Advanced Catalytic Production of High-Purity Quinolinone Derivatives for Scalable Pharmaceutical Intermediate Manufacturing
The recently granted Chinese patent CN113045489B represents a significant advancement in heterocyclic compound synthesis through its innovative approach to producing structurally diverse quinolinone derivatives. This patented methodology addresses critical challenges in pharmaceutical intermediate manufacturing by introducing a dual-functional benzisoxazole reagent that serves simultaneously as both nitrogen source and formyl donor within a palladium-catalyzed framework. The process demonstrates remarkable versatility across various substrate combinations while maintaining operational simplicity that directly translates to enhanced commercial viability for global pharmaceutical supply chains. By eliminating multi-step sequences required by conventional methods like Vilsmeier Haack or Knorr reactions, this technology establishes a new paradigm for efficient production of bioactive quinoline scaffolds essential in modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for quinolinone derivatives typically involve harsh reaction conditions that present substantial operational challenges for pharmaceutical manufacturers. Vilsmeier Haack reactions require highly corrosive reagents under strong acidic conditions at elevated temperatures, creating significant safety hazards and complex waste streams that increase environmental compliance costs. Knorr cyclization methods often suffer from narrow substrate scope with poor functional group tolerance, particularly when handling sensitive moieties common in modern drug molecules. Friedlander condensations frequently produce low yields due to competitive side reactions and necessitate extensive purification procedures that diminish overall process efficiency. These conventional approaches also demand multiple synthetic steps with intermediate isolations, substantially increasing both production timelines and raw material consumption while introducing additional quality control variables that compromise batch consistency.
The Novel Approach
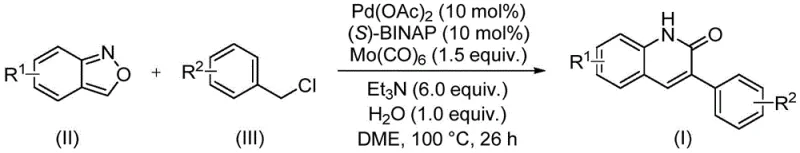
The patented methodology overcomes these limitations through an elegant single-step transformation that leverages benzisoxazole's unique dual functionality within a carefully optimized catalytic system. By employing palladium acetate with (S)-BINAP ligand alongside molybdenum carbonyl as carbon monoxide source, the process achieves high-yielding conversions under mild conditions at precisely controlled temperatures of 100°C without requiring specialized high-pressure equipment. This approach maintains exceptional functional group tolerance across diverse substituents including halogens, alkoxy groups, and cyano functionalities while utilizing commercially available starting materials that are both cost-effective and readily accessible in bulk quantities. The streamlined reaction profile eliminates multiple intermediate steps required by traditional methods, significantly reducing overall process complexity while delivering consistent product quality essential for pharmaceutical applications.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The catalytic cycle begins with oxidative addition of benzyl chloride to palladium(0), generated in situ from palladium acetate reduction by triethylamine. This forms an arylpalladium species that undergoes transmetalation with benzisoxazole through its nitrogen atom, followed by CO insertion from molybdenum carbonyl to create an acylpalladium intermediate. Subsequent intramolecular nucleophilic attack by the carbonyl oxygen leads to ring closure and reductive elimination releases the quinolinone product while regenerating the active catalyst species. This mechanism explains the exceptional functional group tolerance observed across various substrates as the reaction proceeds through stable organopalladium intermediates that minimize undesired side reactions. The dual role of benzisoxazole as both nitrogen donor and formyl source eliminates external carbon monoxide handling requirements while maintaining precise control over the carbonylation step.
Impurity control is achieved through the selective nature of the catalytic transformation where competing pathways are minimized by the specific coordination environment created by the (S)-BINAP ligand. The reaction demonstrates remarkable consistency across diverse substituents as evidenced by high yields maintained even with electron-withdrawing groups like cyano or trifluoromethyl moieties that typically complicate traditional syntheses. The aqueous workup procedure effectively removes polar impurities while column chromatography purification targets residual catalyst species without requiring specialized techniques. This inherent selectivity reduces the formation of regioisomeric byproducts common in alternative methods while maintaining excellent product purity profiles essential for pharmaceutical applications where strict impurity thresholds must be met.
How to Synthesize Quinolinone Derivatives Efficiently
This innovative synthesis pathway represents a significant advancement in quinolinone production methodology through its strategic use of commercially available starting materials under precisely controlled catalytic conditions. The process demonstrates exceptional versatility across multiple substrate combinations while maintaining operational simplicity that directly translates to enhanced commercial viability for global pharmaceutical manufacturers seeking reliable sources of high-purity intermediates. Detailed standardized synthesis procedures have been developed based on extensive optimization studies documented in the patent literature; these protocols ensure consistent product quality while accommodating diverse manufacturing scales from laboratory to commercial production environments.
- Combine benzisoxazole compound (II), benzyl chloride derivative (III), palladium acetate catalyst (0.1 mol%), (S)-BINAP ligand (0.1 mol%), molybdenum carbonyl (1.5 equiv.), triethylamine (6.0 equiv.), and water (1.0 equiv.) in ethylene glycol dimethyl ether solvent under inert atmosphere.
- Heat the reaction mixture at precisely 100°C for a controlled duration of 26 hours to ensure complete conversion while maintaining optimal catalyst activity and minimizing side reactions.
- Execute post-processing through filtration to remove catalyst residues, followed by silica gel mixing and column chromatography purification to isolate high-purity quinolinone derivatives meeting stringent pharmaceutical specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial value across procurement and supply chain functions by addressing critical pain points inherent in traditional quinolinone production systems. The elimination of multi-step sequences required by conventional approaches significantly reduces raw material consumption while minimizing quality control variables that impact batch consistency and regulatory compliance timelines. By utilizing readily available starting materials that can be sourced from multiple global suppliers without specialized handling requirements, the process enhances supply chain resilience while reducing dependency on single-source vendors that create vulnerability to market fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts required by alternative methods substantially reduces raw material costs while simplifying waste stream management through aqueous workup procedures that avoid hazardous reagents. The use of commercially available starting materials at favorable price points combined with high-yielding transformations minimizes overall production expenses without requiring capital-intensive equipment upgrades or specialized infrastructure investments.
- Enhanced Supply Chain Reliability: The broad availability of starting materials from multiple global suppliers significantly reduces sourcing risks while enabling flexible production scheduling that accommodates fluctuating demand patterns common in pharmaceutical manufacturing environments. The simplified process flow with fewer critical control points enhances production predictability while minimizing potential disruption points that could impact delivery timelines for time-sensitive pharmaceutical development programs.
- Scalability and Environmental Compliance: The robust reaction parameters operate effectively across multiple scales without requiring specialized equipment modifications, enabling seamless transition from laboratory development to commercial production volumes while maintaining consistent product quality profiles. The aqueous-based workup procedure generates less hazardous waste compared to traditional methods using strong acids or toxic reagents, significantly reducing environmental compliance burdens while aligning with increasingly stringent global sustainability regulations.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patented methodology's implementation parameters and performance characteristics observed during extensive validation studies. These insights reflect practical experience gained from scaling similar catalytic processes within our manufacturing facilities while addressing specific concerns raised by global pharmaceutical clients regarding process reliability and quality assurance protocols.
Q: How does this method overcome limitations of traditional quinolinone synthesis routes?
A: The patented approach eliminates harsh reaction conditions required by conventional Vilsmeier Haack or Friedlander methods by utilizing benzisoxazole as a dual nitrogen and formyl source. This innovation avoids toxic reagents and high-pressure systems while maintaining broad functional group tolerance across diverse substrates.
Q: What specific advantages does this process offer for pharmaceutical intermediate manufacturing?
A: The methodology provides significant operational advantages through inexpensive starting materials that are commercially available in bulk quantities. The simplified workup procedure using standard column chromatography reduces purification complexity while delivering consistent high-purity outputs essential for API synthesis.
Q: Can this synthesis be reliably scaled for commercial production volumes?
A: Yes, the robust reaction parameters operate effectively across multiple scales without requiring specialized equipment. The consistent yields demonstrated across varied substituents confirm scalability from laboratory to industrial production while maintaining product quality standards required by regulatory agencies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolinone Derivative Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities through our state-of-the-art QC labs equipped with advanced analytical instrumentation. We have successfully implemented this patented methodology within our manufacturing infrastructure to produce high-purity quinolinone derivatives meeting exacting pharmaceutical standards while optimizing cost structures through continuous process improvement initiatives that enhance overall operational efficiency across our supply chain network.
Request our Customized Cost-Saving Analysis today to evaluate how this innovative synthesis approach can optimize your specific production requirements; our technical procurement team stands ready to provide detailed COA data and comprehensive route feasibility assessments tailored to your unique manufacturing needs.
