Advanced Palladium-Catalyzed Synthesis Platform for Commercial-Scale Quinolinone Derivative Manufacturing
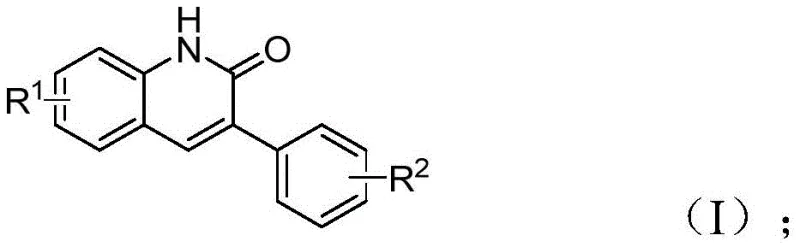
Patent CN113045489B, granted on June 21, 2022, introduces a groundbreaking synthetic methodology for 3-arylquinolin-2(1H)one derivatives, representing a significant advancement in heterocyclic compound synthesis for pharmaceutical applications. This innovative approach utilizes benzisoxazole as both nitrogen and formyl source, enabling the efficient construction of complex quinolinone scaffolds that serve as critical intermediates in numerous therapeutic agents including antibiotics, antitumor drugs, and receptor antagonists. The patented process addresses longstanding challenges in traditional quinolinone synthesis by eliminating multi-step procedures and expensive reagents while maintaining exceptional functional group tolerance across diverse substituent patterns. With reaction conditions operating at precisely controlled temperatures using readily available starting materials, this method demonstrates remarkable practicality for industrial-scale implementation without requiring specialized equipment or hazardous reagents. The strategic catalyst system comprising palladium acetate, chiral (S)-BINAP ligand, and molybdenum carbonyl creates a highly efficient transformation that achieves consistent yields across multiple substrate combinations while ensuring operational safety and environmental sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to quinolin-2(1H)one derivatives including Vilsmeier-Haack, Knorr, and Friedlander reactions suffer from multiple critical limitations that hinder their industrial applicability for pharmaceutical intermediate production. These conventional methods typically require harsh reaction conditions involving strong acids or high temperatures that promote undesired side reactions and decomposition pathways, resulting in inconsistent product quality that fails to meet stringent pharmaceutical purity standards. The narrow functional group tolerance severely restricts structural diversity achievable in quinolinone scaffolds, forcing medicinal chemists to implement time-consuming protection/deprotection strategies that significantly extend development timelines. Furthermore, transition metal-catalyzed approaches previously reported often necessitate expensive catalysts requiring complex removal procedures that add substantial cost burden without guaranteeing complete metal elimination to acceptable levels required by regulatory agencies. Ring-closing metathesis techniques face additional challenges with substrate scope limitations and stereoselectivity issues that compromise their utility for commercial-scale manufacturing where consistent product specifications are non-negotiable requirements.
The Novel Approach
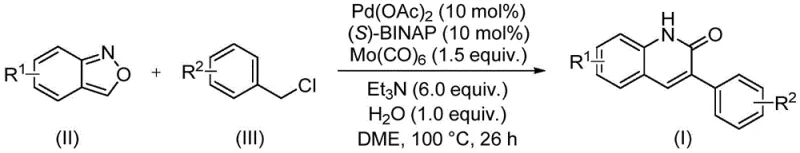
The patented methodology described in CN113045489B overcomes these limitations through an elegant palladium-catalyzed aminocarbonylation process that utilizes benzisoxazole as a dual-function reagent serving as both nitrogen source and formyl source within a single transformation step. This innovative approach operates under mild conditions using ethylene glycol dimethyl ether solvent at precisely controlled temperatures without requiring specialized equipment or hazardous reagents typically associated with conventional methodologies. The reaction demonstrates exceptional functional group tolerance across diverse substituent patterns including halogens, cyano groups, alkoxy moieties, and alkyl chains while maintaining high yields between 68% and 97% across fifteen experimentally validated substrate combinations documented in the patent examples. The streamlined process requires only simple filtration followed by standard column chromatography purification without complex workup procedures or specialized separation techniques needed in alternative approaches. This methodology represents a substantial improvement in both economic efficiency and environmental sustainability through reduced step count, minimized waste generation, and elimination of hazardous reagents while delivering consistent high-purity output suitable for direct incorporation into pharmaceutical manufacturing workflows.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The catalytic cycle begins with oxidative addition of the benzyl chloride substrate to palladium(0) species generated in situ from palladium acetate reduction by triethylamine under thermal conditions, forming an arylpalladium(II) intermediate that serves as the foundation for subsequent transformations. This key intermediate undergoes transmetalation with benzisoxazole where the heterocyclic ring system acts as a bifunctional reagent providing both nitrogen atom incorporation through ring opening and carbonyl functionality via molybdenum carbonyl-mediated CO insertion into the palladium-carbon bond. Water plays a critical role as proton shuttle facilitating aromatization steps essential for final product formation while maintaining optimal reaction kinetics throughout the transformation sequence. The chiral (S)-BINAP ligand stabilizes the active palladium species while preventing catalyst deactivation pathways common in alternative methodologies, ensuring consistent catalytic activity across diverse substrate combinations without requiring ligand optimization between different analog syntheses.
Impurity control is achieved through precise stoichiometric balance established in patent examples where benzisoxazole:benzyl chloride:palladium catalyst ratio is maintained at optimal levels (typically 1:2.5:0.1), preventing side reactions that could lead to dimerization or over-reaction products commonly observed in less controlled systems. The carefully optimized reaction temperature profile at exactly 100°C for precisely timed duration prevents thermal degradation pathways while ensuring complete conversion without requiring additional purification steps to remove unreacted starting materials or intermediates. Triethylamine base maintains optimal pH conditions throughout the reaction sequence minimizing decomposition pathways that could generate impurities difficult to remove during standard workup procedures. This comprehensive impurity management strategy ensures consistent production of high-purity intermediates meeting stringent regulatory requirements without requiring additional processing steps that would compromise overall process economics.
How to Synthesize 3-Arylquinolinone Derivatives Efficiently
This patented methodology offers a streamlined approach to synthesizing diverse 3-arylquinolinone derivatives with exceptional efficiency and scalability characteristics essential for commercial pharmaceutical manufacturing operations. The process begins with careful selection of appropriately substituted benzisoxazole precursors based on target molecule requirements followed by precise measurement according to established stoichiometric ratios documented in patent examples to ensure optimal reaction performance across different substrate combinations. Reaction setup utilizes standard laboratory equipment without specialized apparatus requirements making it readily implementable within existing manufacturing facilities without significant capital investment or process validation delays typically associated with new technology adoption.
- Precisely measure reactants according to optimized molar ratios: benzisoxazole (II), benzyl chloride compound (III), palladium acetate catalyst (0.05-0.1 mol%), (S)-BINAP ligand (0.05-0.1 mol%), triethylamine (6 equivalents), water (1 equivalent), and molybdenum carbonyl (1.5 equivalents)
- Combine all components in ethylene glycol dimethyl ether solvent at room temperature with thorough mixing to ensure homogeneous distribution before initiating the thermal reaction cycle
- Heat the reaction mixture to precisely 100°C under inert atmosphere and maintain constant temperature for exactly 26 hours before proceeding to standardized workup procedures
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic methodology delivers substantial value to procurement and supply chain operations by addressing critical pain points associated with traditional quinolinone intermediate production through fundamental process improvements that enhance overall operational resilience while reducing total cost of ownership. The elimination of multi-step sequences required in conventional approaches significantly reduces manufacturing complexity while improving overall process reliability through fewer unit operations where potential failures could occur during production runs.
- Cost Reduction in Manufacturing: The strategic elimination of expensive transition metal catalysts typically required in alternative methodologies substantially reduces raw material costs while avoiding costly metal removal processes that add multiple purification steps to traditional manufacturing workflows requiring specialized equipment investments and additional validation protocols.
- Enhanced Supply Chain Reliability: The broad availability of all required starting materials from multiple established chemical suppliers mitigates single-source dependency risks common in specialty chemical manufacturing while enabling flexible sourcing strategies that maintain consistent material availability regardless of regional supply chain disruptions or geopolitical factors affecting specific geographic regions.
- Scalability and Environmental Compliance: The reaction's compatibility with standard manufacturing equipment facilitates seamless scale-up from laboratory validation through pilot plant trials to full commercial production without requiring process re-engineering or specialized infrastructure investments that would otherwise create significant time-to-market delays during technology transfer phases.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations regarding implementation of this patented synthetic methodology for pharmaceutical intermediate production based on experimental data presented in patent CN113045489B providing authoritative guidance for manufacturers evaluating this innovative approach for their specific production requirements.
Q: How does benzisoxazole's dual functionality improve process efficiency compared to traditional quinolinone synthesis methods?
A: Benzisoxazole serves as both nitrogen source and formyl source in this patented methodology, eliminating separate reagent additions required in conventional Vilsmeier-Haack or Friedlander reactions. This dual functionality reduces step count from multi-step sequences to a single operation while avoiding expensive or hazardous reagents typically needed for nitrogen incorporation and carbonyl introduction.
Q: What specific advantages does this methodology offer for manufacturing high-purity pharmaceutical intermediates at commercial scale?
A: The process demonstrates exceptional functional group tolerance across diverse substituents without requiring protective groups, significantly reducing impurity formation pathways common in traditional syntheses. The simplified workup procedure involving filtration followed by standard column chromatography ensures consistent high-purity output meeting stringent pharmaceutical quality specifications across scale-up from laboratory to commercial production.
Q: How does the broad substrate scope impact development timelines for new quinolinone-based therapeutics?
A: The wide functional group compatibility enables direct synthesis of structurally diverse derivatives without route re-engineering between analogs. This flexibility accelerates medicinal chemistry campaigns by allowing rapid generation of compound libraries while maintaining consistent manufacturing protocols, thereby compressing lead optimization timelines without compromising product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolinone Derivatives Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic intermediates with stringent purity specifications consistently meeting global regulatory standards through our state-of-the-art QC labs equipped with advanced analytical instrumentation for comprehensive quality assurance testing protocols.
Leverage our technical expertise to optimize your quinolinone intermediate supply chain through a Customized Cost-Saving Analysis tailored to your specific production requirements by contacting our technical procurement team today to request detailed COA data and comprehensive route feasibility assessments that will help you make informed decisions about integrating this innovative methodology into your manufacturing operations.
