Advanced Synthesis of Indolizine-Containing Diarylmethane Derivatives for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking novel heterocyclic scaffolds that offer enhanced biological profiles, particularly in the realms of oncology and infectious disease treatment. Patent CN114163436A introduces a significant breakthrough in this domain by disclosing a series of diarylmethane derivatives containing an indolizine core. These unique molecular architectures simultaneously incorporate indolizine and either benzofuran or indole moieties, creating a privileged structure known for its potent antibacterial and anticancer activities. Unlike previous iterations of indolizine-containing compounds which often suffered from limited biological scope, such as single-activity phospholipase inhibition, these new derivatives offer a broader therapeutic potential. Furthermore, beyond their direct pharmacological value, these compounds serve as versatile intermediates for synthesizing other functional organic molecules, including fluorescent probes, thereby expanding their utility in both drug research and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of complex diarylmethane frameworks bearing multiple fused heterocycles has been a formidable challenge for synthetic chemists. Conventional methodologies often rely on stepwise assembly, where the indolizine core and the pendant aryl groups are constructed in separate, discrete stages. This fragmented approach not only results in lower overall yields due to cumulative losses at each isolation step but also generates substantial chemical waste, violating the principles of green chemistry. Moreover, traditional routes frequently require harsh reaction conditions, such as strong acids or high temperatures, which can lead to the decomposition of sensitive functional groups or the formation of difficult-to-remove impurities. The reliance on pre-functionalized starting materials that are expensive or difficult to source further exacerbates the cost and supply chain risks associated with these legacy manufacturing processes.
The Novel Approach
In stark contrast, the methodology described in the patent utilizes an elegant one-pot tandem cyclization strategy that dramatically streamlines the synthesis. By reacting a 2-iodophenyl allene compound with a propargyl pyridine derivative under palladium catalysis, the entire fused ring system is assembled in a single operational step. This cascade reaction efficiently forms multiple carbon-carbon and carbon-heteroatom bonds simultaneously, significantly improving atom economy and reducing the environmental footprint. The versatility of this approach is highlighted by its tolerance for various substituents, allowing for the rapid generation of diverse libraries of compounds for structure-activity relationship studies. 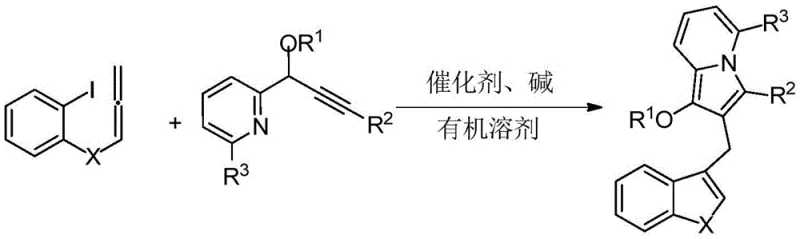
Mechanistic Insights into Pd-Catalyzed Tandem Cyclization
The core of this transformative synthesis lies in the sophisticated palladium-catalyzed mechanism that drives the tandem cyclization. The reaction initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the 2-iodophenyl allene substrate, generating a reactive organopalladium intermediate. This species then undergoes a migratory insertion into the alkyne moiety of the propargyl pyridine component, setting the stage for the subsequent ring-closing events. The precise control over regioselectivity is crucial here, ensuring that the indolizine core is formed with the correct substitution pattern, which is vital for the resulting biological activity. The presence of the base, typically potassium carbonate, facilitates the deprotonation steps necessary to regenerate the active catalyst and drive the equilibrium towards the desired product.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The mild reaction conditions, typically maintained between 60°C and 90°C, minimize the risk of thermal degradation or polymerization of the reactive allene and alkyne precursors. Furthermore, the use of standard ligands like triphenylphosphine helps stabilize the palladium center, preventing the formation of palladium black which can act as a sink for the catalyst and a source of metal contamination. The selectivity of the cyclization ensures that side reactions, such as homocoupling of the alkyne or allene, are suppressed, leading to a cleaner crude reaction profile that simplifies downstream purification efforts significantly.
How to Synthesize Indolizine Diarylmethane Derivatives Efficiently
The practical execution of this synthesis is designed to be robust and scalable, utilizing reagents and solvents that are readily available in standard chemical inventories. The process begins by combining the 2-iodophenyl allene and the propargyl pyridine substrates in a polar aprotic solvent such as N,N-dimethylformamide, which effectively solubilizes both organic reactants and the inorganic base. The reaction is conducted under an inert nitrogen atmosphere to protect the sensitive palladium catalyst from oxidation, ensuring consistent performance throughout the batch. Detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below to ensure reproducibility and high purity for your R&D teams.
- Mix 2-iodophenyl allene compound, propargyl pyridine compound, palladium catalyst, alkali, and organic solvent in a reaction vessel.
- Stir and react the mixture for 4-12 hours at a temperature range of 60-90°C under an inert nitrogen atmosphere.
- Perform post-treatment including extraction with ethyl acetate, drying, filtration, solvent removal, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route presents a compelling value proposition centered on cost efficiency and supply reliability. The elimination of multiple synthetic steps translates directly into reduced labor costs, lower energy consumption, and decreased solvent usage, all of which contribute to a significantly reduced cost of goods sold. By consolidating the synthesis into a single pot, manufacturers can also reduce the turnaround time for production batches, allowing for more responsive inventory management and faster time-to-market for new drug candidates. This efficiency is critical in the competitive landscape of pharmaceutical intermediates where speed and cost are paramount.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of inexpensive and commercially accessible starting materials, such as substituted pyridines and iodobenzenes, which avoids the premium pricing associated with exotic reagents. Additionally, the high atom economy of the tandem cyclization means that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal costs. The avoidance of complex protection and deprotection strategies further simplifies the bill of materials, leading to substantial cost savings in the overall manufacturing budget without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the fact that the key reagents, including the palladium catalyst and inorganic bases like potassium carbonate, are commodity chemicals with stable global supply lines. This reduces the risk of production delays caused by the shortage of specialized custom synthons that often plague complex multi-step syntheses. Furthermore, the robustness of the reaction conditions allows for flexibility in sourcing, as the process tolerates minor variations in reagent grade, ensuring that production can continue even if specific supplier specifications fluctuate slightly.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard solvents like DMF and toluene, which are well-understood in large-scale reactor operations. The simplified workup procedure, involving standard extraction and chromatography, is easily adaptable to continuous processing or large-batch manufacturing, ensuring consistent quality at higher volumes. From an environmental standpoint, the reduction in step count inherently lowers the E-factor (mass of waste per mass of product), aiding facilities in meeting increasingly stringent regulatory requirements for green manufacturing and waste minimization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indolizine derivatives. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy for your decision-making process. Understanding these details is essential for evaluating the feasibility of integrating this technology into your existing pipeline.
Q: What are the key biological activities of these indolizine derivatives?
A: According to patent CN114163436A, these compounds exhibit significant antibacterial and anticancer activities, making them valuable candidates for drug development.
Q: What catalyst system is used in this novel synthesis method?
A: The process utilizes a palladium catalyst, such as tetrakis(triphenylphosphine)palladium, in combination with an inorganic base like potassium carbonate.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the one-pot tandem cyclization strategy simplifies the workflow and uses common solvents like DMF, facilitating easier scale-up compared to multi-step conventional routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine Diarylmethane Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed technology in accelerating the development of next-generation therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of indolizine intermediate delivered meets the highest standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this efficient methodology can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a reliable supply of these high-value pharmaceutical intermediates.
