Advanced Quinolinone Derivative Manufacturing: Scalable Palladium-Catalyzed Process for Pharmaceutical Intermediates
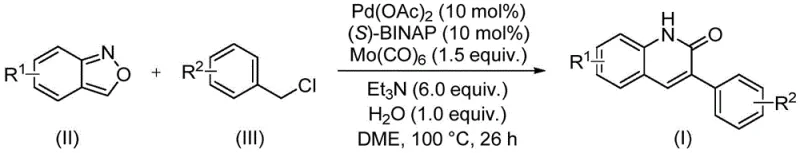
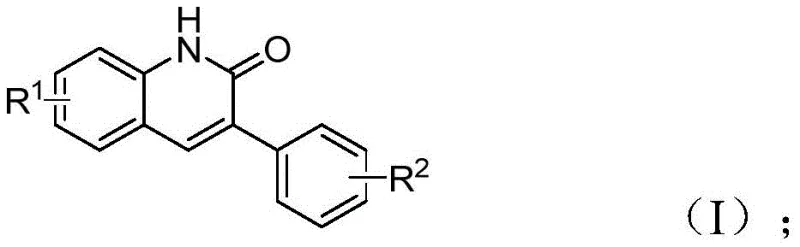
The patent CN113045489B introduces a groundbreaking methodology for synthesizing 3-arylquinoline-2(1H) ketone derivatives through a novel palladium-catalyzed aminocarbonylation process that fundamentally reimagines traditional synthetic approaches. This innovative technique leverages benzisoxazole as a dual-function reagent serving simultaneously as both nitrogen source and formyl source, thereby eliminating multiple synthetic steps required in conventional Vilsmeier Haack or Friedlander reactions. The process demonstrates exceptional operational simplicity while maintaining high substrate versatility across diverse functional groups, representing a significant advancement in the production of pharmacologically relevant heterocyclic compounds. By utilizing commercially available catalysts and reagents under mild reaction conditions, this method addresses critical limitations in existing synthetic routes while offering substantial advantages for industrial-scale implementation in pharmaceutical manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for quinolinone derivatives, including Vilsmeier Haack, Knorr, and Friedlander reactions, often require multiple protection/deprotection steps and harsh reaction conditions that compromise both yield and purity. These methods typically involve toxic reagents, generate significant waste streams, and exhibit limited functional group tolerance, particularly with sensitive substituents common in pharmaceutical intermediates. The multi-step nature of conventional syntheses creates substantial challenges for scale-up, with each additional step introducing potential points of failure and increasing the risk of impurity formation that requires extensive purification. Furthermore, traditional routes frequently depend on specialized equipment and stringent environmental controls that significantly elevate production costs while complicating supply chain logistics for global pharmaceutical manufacturers seeking reliable intermediate sources.
The Novel Approach
The patented methodology overcomes these limitations through an elegant one-pot transformation that utilizes benzisoxazole as a dual-function reagent, eliminating the need for separate nitrogen and carbonyl sources while dramatically simplifying the synthetic sequence. This approach operates under mild conditions (100°C in DME solvent) with readily available catalysts including palladium acetate (10 mol%) and (S)-BINAP ligand (10 mol%), achieving high yields across diverse substrates without requiring specialized equipment or hazardous reagents. The process demonstrates exceptional functional group tolerance, accommodating halogens, cyano groups, alkoxy substituents, and various alkyl functionalities while maintaining consistent product quality. By streamlining the synthetic pathway and utilizing cost-effective starting materials, this method establishes a new benchmark for efficient quinolinone derivative production that directly addresses the critical pain points of traditional manufacturing approaches.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The catalytic cycle begins with oxidative addition of benzyl chloride to the palladium(0) species generated in situ from palladium acetate and (S)-BINAP ligand, forming an arylpalladium intermediate that subsequently coordinates with benzisoxazole. This key intermediate undergoes ring-opening of the isoxazole moiety, releasing the nitrogen functionality while simultaneously providing the formyl group through a unique fragmentation pathway that avoids traditional CO gas handling requirements. The resulting palladium-bound species then undergoes migratory insertion followed by reductive elimination to yield the quinolinone core structure with high regioselectivity. The water additive plays a critical role in facilitating proton transfer steps while suppressing unwanted side reactions, contributing to the exceptional functional group tolerance observed across diverse substrate combinations.
Impurity control is achieved through precise optimization of the catalyst system and reaction parameters, where the combination of palladium acetate with (S)-BINAP ligand creates a highly selective catalytic environment that minimizes dimerization and other common side reactions. The use of molybdenum carbonyl as a CO surrogate ensures controlled release of carbon monoxide equivalents without requiring pressurized gas handling systems, significantly enhancing process safety while maintaining consistent product quality. The triethylamine base serves dual purposes as both proton scavenger and reaction accelerator, while the water additive promotes hydrolysis of intermediate species to prevent accumulation of potential impurities. This carefully balanced system delivers products with consistently high purity profiles suitable for pharmaceutical applications without requiring extensive post-reaction purification steps.
How to Synthesize Quinolinone Derivatives Efficiently
This patented methodology represents a significant advancement in quinolinone derivative synthesis through its innovative use of benzisoxazole as a dual-function reagent that simultaneously provides both nitrogen and formyl components within a single catalytic cycle. The process eliminates multiple synthetic steps required in traditional approaches while maintaining exceptional substrate versatility across diverse functional groups commonly encountered in pharmaceutical intermediates. By operating under mild reaction conditions with readily available catalysts and reagents, this method offers substantial operational advantages for manufacturing environments seeking to optimize both quality and efficiency. Detailed standardized synthesis procedures are provided below to ensure consistent implementation across various production scales.
- Prepare reaction mixture with precise molar ratios of palladium acetate, (S)-BINAP ligand, molybdenum carbonyl, triethylamine, and water in DME solvent
- Combine benzisoxazole compound as dual nitrogen/formyl source with benzyl chloride substrate under controlled temperature conditions
- Execute reaction at 100°C for optimized duration followed by standard purification procedures to obtain high-purity quinolinone derivatives
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic approach delivers substantial value to procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate sourcing through multiple strategic advantages. The methodology significantly reduces complexity in raw material sourcing by utilizing widely available starting materials that are not subject to supply chain bottlenecks common with specialized reagents required in traditional syntheses. By eliminating multiple synthetic steps and associated purification requirements, the process creates inherent cost advantages while enhancing production reliability through simplified operational workflows that minimize potential failure points during scale-up.
- Cost Reduction in Manufacturing: The elimination of specialized reagents and multi-step purification sequences creates substantial cost savings through reduced raw material expenses and lower processing requirements. By utilizing benzisoxazole as a dual-function reagent, the process avoids costly separate nitrogen and carbonyl sources while minimizing waste generation through its streamlined reaction pathway. The use of standard laboratory equipment instead of specialized high-pressure systems further reduces capital investment needs while maintaining consistent product quality across different production scales.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials with stable global supply chains ensures consistent access to necessary reagents regardless of regional market fluctuations or geopolitical factors that might impact specialized chemical suppliers. The process demonstrates exceptional robustness across different production environments while maintaining consistent yield profiles, enabling reliable forecasting and planning for procurement teams managing complex global supply networks. This stability translates directly into improved delivery predictability without requiring extensive qualification processes for new raw material sources.
- Scalability and Environmental Compliance: The methodology's compatibility with standard manufacturing equipment facilitates seamless scale-up from laboratory to commercial production without requiring significant process revalidation or capital investment in specialized infrastructure. The elimination of hazardous reagents and reduction in waste streams through its streamlined synthetic pathway significantly lowers environmental impact while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices. This inherent scalability combined with improved environmental profile creates substantial competitive advantages for manufacturers seeking to optimize both operational efficiency and regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations based on detailed analysis of the patented methodology's implementation parameters and performance characteristics. These insights have been developed through comprehensive evaluation of the reaction mechanism, substrate scope limitations, and scale-up considerations documented in the patent literature.
Q: How does this method improve upon traditional quinolinone synthesis techniques?
A: The patented process eliminates multiple synthetic steps by utilizing benzisoxazole as both nitrogen and formyl source, significantly reducing purification complexity while maintaining high functional group tolerance across diverse substrates.
Q: What are the key advantages for pharmaceutical manufacturing scalability?
A: The methodology demonstrates exceptional substrate versatility with consistent high yields across varied functional groups, enabling seamless scale-up from laboratory to commercial production without reoptimization of reaction parameters.
Q: How does this approach address supply chain reliability concerns?
A: By employing readily available starting materials and eliminating rare metal catalysts, the process ensures consistent raw material sourcing while maintaining stringent purity specifications required for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolinone Derivatives Supplier
Our patented methodology represents a significant advancement in quinolinone derivative synthesis that directly addresses the critical challenges facing modern pharmaceutical manufacturing operations. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs and rigorous analytical protocols. Our dedicated technical team specializes in optimizing complex synthetic routes like this palladium-catalyzed aminocarbonylation process to ensure consistent product quality and reliable supply chain performance for global pharmaceutical partners requiring high-purity intermediates.
Leverage our expertise to implement this innovative synthesis route through our Customized Cost-Saving Analysis service that evaluates your specific manufacturing requirements against our optimized processes. Contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your production needs, ensuring seamless integration of this advanced methodology into your existing supply chain infrastructure.
