics planning while reducing vulnerability to supply chain disruptions. This inherent robustness creates more predictable lead times compared to conventional methods requiring specialized handling protocols.
- Scalability and Environmental Compliance: The straightforward reaction protocol scales seamlessly from laboratory to commercial production without requiring process re-engineering, enabling rapid technology transfer across different manufacturing capacities. The absence of heavy metal catalysts eliminates hazardous waste streams associated with metal removal processes while reducing overall environmental impact through simplified purification requirements. This green chemistry approach aligns with evolving regulatory expectations while supporting corporate sustainability initiatives through reduced resource consumption.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement and R&D teams during initial technology assessment phases. These responses are derived directly from the patent documentation and experimental validation data provided in CN114920707B, ensuring accurate representation of the technology's capabilities and limitations for pharmaceutical manufacturing applications. Each answer reflects practical implementation experience gained through extensive laboratory validation under conditions mirroring commercial production environments.
Q: How does this method eliminate the need for anhydrous and anaerobic conditions?
A: The iodine-catalyzed reaction operates effectively in air atmosphere due to DMF's dual role as solvent and carbon source, which stabilizes reactive intermediates without requiring moisture-sensitive handling. This eliminates costly glovebox systems and inert gas infrastructure.
Q: What makes DMF uniquely suitable as both solvent and carbon source in this synthesis?
A: DMF provides both methyl and formyl groups that participate in the cyclization mechanism, enabling direct conversion without additional carbon donors. Its high boiling point (153°C) accommodates the required reaction temperature while maintaining solubility across diverse substrate variations.
Q: How does this process ensure high purity for pharmaceutical intermediates?
A: The reaction's inherent selectivity minimizes byproduct formation, while the straightforward post-treatment (filtration followed by column chromatography) effectively removes residual iodine and unreacted materials. This yields consistently high-purity products meeting stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazole Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex fluorinated heterocycles while maintaining stringent purity specifications through rigorous QC labs and advanced analytical capabilities. As a specialized CDMO partner with deep expertise in triazole chemistry, we have successfully implemented this patented methodology across multiple client programs requiring high-purity intermediates for clinical development and commercial manufacturing stages. Our integrated approach combines process chemistry excellence with robust supply chain management to deliver consistent quality and reliable delivery timelines essential for pharmaceutical development programs where material continuity directly impacts clinical trial progression.
For procurement teams seeking tangible value demonstration, we offer a Customized Cost-Saving Analysis that quantifies potential savings through process optimization specific to your manufacturing requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your compound development needs—enabling informed decision-making based on actual technical capabilities rather than theoretical projections.
