Advanced One-Pot Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
Advanced One-Pot Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. A significant breakthrough in this domain is detailed in patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. This technology represents a paradigm shift from traditional step-wise syntheses to a streamlined, one-pot palladium-catalyzed cascade reaction. By integrating trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, this process constructs indolinone-imidazole hybrid structures with remarkable precision. For R&D directors and procurement managers, the implications are profound: a route that eliminates the need for hazardous carbon monoxide gas while maintaining high reaction efficiency and substrate compatibility. This report analyzes the technical merits and commercial viability of this innovation, positioning it as a cornerstone for reliable pharmaceutical intermediate supplier strategies aiming for cost-effective and safe manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems containing both indolinone and imidazole moieties has been fraught with synthetic challenges. Conventional literature typically describes three main approaches: direct coupling of two heterocyclic substrates, oxidative cyclization of substrates bearing dual nucleophiles, or transition metal-catalyzed tandem reactions that often lack generality. The direct coupling methods frequently suffer from poor atom economy and require harsh conditions that can degrade sensitive functional groups. Furthermore, oxidative cyclization routes often necessitate stoichiometric amounts of oxidants, generating substantial waste and complicating downstream purification. Perhaps most critically, traditional carbonylation strategies to install the bridging carbonyl group rely heavily on the use of toxic carbon monoxide gas. This not only poses severe safety risks requiring specialized high-pressure equipment but also introduces significant regulatory and logistical hurdles for commercial scale-up of complex pharmaceutical intermediates. These limitations collectively drive up the cost of goods and extend lead times for high-purity intermediates.
The Novel Approach
In stark contrast, the methodology described in patent CN115353511A offers a transformative solution by employing a palladium-catalyzed carbonylation cascade that operates under exceptionally mild conditions. The core innovation lies in the use of a formic acid and acetic anhydride mixture as a safe, in situ source of carbon monoxide, effectively bypassing the need for external CO gas cylinders. This allows the reaction to proceed at a温和 temperature of 30°C in common organic solvents like tetrahydrofuran (THF). The process seamlessly integrates three distinct building blocks—trifluoroethylimidoyl chloride, propargylamine, and an acrylamide derivative—into a single reaction vessel. This one-pot strategy not only simplifies the operational workflow by eliminating intermediate isolation steps but also significantly enhances the overall yield by minimizing material loss during transfer. The ability to tolerate a wide range of functional groups, including halogens and electron-withdrawing nitro groups, underscores the versatility of this approach for generating diverse chemical libraries.
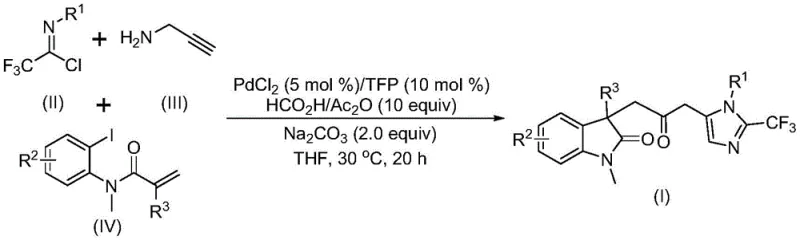
The general reaction scheme illustrates the convergence of the three components into the final carbonyl-bridged structure. As shown in the visual representation, the transformation involves the formation of multiple chemical bonds in a single operation, driven by the palladium catalyst system. This level of molecular complexity achieved in one step is rare and highly valuable for process chemistry teams looking to reduce the number of unit operations. The use of commercially available and inexpensive starting materials further bolsters the economic case for adopting this technology. By replacing dangerous reagents with benign alternatives and reducing the step count, this novel approach directly addresses the pain points of both safety compliance and manufacturing efficiency.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this reaction is crucial for R&D directors aiming to optimize the process for specific analogs. The proposed mechanism begins with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This is followed by an intramolecular Heck-type reaction, which generates a divalent alkyl-palladium intermediate. Crucially, the carbon monoxide required for the subsequent carbonylation step is released from the decomposition of the formic acid and acetic anhydride mixture. This CO then inserts into the palladium-carbon bond to form an acyl-palladium intermediate. Simultaneously, a base-promoted intermolecular reaction between the trifluoroethylimidoyl chloride and propargylamine occurs, forming a trifluoroacetamidine species which subsequently undergoes isomerization. The final ring-closing step is catalyzed by the activated acyl-palladium intermediate, which facilitates the intramolecular cyclization to yield the target carbonyl-bridged biheterocyclic compound. This intricate dance of catalytic cycles ensures high selectivity and minimizes the formation of byproducts.
From an impurity control perspective, the mild reaction temperature of 30°C plays a pivotal role. High-temperature processes often promote non-selective radical pathways or thermal degradation of sensitive intermediates, leading to complex impurity profiles that are difficult to purge. By maintaining the reaction at near-ambient temperatures, this method suppresses these side reactions, resulting in a cleaner crude reaction mixture. The use of trifurylphosphine (TFP) as a ligand further stabilizes the palladium center, preventing the formation of palladium black and ensuring sustained catalytic activity throughout the 12 to 20-hour reaction window. This stability is essential for maintaining consistent quality across different batches, a key requirement for any reliable agrochemical intermediate supplier or pharma partner. The specific interaction between the trifluoromethyl group and the developing heterocyclic ring also contributes to the thermodynamic stability of the final product, driving the equilibrium towards completion.
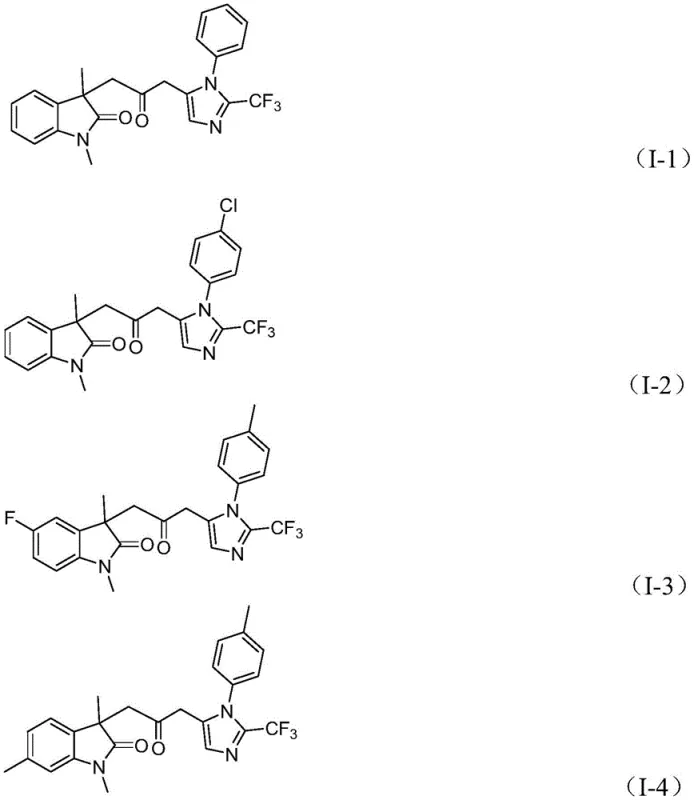
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Implementing this synthesis route requires careful attention to reagent quality and mixing protocols to ensure reproducibility. The patent outlines a straightforward procedure where the catalyst system is prepared first, followed by the sequential or simultaneous addition of the substrates. The choice of solvent is critical; while several aprotic solvents were screened, tetrahydrofuran (THF) was identified as the optimal medium due to its ability to dissolve all reactants effectively while promoting high conversion rates. The molar ratios are also tuned for efficiency, typically employing a slight excess of propargylamine and acrylamide relative to the trifluoroethylimidoyl chloride to drive the reaction to completion. Post-reaction workup is equally simple, involving filtration to remove inorganic salts and silica gel treatment, followed by standard column chromatography. For detailed standardized synthesis steps, please refer to the guide below.
- Combine palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and a formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the acrylamide derivative to the reaction vessel under stirring.
- Maintain the reaction mixture at 30°C for 12 to 20 hours, followed by filtration and column chromatography purification to isolate the target biheterocyclic compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic advantages beyond mere chemical elegance. The primary driver for cost reduction in pharmaceutical intermediate manufacturing here is the elimination of specialized high-pressure infrastructure. Traditional carbonylations require autoclaves capable of withstanding high pressures of toxic CO, which represent a significant capital expenditure (CapEx) and maintenance burden. By generating CO in situ from liquids, this process can be run in standard glass-lined reactors, drastically lowering the barrier to entry for production. Furthermore, the raw materials—propargylamine, acrylamides, and imidoyl chlorides—are commodity chemicals available from multiple global suppliers, mitigating the risk of supply chain disruptions. This abundance ensures that scaling up from gram-scale to multi-ton production does not encounter raw material bottlenecks.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the simplification of the reaction setup and the reduction in hazard management costs. By avoiding the use of gaseous carbon monoxide, facilities save significantly on safety monitoring systems, gas detection equipment, and specialized ventilation requirements. Additionally, the one-pot nature of the reaction reduces solvent consumption and labor hours associated with intermediate isolations and purifications. The high atom economy of the multicomponent reaction means that a larger proportion of the input mass ends up in the final product, reducing waste disposal costs. These factors combine to create a leaner manufacturing process that is inherently more cost-effective than legacy multi-step routes.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and flexibility of the synthetic route. The tolerance for various substituents on the aromatic rings means that the same core process can be used to manufacture a wide family of analogs without needing to revalidate entirely new chemistries. This flexibility allows manufacturers to respond quickly to changing market demands or clinical trial requirements. Moreover, the use of stable, shelf-stable reagents like palladium chloride and trifurylphosphine ensures that catalyst preparation is not a critical path item subject to degradation. The ability to source starting materials from diverse geographic regions further de-risks the supply chain, ensuring continuous availability of high-purity intermediates even during regional logistical challenges.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden pitfalls, but this methodology has already demonstrated feasibility at the gram scale with promising results for expansion. The mild reaction conditions reduce the energy footprint of the process, as there is no need for extensive heating or cooling loops to manage exotherms typical of high-pressure gas reactions. From an environmental standpoint, the avoidance of toxic gas emissions aligns with increasingly stringent global environmental regulations. The waste stream is primarily composed of organic solvents and inorganic salts, which are easier to treat and dispose of compared to heavy metal-contaminated wastes or toxic gas scrubber effluents. This environmental profile facilitates smoother regulatory approvals and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this technology into their pipelines. Understanding these nuances is vital for making informed decisions about process adoption and resource allocation.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: Unlike conventional carbonylation reactions that require toxic and high-pressure carbon monoxide gas, this method utilizes a formic acid and acetic anhydride mixture to generate CO in situ, significantly enhancing operational safety and reducing equipment costs.
Q: What is the substrate compatibility of this Pd-catalyzed cascade reaction?
A: The process demonstrates excellent functional group tolerance, accommodating various substituents on the aryl rings including alkyl, alkoxy, halogen, trifluoromethyl, and nitro groups, allowing for the synthesis of diverse derivatives.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly mentions that the method has been expanded to gram-scale reactions with high efficiency and simple post-treatment, indicating strong potential for commercial scale-up in pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in patent CN115353511A for the production of advanced pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facility is equipped with the necessary infrastructure to handle palladium-catalyzed reactions safely and efficiently, adhering to stringent purity specifications required by top-tier pharmaceutical clients. Our rigorous QC labs employ state-of-the-art analytical techniques to verify the identity and purity of every batch, guaranteeing that the carbonyl-bridged biheterocyclic compounds you receive meet the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to contact our technical procurement team to request specific COA data for related analogs and to discuss route feasibility assessments for your unique molecular targets. By partnering with us, you gain access to a supply chain that prioritizes safety, efficiency, and reliability, empowering you to bring life-saving medicines to market faster and more economically.
