Advanced Palladium-Catalyzed Multi-Component Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates
Advanced Palladium-Catalyzed Multi-Component Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates
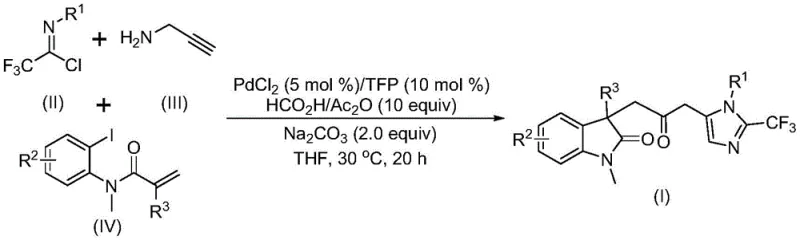
The pharmaceutical industry continuously seeks efficient pathways to construct complex heterocyclic scaffolds that serve as the core backbone for next-generation therapeutics. A significant breakthrough in this domain is documented in Chinese Patent CN115353511A, which discloses a sophisticated multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. This technology addresses the critical need for streamlined access to indolinone-imidazole fused systems, which are prevalent in molecules exhibiting broad-spectrum biological activities. By leveraging a transition metal palladium-catalyzed carbonylation cascade, this methodology eliminates the reliance on hazardous gaseous carbon monoxide while maintaining high reaction efficiency and exceptional substrate tolerance. For research and development teams focusing on oncology or metabolic disease targets, this protocol offers a reliable route to diversify chemical libraries with metabolically stable trifluoromethyl-containing motifs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic frameworks has been fraught with synthetic challenges that hinder rapid drug discovery cycles. Traditional approaches often rely on the direct coupling of two pre-formed heterocyclic substrates, a strategy that frequently suffers from low atom economy and requires harsh reaction conditions that limit functional group compatibility. Alternatively, oxidative cyclization reactions involving bis-nucleophilic substrates and activated methyl-substituted heterocycles often necessitate strong oxidants and elevated temperatures, leading to significant impurity profiles and difficult downstream purification. Furthermore, conventional carbonylation strategies to install the crucial bridging carbonyl moiety typically mandate the use of toxic carbon monoxide gas under high pressure, posing severe safety risks and requiring specialized infrastructure that many contract research organizations lack. These limitations collectively result in prolonged lead times for high-purity pharmaceutical intermediates and inflated manufacturing costs due to complex safety protocols and low yields.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN115353511A introduces a transformative one-pot multi-component reaction that seamlessly integrates bond formation and ring closure. This novel approach utilizes cheap and readily available starting materials, specifically trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, to construct the complex architecture in a single operational step. The brilliance of this method lies in its use of a formic acid and acetic anhydride mixture as a safe, liquid carbon monoxide surrogate, thereby obviating the need for dangerous gas cylinders. The reaction proceeds under exceptionally mild conditions at 30°C, demonstrating remarkable chemoselectivity that preserves sensitive functional groups such as halogens and nitro groups. This operational simplicity not only accelerates the synthesis timeline but also drastically reduces the barrier to entry for producing these valuable scaffolds on a commercial scale.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
For the discerning R&D director, understanding the mechanistic underpinnings of this transformation is vital for troubleshooting and further optimization. The reaction is hypothesized to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This is immediately followed by an intramolecular Heck-type reaction that constructs the indolinone core, yielding a divalent alkyl-palladium species. Subsequently, the carbon monoxide generated in situ from the formic acid/acetic anhydride mixture inserts into the palladium-carbon bond, forming an acyl-palladium intermediate. Concurrently, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine species, which undergoes isomerization. The final step involves the activation of this amidine by the acyl-palladium complex, triggering an intramolecular cyclization that forges the imidazole ring and releases the final carbonyl-bridged biheterocyclic product while regenerating the active catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses. By telescoping multiple bond-forming events into a single pot, the exposure of reactive intermediates to the bulk environment is minimized, thereby reducing the formation of hydrolysis byproducts or oligomerization impurities common in open-vessel procedures. The use of tris(2-furyl)phosphine (TFP) as a ligand is particularly strategic; its electron-rich nature stabilizes the palladium center against aggregation into inactive palladium black, ensuring consistent catalytic turnover throughout the 12 to 20-hour reaction window. Furthermore, the mild basicity of sodium carbonate ensures that acid-sensitive groups on the aromatic rings remain intact, resulting in a cleaner crude reaction profile that simplifies the subsequent column chromatography purification steps required to meet stringent purity specifications.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Implementing this synthesis requires precise attention to reagent stoichiometry and solvent quality to maximize yield and reproducibility. The protocol dictates a specific molar ratio where trifluoroethylimidoyl chloride serves as the limiting reagent, while propargylamine and the acrylamide derivative are used in slight excess to drive the equilibrium forward. The choice of tetrahydrofuran (THF) as the solvent is critical, as it provides the optimal solubility for all three diverse substrates and facilitates the homogeneous catalytic cycle. While the reaction is robust, maintaining the temperature strictly at 30°C is recommended to balance reaction rate with selectivity, preventing potential thermal degradation of the sensitive imidazole moiety. For detailed operational parameters and specific workup procedures, please refer to the standardized guide below.
- Prepare the catalytic system by mixing palladium chloride (PdCl2), tris(2-furyl)phosphine (TFP) ligand, sodium carbonate base, and a carbon monoxide source mixture of formic acid and acetic anhydride in an organic solvent like THF.
- Introduce the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative into the reaction vessel under stirring.
- Maintain the reaction mixture at a mild temperature of 30°C for 12 to 20 hours to allow the cascade cyclization and carbonylation to proceed to completion.
- Upon completion, perform standard post-treatment including filtration, silica gel mixing, and column chromatography purification to isolate the high-purity biheterocyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits regarding cost structure and supply continuity. The primary economic driver is the substitution of expensive or hazardous reagents with commodity chemicals; propargylamine and acrylamide derivatives are produced globally at massive scales, ensuring a stable supply chain immune to the bottlenecks often seen with exotic heterocyclic building blocks. Moreover, the elimination of high-pressure carbon monoxide equipment removes a significant capital expenditure requirement for manufacturing facilities, allowing production to occur in standard glass-lined reactors without specialized gas handling infrastructure. This reduction in operational complexity directly correlates to lower overhead costs and faster batch turnover times, enabling suppliers to respond more agilely to fluctuating market demands for clinical trial materials.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of palladium chloride, which, while a precious metal, is used in catalytic quantities (5 mol%) and can potentially be recovered or scavenged efficiently due to the homogeneous nature of the reaction. More importantly, the avoidance of cryogenic conditions or high-pressure autoclaves drastically reduces energy consumption per kilogram of product. The high atom economy of the multi-component reaction means that a larger proportion of the raw material mass ends up in the final product, minimizing waste disposal costs associated with solvent-heavy stepwise syntheses. Consequently, this leads to substantial cost savings in API manufacturing without compromising the quality of the intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the commercial availability of the key starting materials. Trifluoroethylimidoyl chloride can be rapidly synthesized from widely available aromatic amines and trifluoroacetic acid, creating a short and robust upstream supply chain. The tolerance of the reaction to various substituents, including halogens and alkyl groups, allows manufacturers to utilize a broad range of commercially sourced acrylamides without needing custom synthesis for each variant. This flexibility ensures that production schedules are not derailed by the unavailability of a single niche reagent, thereby reducing lead time for high-purity pharmaceutical intermediates and securing continuity of supply for downstream drug substance production.
- Scalability and Environmental Compliance: The patent data confirms successful expansion to gram-scale reactions, providing a clear pathway for commercial scale-up of complex pharmaceutical intermediates. The mild reaction conditions (30°C) inherently reduce the risk of thermal runaway incidents, a critical factor for safety compliance in large-scale manufacturing. Additionally, the use of formic acid as a CO source generates water and carbon dioxide as benign byproducts, aligning with green chemistry principles and simplifying environmental permitting processes. The simplified workup involving filtration and standard chromatography reduces the volume of hazardous waste generated, supporting corporate sustainability goals and reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy for process chemists evaluating this route. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing production workflows or research pipelines.
Q: What is the carbon monoxide source in this synthesis?
A: Instead of using toxic high-pressure carbon monoxide gas, this protocol utilizes an in situ generation system comprising a mixture of formic acid and acetic anhydride, which safely releases CO under the reaction conditions.
Q: What are the optimal reaction conditions described in the patent?
A: The reaction operates under remarkably mild conditions, specifically at 30°C in tetrahydrofuran (THF) solvent, typically requiring 12 to 20 hours for completion with a palladium chloride catalyst and TFP ligand.
Q: Is this process suitable for large-scale production?
A: Yes, the patent explicitly demonstrates the feasibility of scaling the reaction to the gram level, indicating robust substrate compatibility and operational simplicity suitable for industrial expansion.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced synthetic methodologies play in accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying the structural integrity and purity of these complex biheterocyclic scaffolds against stringent purity specifications. We are committed to leveraging innovations like the Pd-catalyzed carbonylation cascade to deliver high-quality intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with our technical team to evaluate how this cost-effective synthesis route can be tailored to your specific project needs. By engaging with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this multi-component protocol. Please contact our technical procurement team today to request specific COA data for related analogues and comprehensive route feasibility assessments. Let us help you optimize your supply chain and bring your life-saving therapies to market faster and more efficiently.
