Scalable Metal-Free Synthesis of Diarylmethanes: A Strategic Breakthrough for Fine Chemical Manufacturing
Introduction to Advanced Diarylmethane Synthesis
The landscape of fine chemical synthesis is constantly evolving, driven by the urgent need for greener, more cost-effective methodologies that do not compromise on purity or scalability. A pivotal development in this domain is detailed in patent CN112679299B, which discloses a novel preparation method for diarylmethane compounds and their derivatives. This technology represents a significant departure from traditional routes by utilizing cheap, green solid phosphorous acid as a dual-function reduction reagent and accelerant. By initiating the reaction from simple and easily obtainable aryl aldehyde compounds in the presence of elemental iodine, this invention efficiently realizes the preparation of diarylmethane derivatives via a one-pot, one-step method. For R&D directors and procurement managers alike, this approach offers a compelling value proposition: it avoids the use of expensive reduction reagents, toxic metal reagents, and sensitive transition metal catalysts, thereby streamlining the path to industrial production while adhering to stringent environmental standards.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diarylmethane scaffolds has relied on several established but flawed methodologies that pose significant challenges for large-scale manufacturing. The first conventional method involves traditional Friedel-Crafts alkylation, utilizing benzyl halides or benzyl alcohols with equivalent amounts of metal Lewis acids. This approach is plagued by the requirement for excessive metal promoters, which leads to difficult post-treatment processes and generates substantial environmentally unfriendly waste. Furthermore, these reactions often suffer from poor regioselectivity and the formation of numerous byproducts, complicating purification and reducing overall yield. The second method relies on the reduction of diaryl ketones or diaryl methanols, which necessitates dangerous reagents such as hydrogen gas, sodium borohydride, or expensive silane reagents. These raw materials are not only costly but also present significant safety hazards and supply chain vulnerabilities, making them unsuitable for robust industrial production. Finally, transition metal-catalyzed coupling reactions, while effective, require highly active coupling reagents like organic zinc, Grignard, or boron reagents. These reagents are air-sensitive, expensive, and dangerous to handle, while the residual transition metals can severely limit the application range of the final product, particularly in pharmaceutical contexts where metal limits are strictly regulated.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN112679299B introduces a paradigm shift by employing a metal-free catalytic system centered on phosphorous acid and iodine. This novel approach leverages the unique redox properties of phosphorous acid to facilitate the reductive alkylation of aromatic hydrocarbons with aryl aldehydes. The reaction proceeds smoothly under a protective atmosphere at moderate temperatures, typically between 50°C and 150°C, with optimal results observed around 60°C. The simplicity of the operation cannot be overstated; it is a true one-pot process where the aromatic hydrocarbon often serves a dual role as both reactant and solvent. This eliminates the need for additional volatile organic solvents, further enhancing the green chemistry profile of the synthesis. The broad substrate scope allows for the efficient synthesis of various derivatives, accommodating diverse electronic environments on the aromatic rings.
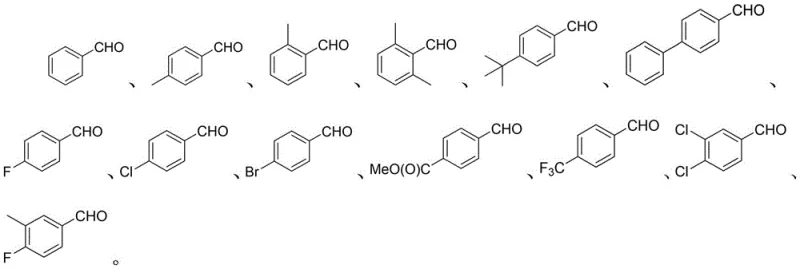
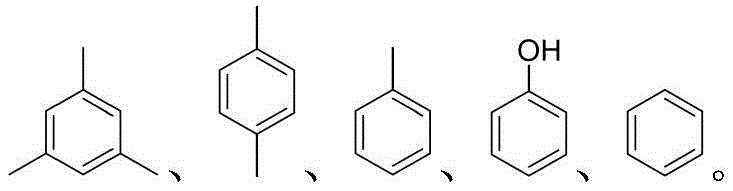
Mechanistic Insights into Phosphorous Acid and Iodine Catalyzed Reductive Alkylation
The mechanistic underpinning of this transformation is a sophisticated interplay between electrophilic activation and in-situ reduction. Elemental iodine likely acts as a Lewis acid activator or generates reactive iodine species that activate the carbonyl group of the aryl aldehyde, facilitating nucleophilic attack by the electron-rich aromatic hydrocarbon. This initial step forms a diaryl methanol intermediate or a related carbocation species. Subsequently, phosphorous acid (H3PO3) functions as a mild yet effective reducing agent. Unlike harsh hydride donors, phosphorous acid selectively reduces the benzylic alcohol or carbocation intermediate to the corresponding methylene bridge without affecting other sensitive functional groups such as esters or halogens. This chemoselectivity is crucial for maintaining the integrity of complex molecular architectures often found in pharmaceutical intermediates. The oxidation product of phosphorous acid is phosphoric acid, which is water-soluble and easily removed during the aqueous workup, leaving no toxic metal residues behind.
From an impurity control perspective, this mechanism offers distinct advantages. The absence of transition metals eliminates the risk of metal-catalyzed side reactions such as homocoupling or over-reduction, which are common pitfalls in palladium or nickel-catalyzed processes. Furthermore, the use of excess aromatic hydrocarbon drives the reaction kinetics towards the desired mono-alkylated product, minimizing the formation of poly-alkylated byproducts. The reaction conditions are mild enough to prevent thermal degradation of sensitive substrates, ensuring a clean impurity profile. Post-reaction treatment with sodium thiosulfate effectively quenches any residual iodine, preventing oxidative side reactions during workup. This streamlined purification process, typically involving extraction and column chromatography, results in high-purity products suitable for downstream applications without the need for complex scavenging resins or recrystallization steps often required to remove metal catalysts.
How to Synthesize Diarylmethane Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and atmospheric conditions to maximize yield and safety. The process begins by charging the reaction vessel with the aryl aldehyde, elemental iodine, and phosphorous acid under a nitrogen atmosphere to prevent oxidation of sensitive reagents. The aromatic hydrocarbon is added in significant excess, serving as the reaction medium. The mixture is then heated to the specified temperature range and maintained with stirring for a duration of 12 to 24 hours. Upon completion, the reaction mixture is treated with an aqueous solution of sodium thiosulfate to neutralize iodine, followed by standard extraction and drying procedures. The detailed standardized synthetic steps, including specific molar ratios and purification parameters for various substrates, are outlined in the guide below.
- Mix aryl aldehyde, elemental iodine, and phosphorous acid with an excess of aromatic hydrocarbon compound (acting as both reactant and solvent) in a reaction flask under a nitrogen atmosphere.
- Heat the reaction mixture to a temperature between 50°C and 150°C (optimally 60°C) and stir continuously for 12 to 24 hours to ensure complete conversion.
- Quench the reaction with aqueous sodium thiosulfate, extract the organic phase with ethyl acetate, dry, filter, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this phosphorous acid-mediated synthesis offers transformative benefits that directly impact the bottom line and operational resilience. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing expensive transition metal catalysts and hazardous hydride reagents with commodity chemicals like phosphorous acid and iodine, manufacturers can achieve substantial cost savings. These reagents are globally available, stable, and inexpensive, insulating the supply chain from the volatility associated with precious metal markets. Furthermore, the elimination of heavy metals from the process flow removes the need for costly metal scavenging steps and rigorous testing for residual metals, which are mandatory for pharmaceutical grade intermediates. This reduction in processing steps translates directly into shorter cycle times and lower utility consumption.
- Cost Reduction in Manufacturing: The economic implications of switching to this metal-free protocol are profound. Traditional methods often incur high costs due to the purchase of noble metal catalysts and the subsequent disposal of metal-contaminated waste. By utilizing phosphorous acid, a low-cost industrial chemical, the direct material cost is significantly lowered. Additionally, the one-pot nature of the reaction reduces labor costs and equipment occupancy time. The avoidance of specialized reagents like Grignard reagents, which require strict anhydrous conditions and low temperatures, further reduces energy consumption and infrastructure requirements. This holistic reduction in operational expenditure makes the production of diarylmethane derivatives far more economically viable on a commercial scale.
- Enhanced Supply Chain Reliability: Supply chain continuity is critical for meeting the demands of downstream pharmaceutical clients. The reagents used in this novel method—aryl aldehydes, aromatic hydrocarbons, phosphorous acid, and iodine—are all bulk commodities with robust global supply chains. Unlike specialized organometallic reagents that may have long lead times or single-source suppliers, these materials are readily accessible from multiple vendors. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. Moreover, the stability of solid phosphorous acid simplifies storage and logistics compared to pyrophoric or moisture-sensitive reagents, enhancing overall warehouse safety and inventory management efficiency.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production is often hindered by safety and environmental concerns. This methodology addresses both issues effectively. The reaction operates at moderate temperatures and does not involve high-pressure hydrogenation or exothermic hydride additions, making it inherently safer for large-scale reactors. From an environmental standpoint, the process aligns with green chemistry principles by minimizing waste generation and avoiding toxic heavy metals. The byproduct, phosphoric acid, is benign and easily managed in wastewater treatment systems. This compliance with increasingly stringent environmental regulations ensures long-term operational sustainability and reduces the regulatory burden on the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diarylmethane synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing workflows.
Q: What are the primary advantages of using phosphorous acid over traditional Lewis acids in diarylmethane synthesis?
A: Phosphorous acid serves as a cheap, green, solid reduction reagent and accelerant, eliminating the need for expensive transition metal catalysts or hazardous reducing agents like sodium borohydride. This significantly simplifies post-treatment by avoiding heavy metal residue removal, thereby enhancing product purity and environmental compliance.
Q: Can this method tolerate diverse functional groups on the aryl aldehyde substrate?
A: Yes, the protocol demonstrates excellent functional group tolerance. It successfully accommodates electron-withdrawing groups such as halogens (F, Cl, Br), trifluoromethyl, and esters, as well as electron-donating alkyl groups, yielding the corresponding diarylmethane derivatives with high efficiency.
Q: How does the molar ratio of reactants influence the yield in this process?
A: Optimization data suggests that using the aromatic hydrocarbon in large excess (molar ratio of aldehyde to aromatic compound up to 1:22.5) acts as both reactant and solvent, driving the equilibrium forward and improving yields. The optimal molar ratio of aryl aldehyde to phosphorous acid is typically between 1:2 and 1:2.5.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diarylmethane Supplier
The technological potential of phosphorous acid-catalyzed reductive alkylation represents a significant opportunity for optimizing the production of high-value fine chemicals. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of diarylmethane intermediate meets the exacting standards required by the global pharmaceutical industry. We understand the critical nature of supply chain consistency and are equipped to handle complex synthetic challenges with precision.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and purity targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply of high-purity diarylmethane derivatives produced via a sustainable and cost-effective methodology, securing your position in a competitive market.
