Advanced Palladium-Catalyzed Carbonylation for Scalable 2-Trifluoromethyl Imidazole Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2-Trifluoromethyl Imidazole Production
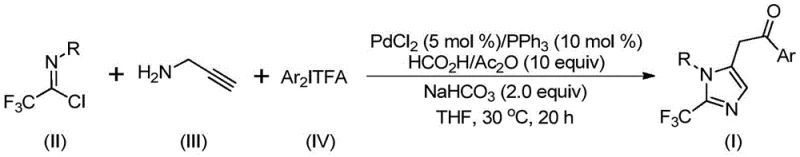
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone of modern medicinal chemistry, driven by the profound impact these motifs have on the metabolic stability, lipophilicity, and bioavailability of drug candidates. As detailed in the groundbreaking patent CN111423381B, a novel and highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds has been developed, addressing critical bottlenecks in the synthesis of these valuable nitrogen-containing five-membered heterocycles. This technology leverages a sophisticated transition metal palladium-catalyzed carbonylation series reaction, utilizing readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this methodology offers a robust pathway to access complex fluorinated building blocks that are essential for the development of next-generation therapeutics and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with significant challenges that hinder large-scale manufacturing and cost-effective production. Traditional approaches often rely on the direct reaction of synthons containing trifluoromethyl groups with suitable substrates, frequently utilizing hazardous reagents such as trifluorodiazoethane or specialized trifluoroethylimidoyl halides under stringent conditions. These conventional routes typically suffer from poor atom economy, the requirement for expensive and difficult-to-handle fluorinating agents, and limited substrate compatibility which restricts the structural diversity of the final products. Furthermore, many existing methods necessitate the use of high-pressure carbon monoxide gas, posing severe safety risks and requiring specialized infrastructure that increases capital expenditure for chemical manufacturing facilities. The reliance on such苛刻 conditions often leads to inconsistent yields and complicates the purification process, resulting in higher impurity profiles that are unacceptable for high-purity API intermediate production.
The Novel Approach
In stark contrast to these legacy methods, the innovative process disclosed in the patent introduces a streamlined, multicomponent reaction strategy that operates under remarkably mild conditions, specifically at a temperature of 30°C. This novel approach utilizes cheap and easily obtained trifluoroethyliminato chloride, propargylamine, and diaryl iodide as the foundational building blocks, effectively bypassing the need for dangerous gaseous CO or unstable diazo compounds. By employing a palladium-catalyzed carbonylation sequence where carbon monoxide is generated in situ from formic acid and acetic anhydride, the method achieves high reaction efficiency and excellent functional group tolerance. This breakthrough not only simplifies the operational complexity but also significantly broadens the utility of the method, allowing for the design and synthesis of diversified substituted imidazole compounds with trifluoromethyl groups at the 1 and 5 positions. For organizations focused on cost reduction in pharmaceutical intermediate manufacturing, this shift towards safer, ambient-temperature chemistry represents a paradigm shift in process development.
Mechanistic Insights into Pd-Catalyzed Multicomponent Carbonylation
The mechanistic elegance of this transformation lies in its intricate cascade of organometallic steps, which are meticulously orchestrated to construct the imidazole core while installing the trifluoromethyl moiety with precision. The reaction initiates with the formation of an intermolecular carbon-nitrogen bond promoted by the alkaline additive, yielding a trifluoroacetamidine compound which subsequently undergoes isomerization. Following this, the palladium catalyst activates the alkyne functionality of the propargylamine derivative through palladation, generating a crucial alkenyl palladium intermediate. This species then isomerizes to form an alkyl palladium intermediate, setting the stage for the pivotal carbonylation step. Under the influence of the carbon monoxide released from the formic acid-acetic anhydride mixture, an acyl palladium intermediate is formed, which serves as the electrophilic center for the subsequent coupling event.
The catalytic cycle culminates with the oxidative addition of the diaryl iodonium salt to the palladium center, generating a high-valent tetravalent palladium intermediate. This transient species then undergoes reductive elimination to release the final 2-trifluoromethyl-substituted imidazole product and regenerate the active palladium catalyst. This detailed mechanistic understanding is vital for R&D teams aiming to optimize the commercial scale-up of complex pharmaceutical intermediates, as it highlights the critical role of each reagent and the delicate balance required to maintain catalytic turnover. The use of sodium bicarbonate as a base and the specific ratio of palladium chloride to triphenylphosphine (0.05:0.1) are optimized to minimize side reactions and ensure high purity specifications, thereby reducing the burden on downstream purification processes and enhancing the overall sustainability of the manufacturing workflow.
How to Synthesize 2-Trifluoromethyl Imidazoles Efficiently
The practical implementation of this synthesis protocol is designed for ease of execution, making it highly attractive for both laboratory-scale discovery and pilot-plant operations. The procedure involves a straightforward one-pot reaction where all components, including the palladium catalyst, ligand, CO surrogate, additives, and substrates, are combined in an organic solvent such as tetrahydrofuran. The reaction is allowed to proceed at a mild 30°C for a duration of 16 to 24 hours, ensuring complete conversion of the starting materials without the need for energy-intensive heating or cooling cycles. Upon completion, the workup is equally simple, involving filtration and standard column chromatography purification to isolate the target compound in high yield. For a detailed breakdown of the standardized operating procedures and safety protocols, please refer to the technical guide below.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid, trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt in an organic solvent such as THF.
- Stir the reaction mixture at a mild temperature of 30°C for a duration of 16 to 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, this patented methodology offers compelling advantages that directly address the pain points of volatility and cost in the fine chemicals sector. The reliance on commercially available and inexpensive starting materials, such as aromatic amines and propargylamine, ensures a stable and resilient supply chain that is less susceptible to the geopolitical and logistical disruptions often associated with exotic fluorinating reagents. By eliminating the need for high-pressure gas infrastructure and specialized containment systems for toxic reagents, the process drastically reduces the barrier to entry for contract manufacturing organizations and internal production facilities alike. This accessibility translates into a more competitive pricing structure for the final intermediates, enabling pharmaceutical companies to optimize their bill of materials without compromising on quality or regulatory compliance standards.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of low-cost catalysts like palladium chloride and the avoidance of expensive, proprietary trifluoromethylating agents. The in-situ generation of carbon monoxide from formic acid and acetic anhydride removes the logistical costs and safety premiums associated with purchasing and storing compressed CO gas cylinders. Furthermore, the mild reaction temperature of 30°C significantly lowers energy consumption compared to traditional high-temperature reflux conditions, contributing to substantial operational expenditure savings over the lifecycle of the product. The high reaction efficiency and substrate compatibility mean that fewer batches are rejected due to poor conversion, maximizing the return on investment for raw material procurement.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions and the widespread availability of the key precursors, such as diaryl iodonium salts and trifluoroethylimidoyl chlorides, mitigate the risk of supply shortages. Unlike methods dependent on single-source specialty chemicals, this approach allows for multi-vendor sourcing strategies for the bulk ingredients, thereby strengthening supply chain continuity. The scalability of the process, which has been demonstrated to be expandable to the gram level with potential for industrial tonnage production, ensures that procurement managers can secure long-term contracts with confidence. This reliability is critical for maintaining uninterrupted production schedules for downstream API manufacturing and meeting tight project milestones.
- Scalability and Environmental Compliance: The simplified post-treatment procedure, which involves basic filtration and chromatography, reduces the generation of complex waste streams that require costly disposal. The use of common organic solvents like THF, which can be readily recovered and recycled, aligns with green chemistry principles and helps facilities meet increasingly stringent environmental regulations. The absence of heavy metal contaminants from exotic catalysts simplifies the purification process, reducing the need for extensive scavenging steps that can lower overall yield. This environmental friendliness not only reduces compliance costs but also enhances the corporate social responsibility profile of the supply chain, a factor of growing importance to global pharmaceutical partners.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction parameters and scope. These insights are derived directly from the experimental data and optimization studies presented in the patent literature, providing a transparent view of the method's capabilities and limitations. Understanding these nuances is essential for effective technology transfer and process validation.
Q: What is the carbon monoxide source in this synthesis?
A: The process utilizes an in-situ generated carbon monoxide source derived from the decomposition of formic acid and acetic anhydride, eliminating the need for hazardous high-pressure CO gas cylinders.
Q: What are the optimal reaction conditions for this transformation?
A: The reaction proceeds efficiently at a mild temperature of 30°C over a period of 16 to 24 hours, using tetrahydrofuran (THF) as the preferred organic solvent.
Q: Which catalyst system is employed for this carbonylation?
A: The method employs a cost-effective palladium catalyst system consisting of palladium chloride (PdCl2) and triphenylphosphine (PPh3) ligands.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology in accelerating the development of fluorinated drug candidates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications for complex heterocyclic intermediates, guaranteeing that every batch meets the exacting standards required by global regulatory bodies. We are committed to leveraging our technical expertise to optimize this specific route for your unique molecular targets, delivering high-quality intermediates that drive your pipeline forward.
We invite you to engage with our technical team to explore how this innovative synthesis method can be integrated into your supply chain to achieve superior economic and operational outcomes. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential financial benefits tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project is built on a foundation of scientific excellence and commercial reliability.
