Advanced Multicomponent Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharma
Advanced Multicomponent Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Pharma
The pharmaceutical industry continuously seeks efficient pathways to construct complex heterocyclic scaffolds that serve as the backbone for novel therapeutic agents. Patent CN115353511A introduces a groundbreaking multicomponent method for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This technology represents a significant leap forward in organic synthesis, offering a streamlined route to high-value intermediates that are notoriously difficult to access via conventional means. By leveraging a palladium-catalyzed cascade reaction, this method avoids the use of hazardous carbon monoxide gas, instead utilizing a safe in situ generation system. For R&D directors and process chemists, this innovation opens new avenues for library synthesis and lead optimization, providing a robust platform for generating diverse chemical space around the biheterocyclic core.
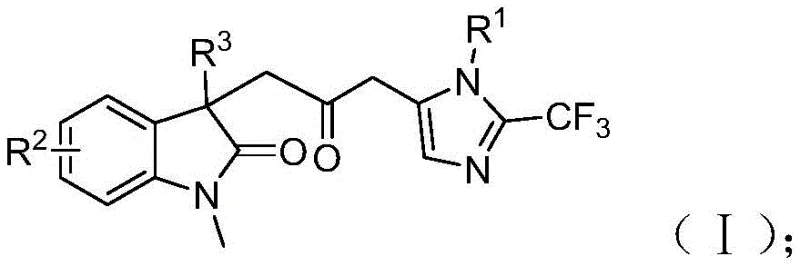
The structural complexity of carbonyl-bridged biheterocycles often necessitates multi-step sequences with protecting group manipulations, which inherently lowers overall yield and increases waste. Traditional approaches to similar scaffolds frequently rely on the direct coupling of pre-formed heterocycles or oxidative cyclization strategies that suffer from poor atom economy and harsh reaction conditions. Furthermore, classical carbonylation reactions typically require high-pressure equipment and toxic CO gas, posing significant safety risks and infrastructure costs for manufacturing facilities. These limitations create bottlenecks in the supply chain for critical pharmaceutical intermediates, driving up costs and extending lead times for drug development programs. The inability to easily introduce diverse functional groups at key positions further restricts the medicinal chemistry utility of compounds produced by older methodologies.
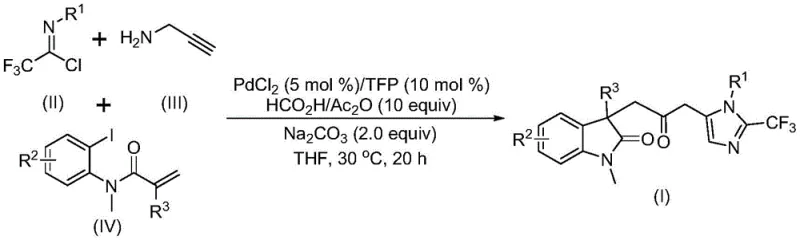
In stark contrast, the novel approach detailed in the patent utilizes a one-pot, three-component coupling strategy that dramatically simplifies the synthetic workflow. By reacting trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives in the presence of a palladium catalyst, the method constructs multiple chemical bonds simultaneously. This telescoped process not only improves step economy but also enhances the overall mass balance of the reaction. The use of mild reaction conditions, specifically operating at 30°C in tetrahydrofuran (THF), ensures compatibility with sensitive functional groups that might degrade under thermal stress. This methodology transforms the synthesis of these complex architectures from a challenging, multi-step ordeal into a straightforward, scalable operation suitable for modern process chemistry.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a sophisticated example of transition metal catalysis, initiating with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This is followed by an intramolecular Heck reaction that generates a divalent alkyl-palladium intermediate, setting the stage for ring closure. Crucially, the carbon monoxide required for the carbonyl bridge is released in situ from the decomposition of the formic acid and acetic anhydride mixture, which then inserts into the palladium-carbon bond to form an acyl-palladium species. This elegant sequence avoids the logistical nightmares associated with external CO sources while maintaining high reaction efficiency. The subsequent activation of the trifluoroacetamidine intermediate, formed from the condensation of the imidoyl chloride and propargylamine, facilitates the final cyclization event.
From an impurity control perspective, this mechanism offers distinct advantages due to its high selectivity and mild conditions. The base-promoted formation of the carbon-nitrogen bond occurs prior to the palladium-catalyzed cyclization, minimizing side reactions such as polymerization of the alkyne or hydrolysis of the acid chloride. The specific choice of trifurylphosphine as a ligand stabilizes the palladium center, preventing the formation of palladium black and ensuring a consistent catalytic turnover number. This stability is critical for maintaining high purity profiles in the crude reaction mixture, thereby reducing the burden on downstream purification processes like column chromatography. For quality control teams, understanding this mechanism confirms that the process is robust against minor variations in reagent stoichiometry, ensuring batch-to-batch consistency.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Executing this synthesis requires careful attention to the order of addition and the quality of the palladium catalyst system. The protocol dictates mixing the catalyst, ligand, base, and CO source mixture before introducing the organic substrates to ensure the active catalytic species is generated prior to substrate exposure. The reaction is typically conducted in anhydrous THF to prevent hydrolysis of the reactive imidoyl chloride intermediate. Maintaining the temperature at 30°C is vital; while higher temperatures might accelerate the rate, they could also promote decomposition of the sensitive intermediates. Upon completion, usually within 12 to 20 hours, the workup involves simple filtration to remove inorganic salts followed by silica gel treatment. Detailed standardized synthesis steps are provided in the guide below.
- Combine palladium chloride, trifurylphosphine ligand, sodium carbonate, and the formic acid/acetic anhydride CO source mixture in a reaction vessel.
- Add trifluoroethylimidoyl chloride, propargylamine, and the acrylamide substrate into the organic solvent (preferably THF) under stirring.
- Maintain the reaction mixture at 30°C for 12 to 20 hours, followed by filtration, silica gel treatment, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits regarding cost structure and operational risk mitigation. The elimination of high-pressure carbon monoxide gas removes the need for specialized autoclaves and rigorous gas handling safety protocols, which translates directly into reduced capital expenditure (CAPEX) for manufacturing sites. Furthermore, the starting materials—trifluoroethylimidoyl chloride, propargylamine, and acrylamides—are commodity chemicals available from multiple global suppliers, ensuring a resilient supply chain that is not dependent on single-source vendors. This diversification of raw material sources significantly de-risks the production schedule against market fluctuations or geopolitical disruptions.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive palladium chloride and avoiding expensive proprietary reagents or cryogenic conditions. The high atom economy of the multicomponent reaction minimizes waste disposal costs, while the mild conditions reduce energy consumption associated with heating and cooling cycles. Additionally, the simplified workup procedure reduces solvent usage and labor hours required for purification, leading to a lower overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: By relying on stable, shelf-stable solid reagents rather than hazardous gases, inventory management becomes safer and more straightforward. The robustness of the reaction across a wide range of substrates means that a single manufacturing line can be adapted to produce various analogues without extensive re-validation. This flexibility allows for rapid response to changing demand signals from R&D or clinical trial phases, ensuring that material is available exactly when needed without long lead times for process redevelopment.
- Scalability and Environmental Compliance: The protocol has been demonstrated to work effectively on a gram scale, indicating a clear path to kilogram and ton-scale production without fundamental changes to the chemistry. The avoidance of toxic CO gas greatly simplifies environmental permitting and reduces the facility's environmental footprint. Waste streams are primarily organic solvents and inorganic salts, which are easier to treat and dispose of compared to heavy metal-contaminated wastes from other transition metal processes, aligning with modern green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the method. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the safety advantages of this carbonylation method compared to traditional CO gas methods?
A: This protocol utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in situ, completely eliminating the need for handling toxic, high-pressure carbon monoxide gas cylinders, thereby significantly enhancing operational safety and reducing regulatory compliance burdens.
Q: What is the substrate scope for the R1 and R3 groups in this synthesis?
A: The method demonstrates excellent functional group tolerance, accommodating alkyl, substituted or unsubstituted aryl groups for R1, and alkyl, phenyl, or benzyl groups for R3, including substituents like halogens, trifluoromethyl, and nitro groups without compromising yield.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the reaction operates under mild conditions (30°C) using commercially available catalysts and solvents, and has been validated for gram-scale synthesis, indicating strong potential for seamless scale-up to industrial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates for the development of next-generation therapeutics. Our team of expert process chemists has extensively evaluated this palladium-catalyzed carbonylation technology and possesses the capability to implement it seamlessly within our GMP-compliant facilities. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale manufacturing. Our rigorous QC labs and stringent purity specifications guarantee that every batch of carbonyl-bridged biheterocyclic compounds meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this innovative synthesis for your drug discovery programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects, ensuring a secure and efficient supply chain for your critical APIs.
