Advanced Catalytic Synthesis of C-3 Benzyl Indolizine Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access nitrogen-containing heterocycles, particularly indolizine derivatives, due to their profound biological significance. Patent CN110357879B introduces a groundbreaking preparation method for C-3 benzyl indolizine compounds that addresses long-standing synthetic challenges. These compounds are not merely academic curiosities; they serve as critical scaffolds in medicinal chemistry, exhibiting potent antitumor activities and phosphodiesterase inhibition capabilities. The structural versatility of the indolizine core allows for extensive modification, making it a high-value target for drug discovery programs aiming to develop next-generation therapeutics. By leveraging a palladium-catalyzed annulation strategy, this technology transforms simple, commercially available starting materials into complex heterocyclic architectures with remarkable efficiency.
Historically, the synthesis of C-3 benzyl indolizine structures has been fraught with significant technical hurdles that limit their widespread adoption in industrial settings. Conventional literature methods often rely on the reaction of 2-vinylpyridine with chlorocarbene under harsh heating or irradiation conditions. These legacy processes are plagued by severe limitations, including poor applicability across diverse substituent groups and consistently low reaction yields. Furthermore, the handling of chlorocarbene precursors poses safety risks and environmental concerns, complicating the regulatory approval process for large-scale manufacturing. The inability to easily tune the molecular structure restricts the medicinal chemist's ability to optimize pharmacokinetic properties, thereby slowing down the overall drug development timeline. Consequently, there is an urgent demand for a safer, more versatile, and higher-yielding synthetic route that can support the rigorous demands of modern pharmaceutical supply chains.
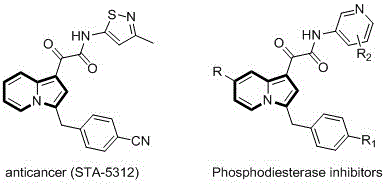
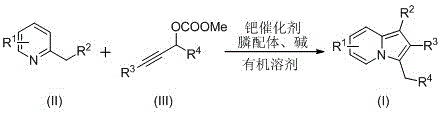
The novel approach detailed in the patent data revolutionizes this landscape by employing a transition metal-catalyzed cyclization strategy that is both operationally simple and chemically robust. Instead of hazardous carbenes, this method utilizes 2-alkylpyridines and propargyl carbonates as the primary building blocks, reacting them in the presence of a metal palladium catalyst and a phosphine ligand. This shift in reagents dramatically expands the scope of accessible derivatives, allowing for the precise installation of various functional groups at the C-3 position. The reaction proceeds smoothly in polar organic solvents at moderate temperatures, eliminating the need for extreme conditions that often degrade sensitive functional groups. This methodological advancement ensures that the resulting C-3 benzyl indolizine compounds are obtained with high purity and structural integrity, providing a reliable foundation for subsequent biological evaluation and process development.
Mechanistic Insights into Palladium-Catalyzed Indolizine Cyclization
At the heart of this synthesis lies a sophisticated palladium-catalyzed mechanism that facilitates the construction of the indolizine ring system through a series of well-defined organometallic steps. The reaction initiates with the oxidative addition of the zero-valent palladium species into the propargyl carbonate, generating a reactive pi-allyl palladium intermediate. This electrophilic species is then attacked by the nucleophilic nitrogen atom of the 2-alkylpyridine, forming a crucial C-N bond that sets the stage for ring closure. The presence of specialized phosphine ligands, such as 4,6-bis(diphenylphosphino)-dibenzofuran, is critical in stabilizing the palladium center and modulating its electronic properties to favor the desired cyclization pathway over competing side reactions. This ligand acceleration effect ensures high turnover numbers and minimizes the formation of palladium black, which can otherwise lead to catalyst deactivation and reduced yields.
Controlling the impurity profile is paramount for any intermediate intended for pharmaceutical use, and this catalytic system offers inherent advantages in selectivity. The use of mild bases like potassium carbonate or cesium carbonate facilitates the final deprotonation and aromatization steps without promoting unwanted hydrolysis of the ester or nitrile functionalities often present in the substrate. By carefully optimizing the molar ratios of the reactants, specifically using an excess of the 2-alkylpyridine, the process drives the equilibrium towards the product while suppressing homocoupling side products. The result is a crude reaction mixture that is significantly cleaner than those produced by traditional methods, simplifying the downstream purification process. This high level of chemoselectivity is essential for maintaining the stringent quality standards required by global regulatory bodies, ensuring that the final API intermediates meet exacting specifications for heavy metal residues and organic impurities.
How to Synthesize C-3 Benzyl Indolizine Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility on a production scale. The process involves dissolving the metal catalyst, ligand, and base in a polar aprotic solvent such as dimethyl sulfoxide (DMSO) or N,N-dimethylformamide (DMF) before introducing the substrates. Heating the mixture to between 100°C and 140°C for a duration of 10 to 20 hours allows the cyclization to reach completion. Following the reaction, the workup procedure is straightforward, involving filtration to remove insoluble salts followed by standard chromatographic purification. For detailed operational parameters and specific stoichiometric ratios validated through experimental examples, please refer to the standardized guide below.
- Combine metal palladium catalyst, phosphine ligand, 2-alkylpyridine, propargyl carbonate, and base in a polar organic solvent such as DMSO or DMF.
- Heat the reaction mixture to a temperature range of 100-140°C and maintain stirring for 10 to 20 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography using ethyl acetate and petroleum ether to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this patented synthesis route offers substantial benefits that extend beyond mere chemical novelty. The shift towards using readily available 2-alkylpyridines and propargyl carbonates significantly mitigates supply chain risks associated with specialty reagents that have limited global sourcing options. By relying on commodity chemicals, manufacturers can secure long-term contracts with multiple vendors, ensuring continuity of supply even during market fluctuations. Furthermore, the operational simplicity of the process reduces the dependency on highly specialized equipment or extreme safety protocols, lowering the barrier to entry for contract manufacturing organizations. This accessibility translates directly into a more resilient supply chain capable of responding rapidly to increased demand from downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of hazardous chlorocarbene reagents and the associated safety infrastructure leads to a drastic simplification of the production workflow. By utilizing inexpensive palladium catalysts in conjunction with efficient ligands, the process achieves high conversion rates that minimize raw material waste. The simplified post-treatment procedure, which avoids complex extraction or distillation steps, further reduces energy consumption and labor costs. These cumulative efficiencies result in significant cost savings per kilogram of the final intermediate, allowing procurement teams to negotiate more competitive pricing structures without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system ensures consistent batch-to-batch reproducibility, a critical factor for maintaining trust with international partners. Since the reaction tolerates a wide range of substituents, the same core process can be adapted to produce various analogues without requiring complete re-validation of the manufacturing line. This flexibility allows suppliers to pivot quickly between different product SKUs based on market needs, reducing lead times for high-purity pharmaceutical intermediates. Additionally, the use of stable starting materials eliminates the risk of degradation during storage and transport, guaranteeing that raw materials arrive at the production site in optimal condition.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial tonnage is facilitated by the use of standard polar solvents and common inorganic bases that are easily handled in large reactors. The absence of toxic gaseous byproducts simplifies waste gas treatment systems, aligning the process with increasingly stringent environmental regulations. The high atom economy of the cyclization step means less chemical waste is generated per unit of product, supporting sustainability goals and reducing disposal costs. This environmentally friendly profile enhances the corporate reputation of manufacturers and ensures compliance with green chemistry initiatives demanded by major multinational corporations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its potential for integration into existing portfolios. The following questions address common inquiries regarding the practical implementation and advantages of this technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a factual basis for decision-making. Clients are encouraged to review these insights to assess the feasibility of adopting this route for their specific project requirements.
Q: What are the key advantages of this Pd-catalyzed method over conventional synthesis?
A: Unlike conventional methods involving chlorocarbene which suffer from poor substituent applicability and low yields, this novel approach utilizes readily available 2-alkylpyridines and propargyl carbonates, offering superior substrate designability and operational simplicity.
Q: Which catalysts and ligands are optimal for this transformation?
A: The process demonstrates high efficiency using zero-valent palladium catalysts such as tris(dibenzylideneacetone)dipalladium paired with bidentate phosphine ligands like 4,6-bis(diphenylphosphino)-dibenzofuran.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method employs inexpensive raw materials and standard post-processing techniques like filtration and column chromatography, making it highly practical for commercial scale-up and consistent supply chain integration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-3 Benzyl Indolizine Supplier
As the demand for complex heterocyclic intermediates continues to grow, partnering with an experienced CDMO is vital for translating innovative chemistry into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to plant. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of C-3 benzyl indolizine meets the highest international standards. We understand the critical nature of timeline and quality in the pharmaceutical sector and are committed to delivering excellence at every stage of the manufacturing process.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Together, we can accelerate your path to market with a reliable, cost-effective, and scalable supply solution.
