Efficient Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Drug Discovery
Efficient Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Drug Discovery
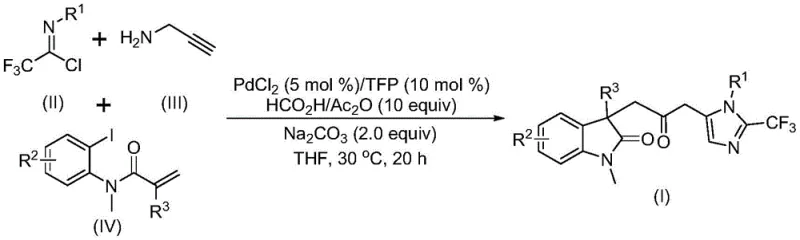
The rapid advancement of medicinal chemistry demands robust and versatile synthetic methodologies capable of constructing complex molecular architectures with high efficiency. In this context, patent CN115353511A introduces a groundbreaking multi-component strategy for the preparation of carbonyl-bridged biheterocyclic compounds, specifically targeting scaffolds that combine indolinone and imidazole motifs. These structural frameworks are ubiquitous in bioactive natural products and pharmaceutical agents, making their efficient synthesis a high priority for R&D teams globally. The disclosed method leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives as accessible starting materials. By operating under mild conditions without the need for hazardous carbon monoxide gas, this technology represents a significant leap forward in safe and sustainable chemical manufacturing, offering a reliable pathway for producing high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems has relied on laborious multi-step sequences that often suffer from poor atom economy and low overall yields. Traditional approaches typically involve the direct coupling of two pre-formed heterocyclic substrates, which requires the prior synthesis and isolation of each distinct ring system, thereby increasing material costs and processing time. Alternatively, oxidative cyclization reactions using activated methyl-substituted heterocycles often necessitate harsh oxidizing agents and elevated temperatures, leading to significant safety concerns and the generation of toxic waste streams. Furthermore, conventional carbonylation strategies frequently depend on the use of high-pressure carbon monoxide gas, which poses severe operational hazards and requires specialized, expensive reactor infrastructure that is not readily available in standard process development laboratories. These limitations collectively hinder the rapid exploration of chemical space and delay the timeline for bringing new drug candidates to clinical trials.
The Novel Approach
In stark contrast to these legacy techniques, the novel methodology described in the patent utilizes a sophisticated one-pot, three-component coupling reaction that streamlines the entire synthetic sequence into a single operational step. This approach ingeniously replaces toxic carbon monoxide gas with a safe and manageable mixture of formic acid and acetic anhydride, which generates the necessary carbonyl species in situ under ambient pressure. The reaction proceeds efficiently at a mild temperature of 30°C, demonstrating exceptional energy efficiency compared to thermal-intensive traditional methods. By merging the formation of the indolinone core and the trifluoromethyl-substituted imidazole ring in a tandem fashion, this process drastically reduces the number of purification steps required, thereby minimizing solvent consumption and waste generation. The ability to tolerate a wide range of functional groups on the substrate allows chemists to rapidly generate diverse libraries of analogs for structure-activity relationship studies without redesigning the core synthetic route.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The success of this transformation relies on a meticulously orchestrated catalytic cycle initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. Following this activation, an intramolecular Heck-type reaction occurs, generating a divalent alkyl-palladium intermediate that sets the stage for the subsequent carbonylation event. The unique combination of formic acid and acetic anhydride serves as a surrogate for carbon monoxide, releasing the reactive carbonyl species which then inserts into the palladium-carbon bond to form a crucial acyl-palladium intermediate. Simultaneously, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine species in situ, which undergoes isomerization to become the active nucleophile for the final cyclization step. This dual-activation strategy ensures that both the electrophilic acyl component and the nucleophilic nitrogen component are generated concurrently, driving the reaction towards the formation of the carbonyl-bridged biheterocyclic product with high regioselectivity.
From an impurity control perspective, the use of trifurylphosphine (TFP) as a ligand plays a critical role in stabilizing the palladium center and preventing the formation of inactive palladium black, which is a common cause of catalyst deactivation in carbonylation reactions. The presence of sodium carbonate as a base not only facilitates the deprotonation steps required for amidine formation but also helps to neutralize acidic byproducts, maintaining a stable pH environment that favors the desired catalytic cycle over competing hydrolysis pathways. The mild reaction temperature of 30°C further suppresses thermal degradation of sensitive functional groups, ensuring that the final product profile remains clean with minimal side products. This mechanistic robustness translates directly to simplified downstream processing, as the crude reaction mixture typically requires only filtration and standard column chromatography to achieve pharmaceutical-grade purity, eliminating the need for complex crystallization protocols or extensive recrystallization cycles.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this advanced synthetic route in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and mixing protocols to maximize yield and reproducibility. The process begins with the preparation of the catalytic system, where palladium chloride and the TFP ligand are combined with the carbon monoxide substitute mixture in an aprotic solvent such as tetrahydrofuran. It is critical to ensure complete dissolution of all solid reagents before introducing the substrates to prevent localized concentration gradients that could lead to uneven reaction rates. Once the reaction mixture is homogenized, the trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivative are added, and the system is maintained at 30°C with continuous stirring for a duration of 12 to 20 hours. Detailed standardized synthesis steps follow below to guide your technical team through the exact execution of this protocol.
- Combine palladium chloride, trifurylphosphine, sodium carbonate, and the CO substitute mixture (formic acid/acetic anhydride) in an organic solvent like THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the acrylamide derivative to the reaction vessel.
- Stir the mixture at 30°C for 12 to 20 hours, then filter and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method offers transformative benefits that extend far beyond simple yield improvements. The elimination of high-pressure carbon monoxide gas removes a major bottleneck in facility qualification, allowing production to occur in standard glass-lined reactors rather than specialized high-pressure autoclaves, which significantly lowers capital expenditure requirements. Furthermore, the reliance on commercially available and inexpensive starting materials such as propargylamine and acrylamide derivatives ensures a stable and resilient supply chain that is less susceptible to market volatility compared to exotic or custom-synthesized precursors. The operational simplicity of the one-pot process reduces labor costs and minimizes the risk of human error during material transfer between steps, contributing to a more predictable and consistent manufacturing output. These factors collectively position this technology as a highly attractive option for cost reduction in pharmaceutical intermediate manufacturing, enabling companies to maintain competitive pricing while adhering to strict quality standards.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substitution of expensive and hazardous reagents with commodity chemicals that are sourced globally at low cost. By avoiding the need for specialized high-pressure equipment and the associated safety monitoring systems, facilities can achieve substantial cost savings in both infrastructure maintenance and operational overhead. The high atom economy of the multi-component reaction ensures that a greater proportion of the input mass is converted into the final product, reducing the volume of raw materials required per kilogram of output. Additionally, the simplified workup procedure involving basic filtration and chromatography minimizes solvent usage and waste disposal fees, further enhancing the overall cost-efficiency of the production line.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the use of robust and widely available reagents that do not rely on single-source suppliers or complex logistics networks. The stability of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuations in demand without lengthy lead times for raw material procurement. The tolerance for various functional groups means that a single platform technology can be adapted to produce a wide array of derivatives, reducing the need to qualify multiple distinct synthetic routes for different projects. This versatility provides a strategic buffer against supply disruptions, ensuring that critical pharmaceutical intermediates can be delivered consistently to downstream customers.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is facilitated by the mild reaction parameters and the absence of gaseous reagents that are difficult to handle in large volumes. The process generates minimal hazardous waste, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The ability to operate at near-ambient temperatures reduces energy consumption for heating and cooling, contributing to a lower carbon footprint for the manufacturing process. As the patent indicates successful gram-scale expansion, the pathway is well-suited for scaling up to multi-kilogram or ton-level production, providing a clear route for meeting the growing demands of the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on reaction scope, safety, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing process development workflows.
Q: What are the primary advantages of this multi-component synthesis over traditional methods?
A: This method avoids the use of toxic carbon monoxide gas by utilizing a formic acid/acetic anhydride system, operates under mild conditions (30°C), and achieves high yields in a one-pot process, significantly simplifying purification and reducing safety risks.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly demonstrates successful expansion to gram-scale reactions with consistent yields, indicating strong potential for commercial scale-up due to the use of cheap, readily available starting materials and standard laboratory equipment.
Q: What is the substrate compatibility of this palladium-catalyzed reaction?
A: The reaction exhibits excellent functional group tolerance, accommodating various substituents such as halogens, alkyl groups, alkoxy groups, and nitro groups on the aromatic rings, allowing for the diverse synthesis of drug-like scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to accelerate drug discovery and development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory results can be seamlessly translated into reliable industrial supply. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging innovations such as the palladium-catalyzed multi-component reaction described here, we empower our partners to overcome synthetic challenges and bring life-saving therapies to patients faster.
We invite you to engage with our technical procurement team to discuss how this cutting-edge methodology can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of this route for your target molecule, we are ready to provide comprehensive support. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your pharmaceutical manufacturing operations.
