Revolutionizing Triazole Synthesis: Iodine-Catalyzed Manufacturing for Commercial-Scale Pharmaceutical Intermediates
Patent CN110467579B introduces a groundbreaking iodine-catalyzed methodology for synthesizing 5-trifluoromethyl-substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This innovative process eliminates the need for transition metal catalysts while operating under ambient atmospheric conditions, addressing critical limitations in conventional triazole synthesis. The methodology leverages readily available starting materials including trifluoroethylimidoyl chloride and hydrazones, enabling the production of structurally diverse triazole derivatives with exceptional functional group tolerance. Crucially, the process maintains high yields across various substitution patterns without requiring specialized anhydrous or anaerobic environments, thereby simplifying manufacturing infrastructure requirements. This patent establishes a robust foundation for producing high-purity triazole intermediates essential for next-generation pharmaceutical development while significantly enhancing process sustainability and operational flexibility in commercial manufacturing settings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches for synthesizing trifluoromethyl-substituted triazoles typically require stringent anhydrous and anaerobic conditions that necessitate specialized equipment and complex operational protocols, significantly increasing both capital expenditure and operational costs. These methods frequently employ transition metal catalysts such as palladium or copper complexes, which introduce substantial contamination risks requiring extensive purification steps to meet pharmaceutical quality standards. The reliance on expensive trifluoromethylation reagents and the narrow substrate scope of existing methodologies severely limit structural diversity and commercial scalability. Furthermore, conventional processes often suffer from inconsistent yields when incorporating different functional groups, creating significant challenges for producing diverse triazole derivatives required in pharmaceutical development pipelines. The multi-step purification protocols needed to remove metal residues also contribute to extended production timelines and higher waste generation, making these approaches economically and environmentally unsustainable for large-scale manufacturing.
The Novel Approach
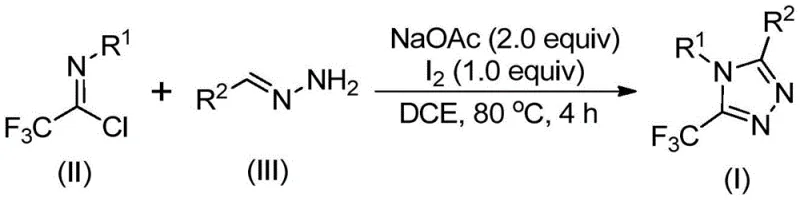
The patented methodology overcomes these limitations through an elegant iodine-catalyzed cyclization process that operates effectively under standard atmospheric conditions without requiring specialized moisture or oxygen exclusion systems. By utilizing elemental iodine as a non-toxic catalyst and inexpensive sodium acetate as a base promoter, the process eliminates both heavy metal contamination risks and the associated purification burdens that plague conventional methods. The reaction demonstrates remarkable substrate flexibility, accommodating a wide range of aryl and heteroaryl substitutions at both R¹ and R² positions while maintaining consistent high yields across diverse structural variants. This approach leverages commercially available starting materials including readily synthesized trifluoroethylimidoyl chloride and hydrazones derived from abundant aldehydes, creating a cost-effective and sustainable manufacturing pathway. The simplified workup procedure involving basic filtration and column chromatography further enhances process efficiency while ensuring high product purity suitable for pharmaceutical applications.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism proceeds through a sophisticated sequence beginning with base-promoted intermolecular carbon-nitrogen bond formation between trifluoroethylimidoyl chloride and hydrazone to generate a trifluoroacetamidine intermediate. This key intermediate undergoes spontaneous isomerization followed by iodine-mediated oxidative iodination to form a critical iodinated species that facilitates intramolecular electrophilic substitution. The subsequent cyclization step drives aromatization through elimination processes, ultimately yielding the thermodynamically stable 5-trifluoromethyl-substituted 1,2,4-triazole core structure. The iodine catalyst plays a dual role in promoting both the oxidation step and facilitating the final cyclization event through halogen bonding interactions that lower the activation energy barrier for ring closure. This mechanistic pathway operates efficiently at moderate temperatures (80°C) in common aprotic solvents like dichloroethane, demonstrating exceptional functional group compatibility across diverse substitution patterns.
Impurity control is inherently achieved through the reaction's self-regulating nature where the iodine catalyst promotes selective cyclization while suppressing common side reactions such as hydrolysis or oxidation that typically plague metal-catalyzed processes. The absence of transition metals eliminates metal-derived impurities that require extensive purification protocols in conventional methods, while the moderate reaction temperature prevents thermal decomposition pathways that could generate byproducts. The process demonstrates excellent regioselectivity for the desired 5-trifluoromethyl substitution pattern across various substrate combinations, with minimal formation of regioisomers even with complex functionalized starting materials. This inherent selectivity significantly reduces downstream purification requirements while ensuring consistent product quality suitable for pharmaceutical intermediate applications where strict impurity profiles are mandated by regulatory authorities.
How to Synthesize 5-Trifluoromethyl Triazoles Efficiently
This iodine-catalyzed methodology represents a significant advancement in triazole synthesis technology that addresses critical pain points in pharmaceutical intermediate manufacturing. The process eliminates traditional barriers associated with metal catalysts and specialized reaction environments while delivering exceptional structural diversity through simple substrate modifications. Detailed standardized synthesis protocols have been developed based on patent CN110467579B to ensure consistent product quality and yield across different manufacturing scales. The following step-by-step guide provides essential operational parameters for successful implementation of this innovative manufacturing approach in commercial settings.
- Combine trifluoroethylimidoyl chloride (II), hydrazone (III), and sodium acetate in dichloroethane solvent under ambient conditions without requiring anhydrous or anaerobic environments.
- Heat the reaction mixture to 80°C for four hours to facilitate base-promoted carbon-nitrogen bond formation and subsequent isomerization into key intermediates.
- Introduce elemental iodine to enable oxidative iodination followed by intramolecular electrophilic substitution and aromatization to yield the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
This novel manufacturing approach delivers substantial value across procurement and supply chain operations by addressing fundamental challenges in pharmaceutical intermediate sourcing. The elimination of transition metal catalysts removes significant cost drivers associated with catalyst procurement, handling, and post-reaction removal processes while simultaneously enhancing supply chain resilience through simplified raw material requirements. The process design inherently supports seamless scale-up from laboratory to commercial production volumes without requiring major equipment modifications or process revalidation, creating significant operational flexibility for manufacturers responding to fluctuating market demands.
- Cost Reduction in Manufacturing: The complete elimination of expensive transition metal catalysts removes both procurement costs and the substantial downstream processing expenses required for metal residue removal from final products. This metal-free approach significantly reduces overall manufacturing costs by streamlining purification protocols while avoiding the need for specialized analytical testing to confirm metal contamination levels below regulatory thresholds. The use of commercially available starting materials at optimal stoichiometric ratios further enhances cost efficiency without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials including sodium acetate, elemental iodine, and common organic solvents creates exceptional supply chain resilience by eliminating dependence on specialized or restricted reagents. The ambient condition operation enables flexible manufacturing across diverse geographical locations without requiring specialized infrastructure investments, while the robust reaction profile maintains consistent performance despite minor variations in raw material quality or environmental conditions. This inherent process robustness significantly reduces supply chain disruption risks while ensuring reliable delivery timelines for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The straightforward scale-up pathway from laboratory to commercial production volumes is facilitated by the absence of hazardous reagents or extreme reaction conditions that typically complicate manufacturing expansion. The simplified workup procedure minimizes waste generation compared to conventional methods requiring multiple purification steps to remove metal contaminants, resulting in a more environmentally sustainable process with reduced EHS management requirements. This green chemistry approach aligns with increasing regulatory pressure for sustainable manufacturing practices while supporting corporate environmental stewardship goals without compromising production efficiency.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations for implementing this innovative triazole synthesis methodology in pharmaceutical manufacturing operations. These FAQs are derived directly from patent CN110467579B's technical specifications and practical implementation experience to provide actionable insights for procurement and R&D decision-makers evaluating this manufacturing approach.
Q: How does this method eliminate heavy metal contamination risks in API manufacturing?
A: The iodine-catalyzed process completely avoids transition metal catalysts that typically require complex purification steps to remove toxic residues. This inherently eliminates heavy metal contamination risks during synthesis of pharmaceutical intermediates, directly addressing stringent regulatory requirements for API purity without additional processing steps.
Q: What supply chain advantages does the ambient condition operation provide?
A: By operating under standard atmospheric conditions without requiring specialized anhydrous or anaerobic equipment, the process significantly reduces facility complexity and operational constraints. This enables more flexible manufacturing locations and faster scale-up timelines while maintaining consistent product quality across different production sites.
Q: How does substrate design flexibility impact commercial production scalability?
A: The modular design allows substitution at both R¹ and R² positions using readily available starting materials, enabling rapid adaptation to specific customer requirements without process revalidation. This structural versatility supports seamless scale-up from laboratory to commercial production while maintaining high functional group tolerance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic intermediates while maintaining stringent purity specifications through rigorous QC labs and advanced analytical capabilities. We have successfully implemented this patented iodine-catalyzed methodology across multiple client projects, demonstrating consistent ability to deliver high-purity triazole intermediates meeting exacting pharmaceutical requirements through our integrated CDMO platform. Our technical team brings deep expertise in process optimization and scale-up challenges specific to fluorinated heterocycles, ensuring seamless technology transfer from laboratory to commercial manufacturing environments while maintaining robust quality control throughout production cycles.
Leverage our technical procurement team's expertise through a Customized Cost-Saving Analysis to evaluate how this innovative manufacturing approach can optimize your specific supply chain requirements. We invite qualified partners to request specific COA data and route feasibility assessments tailored to your unique production needs through our dedicated technical support channels.
