Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Manufacturing
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which enhance metabolic stability and bioavailability. Patent CN111423381A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical synthetic challenges in modern drug discovery. This technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts. Unlike traditional approaches that often rely on hazardous reagents or harsh conditions, this novel process operates efficiently at a mild temperature of 30°C. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant leap forward in accessing high-purity OLED material precursors and complex API scaffolds with improved safety profiles and cost-efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl groups has been fraught with significant safety and operational hurdles. Conventional literature methods predominantly rely on trifluoromethyl synthons such as trifluorodiazoethane, which poses severe explosion risks and requires specialized handling equipment to mitigate hazards. Furthermore, alternative routes using trifluoroethylimide acid halides have seen limited application due to perceived difficulties in reactivity control and narrow substrate scope. These traditional pathways often necessitate extreme temperatures or stoichiometric amounts of toxic reagents, leading to complex impurity profiles that complicate downstream purification. For supply chain heads, these factors translate into increased lead times for high-purity pharmaceutical intermediates and elevated costs associated with waste disposal and safety compliance. The inability to easily diversify the molecular scaffold limits the speed at which medicinal chemists can iterate on lead compounds, creating a bottleneck in the drug development pipeline.
The Novel Approach
The methodology disclosed in CN111423381A fundamentally reshapes the synthetic landscape by employing a palladium-catalyzed carbonylation tandem reaction that is both safe and highly efficient. By utilizing trifluoroethylimidoyl chloride and propargylamine as key building blocks, the process avoids the generation of unstable diazo intermediates entirely. The reaction proceeds smoothly in common organic solvents like tetrahydrofuran (THF) at a constant 30°C, demonstrating exceptional functional group tolerance across a wide array of aryl substituents. 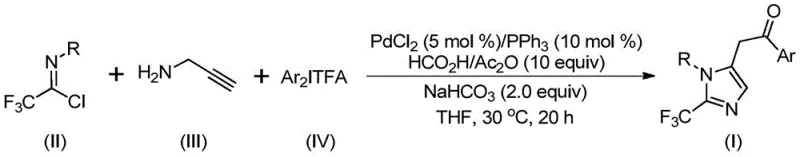 As illustrated in the reaction scheme, the convergence of three distinct components allows for the rapid assembly of complex 1,5-disubstituted imidazole cores. This modularity enables the synthesis of diversified libraries, such as compounds I-1 through I-5 shown in the patent examples, facilitating cost reduction in electronic chemical manufacturing and pharmaceutical applications by streamlining the route to valuable fluorinated heterocycles.
As illustrated in the reaction scheme, the convergence of three distinct components allows for the rapid assembly of complex 1,5-disubstituted imidazole cores. This modularity enables the synthesis of diversified libraries, such as compounds I-1 through I-5 shown in the patent examples, facilitating cost reduction in electronic chemical manufacturing and pharmaceutical applications by streamlining the route to valuable fluorinated heterocycles.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific targets. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, generating a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization, setting the stage for the pivotal palladium catalytic cycle. The palladium catalyst, typically generated in situ from PdCl2 and PPh3, facilitates the aminopalladation of the alkyne moiety to form a vinyl-palladium intermediate. Through a series of precise isomerizations, this evolves into an alkyl-palladium species which then undergoes carbonylation. The carbon monoxide required for this step is conveniently released in situ from the formic acid and acetic anhydride mixture, eliminating the need for high-pressure CO gas cylinders. Finally, oxidative addition of the diaryliodonium salt generates a tetravalent palladium intermediate, which collapses via reductive elimination to release the final 2-trifluoromethyl substituted imidazole product and regenerate the active catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based trifluoromethylation strategies. The stepwise nature of the palladium cycle ensures high regioselectivity, minimizing the formation of positional isomers that are notoriously difficult to separate. The mild reaction temperature of 30°C further suppresses thermal decomposition pathways and side reactions such as polymerization of the alkyne starting material. Moreover, the use of diaryliodonium salts as arylating agents provides a clean leaving group profile, reducing the burden on downstream purification processes like column chromatography. For manufacturers focused on stringent purity specifications, this mechanistic clarity allows for better prediction and control of the impurity profile, ensuring that the resulting high-purity pharmaceutical intermediates meet rigorous regulatory standards for clinical use.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale discovery and pilot-scale production. The protocol involves a straightforward one-pot procedure where all reagents, including the palladium catalyst, ligand, base, and carbonyl source, are combined in an organic solvent. The reaction mixture is stirred at 30°C for a duration of 16 to 24 hours, ensuring complete conversion of the starting materials. Post-reaction processing is equally simplified, involving basic filtration to remove inorganic salts followed by silica gel treatment and standard column chromatography. This streamlined workflow eliminates the need for cryogenic conditions or inert atmosphere gloveboxes typically associated with sensitive organometallic chemistry, thereby lowering the barrier to entry for adopting this technology in diverse manufacturing settings.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, followed by filtration and column chromatography purification to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers tangible strategic benefits beyond mere chemical novelty. The shift towards safer, more stable starting materials directly correlates with reduced operational risks and lower insurance premiums for manufacturing facilities. By eliminating the need for hazardous diazo compounds, companies can avoid the substantial costs associated with specialized storage and handling infrastructure. Furthermore, the high atom economy and efficient catalyst turnover contribute to substantial cost savings in raw material consumption. The ability to source cheap and readily available aromatic amines for the preparation of trifluoroethylimidoyl chloride ensures a stable supply chain, mitigating the risk of raw material shortages that often plague the production of exotic fluorinated building blocks.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive palladium chloride as the catalyst precursor compared to more exotic noble metal complexes. The reaction conditions are energy-efficient, operating at near-ambient temperatures which drastically reduces heating and cooling loads in large-scale reactors. Additionally, the high yields reported, reaching up to 97% for certain substrates, minimize waste generation and maximize the output per batch. This efficiency translates directly into a lower cost of goods sold (COGS) for the final API intermediate, providing a competitive edge in pricing negotiations with downstream pharmaceutical clients.
- Enhanced Supply Chain Reliability: The robustness of the reaction against various functional groups means that a single standardized protocol can be applied to synthesize a wide library of derivatives. This flexibility reduces the need for multiple specialized production lines, simplifying inventory management and scheduling. The starting materials, including propargylamine and various diaryliodonium salts, are commercially available from multiple global suppliers, ensuring redundancy in the supply chain. This diversification protects against single-source bottlenecks and ensures consistent delivery schedules for critical project milestones.
- Scalability and Environmental Compliance: The patent explicitly notes the potential for scale-up to the gram level and beyond, indicating that the chemistry is not limited to milligram-scale discovery. The use of THF as a solvent, while requiring recovery systems, is well-established in industrial settings with mature recycling protocols. The avoidance of toxic carbon monoxide gas cylinders in favor of in situ generation from formic acid significantly improves the environmental, health, and safety (EHS) profile of the process. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction parameters and scope. These insights are derived directly from the experimental data and descriptions provided in the patent documentation, ensuring accuracy and relevance for process development discussions. Understanding these nuances is essential for integrating this synthetic route into existing manufacturing workflows and maximizing its potential for producing high-value fluorinated heterocycles.
Q: What are the key advantages of this palladium-catalyzed method over traditional diazo-based routes?
A: This method avoids the use of hazardous trifluorodiazoethane, utilizing safer trifluoroethylimidoyl chlorides instead. It operates under mild conditions (30°C) with excellent functional group tolerance, significantly improving operational safety and substrate compatibility for diverse API intermediates.
Q: Can this synthesis protocol be scaled for industrial production?
A: Yes, the patent explicitly demonstrates scalability to the gram level with simple post-processing steps like filtration and column chromatography. The use of commercially available catalysts and mild temperatures facilitates potential scale-up for commercial manufacturing.
Q: What is the typical yield range for these 2-trifluoromethyl imidazole derivatives?
A: Experimental data indicates high reaction efficiency, with isolated yields ranging significantly depending on the substrate. Specific examples in the patent demonstrate yields reaching up to 97% for optimized substrates, confirming the robustness of the catalytic system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN111423381A for accelerating drug discovery pipelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications for complex fluorinated intermediates. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global market, leveraging our deep technical expertise to optimize yield and quality.
We invite you to collaborate with us to explore the full capabilities of this palladium-catalyzed synthesis for your specific API candidates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. Please contact us today to request specific COA data for related imidazole derivatives and comprehensive route feasibility assessments. Let us help you secure a reliable supply chain for your next-generation therapeutics while optimizing your manufacturing costs.
