Advanced Electrochemical Synthesis of 1,1'-Binaphthyl Compounds for Commercial Scale
The chemical industry is currently witnessing a paradigm shift towards greener, more sustainable synthesis methodologies, particularly in the production of high-value chiral intermediates. Patent CN113957461A introduces a groundbreaking electrochemical synthesis method for 1,1'-binaphthyl compounds, which are critical precursors for bioactive molecules, functional materials, and chiral ligands. This innovation addresses the longstanding challenges associated with traditional coupling reactions by eliminating the need for toxic transition metals and stoichiometric oxidants. The technology leverages constant current electrolysis in a diaphragm-free cell, operating under mild room temperature conditions to achieve considerable yields. For R&D directors and procurement managers, this represents a significant opportunity to streamline supply chains and reduce environmental compliance costs. The method's simplicity and safety profile make it an ideal candidate for commercial scale-up, ensuring a reliable supply of high-purity pharmaceutical intermediates for global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of the 1,1'-binaphthyl skeleton has relied heavily on reduction coupling reactions such as Kumada, Negishi, or Suzuki couplings, which necessitate the use of expensive transition metal catalysts. These conventional pathways often require pre-functionalized halogenated naphthalene substrates, adding multiple synthetic steps and increasing the overall cost of goods. Furthermore, oxidative coupling methods typically employ stoichiometric amounts of strong chemical oxidants, generating substantial quantities of hazardous waste and byproducts that complicate downstream purification. The reliance on heavy metals introduces stringent regulatory hurdles regarding residual metal limits in final API intermediates, posing risks to supply chain continuity. Additionally, many existing electrochemical methods are limited to substrates with strong electron-donating groups, restricting their utility for diverse chemical libraries. These factors collectively contribute to higher manufacturing costs and reduced atomic efficiency, creating a pressing need for more sustainable alternatives.
The Novel Approach
The electrochemical method disclosed in patent CN113957461A offers a transformative solution by utilizing direct anodic oxidation to drive the coupling reaction without external oxidants or metal catalysts. This approach employs common inert electrodes, such as carbon rods and platinum sheets, in a simple solvent system comprising acetonitrile and electrolytes like lithium perchlorate. The process operates at room temperature under air conditions, significantly reducing energy consumption and safety risks associated with high-temperature or high-pressure reactions. By avoiding the use of toxic catalysts and hazardous oxidants, the method simplifies the workup procedure and minimizes environmental impact, aligning with modern green chemistry principles. The broad substrate scope allows for the synthesis of various substituted 1,1'-binaphthyl derivatives, including those with alkyl and alkoxy groups, which were previously difficult to access efficiently. 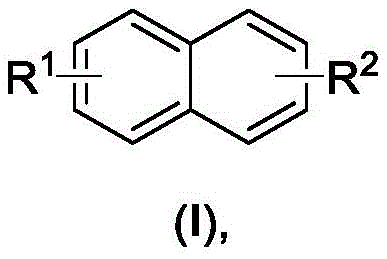
Mechanistic Insights into Electrochemical Oxidative Coupling
The core mechanism of this synthesis involves the anodic oxidation of the naphthalene substrate to generate a radical cation intermediate, which subsequently undergoes dimerization to form the 1,1'-binaphthyl skeleton. This electrochemical activation bypasses the need for chemical oxidants, as the electrode surface serves as the electron acceptor, driving the reaction forward with high atom economy. The use of a diaphragm-free cell simplifies the reactor design, allowing for easier scale-up and operation compared to divided cell systems. The reaction kinetics are controlled by the applied constant current, ensuring consistent conversion rates and minimizing over-oxidation side reactions that could lead to impurities. This precise control over the oxidation potential is crucial for maintaining high selectivity and yield, particularly when dealing with sensitive functional groups on the naphthalene ring. 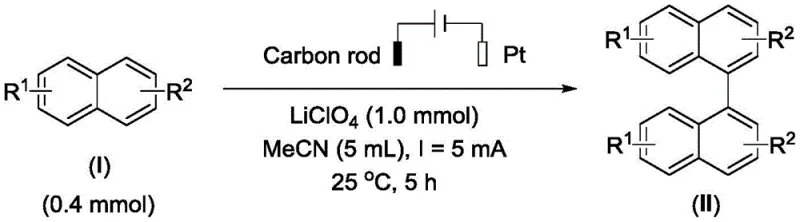
Impurity control is inherently enhanced in this metal-free system, as there are no transition metal residues to remove during the purification stage. The absence of stoichiometric oxidants also eliminates the formation of reduced oxidant byproducts, which often co-elute with the desired product in chromatographic separations. The reaction conditions, specifically the use of acetonitrile and lithium perchlorate, provide a stable environment that suppresses unwanted side reactions such as polymerization or over-oxidation. Post-reaction workup involves simple solvent removal and column chromatography, yielding products with high purity suitable for downstream applications. This streamlined purification process reduces solvent consumption and waste generation, further contributing to the overall cost reduction in fine chemical manufacturing. For quality control teams, the consistent impurity profile ensures reliable batch-to-batch reproducibility, a critical factor for regulatory compliance.
How to Synthesize 1,1'-Binaphthyl Compounds Efficiently
To implement this synthesis effectively, operators must first prepare the electrolytic cell by sequentially adding the solvent, electrolyte, and naphthalene compound into a diaphragm-free vessel equipped with inert electrodes. The reaction is then initiated by applying a constant current at room temperature, with progress monitored via TLC to ensure optimal conversion without over-reaction. Detailed standard operating procedures regarding electrode preparation, current density optimization, and workup protocols are essential for maximizing yield and safety.
- Prepare the electrolytic cell by adding solvent, electrolyte, and naphthalene compound into a diaphragm-free cell with inert electrodes.
- Apply constant current at room temperature under air conditions to initiate the oxidative coupling reaction.
- Concentrate the reaction mixture and purify the organic phase via column chromatography to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this electrochemical methodology offers substantial advantages by eliminating the procurement of expensive transition metal catalysts and hazardous chemical oxidants. The reduction in raw material complexity directly translates to significant cost savings, as the supply chain is no longer vulnerable to price fluctuations of precious metals like palladium or nickel. Furthermore, the simplified reaction system reduces the need for specialized equipment for handling toxic reagents, lowering capital expenditure for manufacturing facilities. The environmental benefits also mitigate regulatory compliance costs, as the process generates less hazardous waste requiring disposal. These factors collectively enhance the economic viability of producing 1,1'-binaphthyl compounds on a large scale.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the need for costly metal scavenging steps and reduces the risk of product contamination, leading to lower overall production costs. By avoiding stoichiometric oxidants, the process minimizes waste disposal fees and reduces the consumption of auxiliary chemicals. The use of common inert electrodes and simple solvents further drives down operational expenses, making the process highly competitive. This cost structure allows for more flexible pricing strategies in the supply of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials and common electrolytes ensures a stable supply chain不受 geopolitical disruptions affecting rare metal markets. The simplicity of the reaction setup reduces the risk of operational delays caused by equipment failure or reagent shortages. Additionally, the ambient temperature conditions lower energy dependencies, making the process resilient to energy price volatility. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for global clients.
- Scalability and Environmental Compliance: The diaphragm-free cell design and mild reaction conditions facilitate easy scale-up from laboratory to industrial production without significant process re-engineering. The absence of toxic heavy metals and hazardous oxidants simplifies environmental permitting and reduces the burden of waste treatment compliance. This green profile aligns with corporate sustainability goals, enhancing the marketability of the final products. The process is well-suited for commercial scale-up of complex polymer additives and chiral ligands, ensuring long-term viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 1,1'-binaphthyl compounds, based on the detailed specifications provided in the patent documentation. These answers are designed to clarify the operational benefits and feasibility of adopting this technology for industrial applications.
Q: Does this electrochemical method require transition metal catalysts?
A: No, the process described in patent CN113957461A operates without any transition metal catalysts or stoichiometric oxidants, eliminating heavy metal residue concerns.
Q: What is the substrate scope for this synthesis?
A: The method accommodates a wide range of naphthalene derivatives, including those with alkyl, alkoxy, and ester substituents, offering broader applicability than previous electrochemical methods.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of common inert electrodes, simple solvent systems, and ambient temperature conditions makes the process highly scalable and safe for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1'-Binaphthyl Compounds Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting innovative synthesis technologies to deliver high-quality chemical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the electrochemical method described in patent CN113957461A can be seamlessly integrated into our manufacturing operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1,1'-binaphthyl compounds meets the highest industry standards. Our commitment to green chemistry and process efficiency allows us to offer competitive pricing without compromising on quality or safety.
We invite procurement leaders and technical directors to collaborate with us on optimizing their supply chains for chiral ligands and functional materials. By leveraging our expertise in electrochemical synthesis, we can provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Together, we can drive innovation and efficiency in the production of high-value fine chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
