Advanced Palladium-Catalyzed Synthesis of C-3 Benzyl Indolizine Compounds for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN110357879B discloses a significant advancement in the preparation of C-3 benzyl indolizine compounds, a scaffold known for potent antitumor and phosphodiesterase inhibitory activities. This novel methodology leverages a palladium-catalyzed annulation strategy that overcomes the historical inefficiencies associated with indolizine construction. By utilizing readily available 2-alkylpyridines and propargyl carbonates, the process achieves high atom economy and operational simplicity. For R&D directors and procurement specialists, this represents a critical opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering complex heterocyclic building blocks with consistent quality. The transition from legacy photochemical or thermal carbene methods to this transition-metal catalyzed approach marks a pivotal shift towards more sustainable and scalable manufacturing paradigms in fine chemical synthesis.
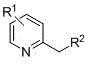
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indolizine derivatives has been plagued by significant technical hurdles that hinder large-scale production. Prior art, such as the reaction of 2-vinylpyridine with chlorocarbene, requires extreme conditions involving high-intensity illumination or rigorous heating, which poses safety risks and energy inefficiencies. Furthermore, these conventional pathways exhibit poor functional group tolerance, severely restricting the chemical space accessible to medicinal chemists. The low yields often observed in these traditional protocols necessitate extensive purification efforts, driving up the cost of goods sold and creating bottlenecks in the supply chain. For a procurement manager, relying on such unstable processes translates to volatile pricing and unpredictable lead times, making it difficult to plan for commercial scale-up of complex pharmaceutical intermediates. The inability to easily modify substituents without crashing the reaction yield further limits the utility of these older methods in modern drug discovery campaigns where rapid analog synthesis is paramount.
The Novel Approach
In stark contrast, the method described in CN110357879B introduces a streamlined catalytic cycle that operates under thermal conditions between 100°C and 140°C, eliminating the need for hazardous photochemical setups. This approach utilizes a synergistic combination of a zero-valent palladium catalyst and specialized phosphine ligands to activate the C-H bond of the 2-alkylpyridine, facilitating a smooth cyclization with the propargyl carbonate. The reaction demonstrates exceptional versatility, accommodating a wide range of electron-withdrawing and electron-donating groups on both the pyridine and alkyne components. This flexibility allows for the direct installation of diverse functional handles, such as esters and nitriles, which are crucial for downstream derivatization into active pharmaceutical ingredients. By simplifying the post-treatment to basic filtration and column chromatography, the novel approach drastically reduces processing time and solvent consumption. This operational ease ensures that the production of high-purity OLED material precursors or drug intermediates can be achieved with minimal technical friction, enhancing overall process reliability.
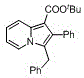
Mechanistic Insights into Pd-Catalyzed Cyclization
The core of this technological breakthrough lies in the intricate mechanistic pathway governed by the palladium catalyst. The reaction initiates with the oxidative addition of the propargyl carbonate to the Pd(0) species, generating a reactive pi-allyl palladium intermediate. This is followed by a nucleophilic attack or migratory insertion involving the 2-alkylpyridine, which is activated by the base present in the system. The choice of ligand, such as 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (Xantphos), is critical as its large bite angle stabilizes the palladium center and promotes the reductive elimination step that closes the five-membered ring. Understanding this catalytic cycle is essential for R&D teams aiming to optimize reaction parameters for specific substrates. The mechanism ensures that side reactions, such as polymerization of the alkyne or decomposition of the sensitive carbonate, are minimized through precise control of the electronic environment around the metal center. This level of mechanistic control is what differentiates a laboratory curiosity from a commercially viable manufacturing process.
Impurity control is another vital aspect addressed by this catalytic system. In traditional syntheses, side products often arise from uncontrolled radical pathways or incomplete conversions, leading to difficult-to-remove impurities that compromise the purity profile of the final API. The palladium-catalyzed route described here favors a concerted mechanism that inherently suppresses the formation of byproducts. The use of polar aprotic solvents like DMSO or DMF further enhances the solubility of ionic intermediates and stabilizes the transition states, leading to cleaner reaction profiles. For quality assurance teams, this means that the resulting C-3 benzyl indolizine compounds exhibit a superior impurity spectrum, reducing the burden on downstream purification units. The ability to tune the steric bulk of the ligand allows for further refinement of selectivity, ensuring that even sterically hindered substrates can be converted with high fidelity. This robustness is a key factor in maintaining stringent purity specifications required for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize C-3 Benzyl Indolizine Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and environmental controls as outlined in the patent examples. The process begins with the careful weighing of the metal catalyst and ligand under inert atmosphere to prevent oxidation of the active Pd(0) species. The detailed standardized synthesis steps see the guide below.
- Combine metal palladium catalyst, phosphine ligand, 2-alkylpyridine, propargyl carbonate, and base in an organic solvent such as DMSO or DMF.
- Heat the reaction mixture to a temperature range of 100-140°C and maintain stirring for 10 to 20 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography using ethyl acetate and petroleum ether to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the adoption of this palladium-catalyzed methodology offers transformative benefits regarding cost structure and logistical stability. The reliance on commodity chemicals like 2-alkylpyridines and simple propargyl carbonates eliminates the need for exotic, high-cost starting materials that are often subject to market volatility. This shift to abundant raw materials significantly de-risks the supply chain, ensuring continuous availability even during global disruptions. Furthermore, the simplified workup procedure reduces the consumption of auxiliary materials and solvents, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. The robustness of the reaction conditions means that batch-to-batch variability is minimized, allowing for more accurate forecasting and inventory management. By partnering with a supplier who has mastered this technology, companies can secure a steady flow of critical building blocks without the fear of production stoppages due to technical failures.
- Cost Reduction in Manufacturing: The elimination of expensive photochemical equipment and the reduction in reaction time lead to substantial operational savings. Since the catalyst loading is kept low and the ligands are recyclable in optimized processes, the overall catalyst cost per kilogram of product is drastically reduced. Additionally, the high conversion rates minimize the loss of valuable starting materials, improving the overall mass balance of the process. These efficiencies compound to lower the total cost of ownership for the intermediate, allowing pharmaceutical companies to allocate resources to other critical areas of drug development. The avoidance of hazardous reagents also lowers waste disposal costs, adding another layer of financial benefit to the operation.
- Enhanced Supply Chain Reliability: The use of thermally stable reagents and standard heating equipment makes this process highly transferable across different manufacturing sites. Unlike light-dependent reactions that require specialized reactors, this thermal process can be executed in standard stainless steel vessels found in most multipurpose plants. This universality ensures that production can be scaled or shifted geographically without significant capital expenditure. The robustness of the chemistry against moisture and air variations further enhances reliability, reducing the risk of batch failures. For procurement managers, this translates to a dependable source of supply that can meet tight deadlines and fluctuating demand schedules with confidence.
- Scalability and Environmental Compliance: The reaction operates in polar solvents that are well-understood in industrial settings, facilitating easy scale-up from gram to ton quantities. The absence of toxic chlorocarbene reagents aligns with modern green chemistry principles, simplifying environmental permitting and compliance reporting. Waste streams are easier to treat due to the lack of halogenated byproducts, reducing the environmental footprint of the manufacturing process. This sustainability profile is increasingly important for multinational corporations aiming to meet their corporate social responsibility goals. The ability to produce large quantities of high-purity material with minimal environmental impact positions this technology as a preferred choice for long-term strategic sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent literature to provide clarity for potential partners.
Q: What are the primary limitations of conventional indolizine synthesis methods?
A: Traditional methods often rely on the reaction of 2-vinylpyridine with chlorocarbene under harsh heating or illumination conditions. These legacy processes suffer from poor applicability regarding substituent groups and frequently result in low yields, limiting their utility for diverse drug discovery programs.
Q: Which catalyst systems are most effective for this transformation?
A: The process utilizes zero-valent palladium catalysts, specifically tris(dibenzylideneacetone)dipalladium or tetrakis(triphenylphosphine)palladium. These are paired with bulky phosphine ligands like 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene to facilitate the cyclization efficiently.
Q: Does this method support structural diversity for medicinal chemistry?
A: Yes, the method exhibits strong substrate designability. It tolerates various substituents on the pyridine ring (such as methyl or hydrogen) and accommodates different ester groups (methyl, ethyl, tert-butyl) or cyano groups on the alkyne component, allowing for the rapid generation of analog libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-3 Benzyl Indolizine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We have invested heavily in rigorous QC labs and state-of-the-art analytical instrumentation to guarantee stringent purity specifications for every batch of C-3 benzyl indolizine we produce. Our commitment to technical excellence means that we can navigate the complexities of palladium catalysis to deliver material that meets the highest industry standards. Whether you require custom analogs for SAR studies or bulk quantities for clinical trials, our infrastructure is designed to support your timeline and quality requirements seamlessly.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this methodology for your specific application. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique molecular targets. Let us collaborate to accelerate your drug development pipeline with reliable, cost-effective, and high-performance chemical solutions.
