Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Pharmaceutical Applications
Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Pharmaceutical Applications
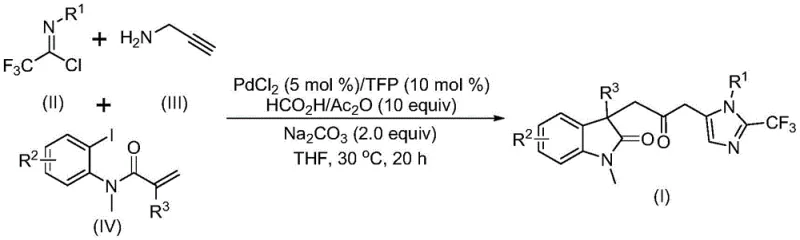
The pharmaceutical and fine chemical industries are constantly seeking more efficient, safer, and cost-effective pathways to construct complex molecular scaffolds, particularly those containing biheterocyclic motifs which are prevalent in bioactive molecules. A significant breakthrough in this domain is documented in Chinese Patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. This technology represents a paradigm shift from traditional step-wise syntheses to a streamlined, one-pot cascade reaction catalyzed by transition metals. By integrating indolinone and imidazole frameworks through a carbonyl bridge, this method addresses critical challenges in medicinal chemistry, offering a robust platform for generating diverse libraries of potential drug candidates. The innovation lies not only in the structural complexity achieved but also in the operational simplicity, utilizing mild temperatures and avoiding the logistical nightmares associated with handling toxic gases, thereby positioning it as a highly attractive route for reliable pharmaceutical intermediate supplier networks aiming to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems, especially those bridged by carbonyl functionalities, has been fraught with synthetic difficulties and safety hazards. Conventional literature reports typically rely on three primary strategies: the direct coupling of two pre-formed heterocyclic substrates, oxidative cyclization of substrates containing dual nucleophiles with activated methyl-substituted heterocycles, or complex transition metal-catalyzed tandem cyclizations. The direct coupling approach often suffers from low atom economy and requires harsh conditions that may degrade sensitive functional groups, while oxidative cyclization methods frequently necessitate stoichiometric amounts of oxidants, generating substantial waste and complicating downstream purification. Furthermore, traditional carbonylation reactions to install the bridging carbonyl group almost universally depend on the use of carbon monoxide gas, which is highly toxic, flammable, and requires specialized high-pressure equipment and rigorous safety protocols, creating significant barriers to entry for many manufacturing facilities and inflating the capital expenditure required for cost reduction in API manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN115353511A introduces a sophisticated yet operationally simple palladium-catalyzed carbonylation cascade reaction. This novel approach utilizes cheap and readily available starting materials, specifically trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, to construct the target biheterocyclic architecture in a single pot. The reaction proceeds under remarkably mild conditions, typically at 30°C, which preserves the integrity of thermally sensitive substituents and minimizes energy consumption. Crucially, this method eliminates the need for external carbon monoxide gas cylinders by employing a formic acid and acetic anhydride mixture as a safe, in situ CO source. This strategic substitution not only enhances laboratory and plant safety but also simplifies the reactor setup, making the process highly amenable to commercial scale-up of complex pharmaceutical intermediates without the need for exotic high-pressure infrastructure.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, involving a intricate sequence of bond-forming events orchestrated by a palladium center. The reaction is believed to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating an aryl-palladium intermediate. This is swiftly followed by an intramolecular Heck-type reaction, which constructs the indolinone core and yields a divalent alkyl-palladium species. Subsequently, the carbon monoxide generated in situ from the decomposition of the formic acid and acetic anhydride mixture inserts into the palladium-carbon bond, forming a reactive acyl-palladium intermediate. This carbonylation step is the linchpin of the process, effectively installing the crucial carbonyl bridge that links the two heterocyclic domains. Concurrently, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine intermediate, which undergoes isomerization to become susceptible to nucleophilic attack or cyclization.
The final stage of the catalytic cycle involves the activation of this trifluoroacetamidine species by the acyl-palladium intermediate, triggering an intramolecular cyclization that closes the imidazole ring and releases the final carbonyl-bridged biheterocyclic product while regenerating the active palladium catalyst. This complex interplay of reactions—heck cyclization, carbonylation, and amidine formation—occurring in a single vessel highlights the high efficiency and atom economy of the process. From a purity and impurity profile perspective, the use of specific ligands like tri-furyl phosphine (TFP) alongside palladium chloride ensures high selectivity, minimizing the formation of side products such as homocoupling byproducts or incomplete cyclization intermediates. The compatibility with various functional groups, including halogens and electron-withdrawing groups like nitro and trifluoromethyl, suggests that the catalytic cycle is robust against electronic variations in the substrate, providing R&D teams with a versatile tool for structure-activity relationship (SAR) studies without compromising on the cleanliness of the crude reaction mixture.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize yield and minimize impurities, leveraging the specific conditions optimized in the patent data. The process begins with the preparation of a catalytic system comprising palladium chloride and the TFP ligand in an aprotic organic solvent, with tetrahydrofuran (THF) identified as the preferred medium due to its superior solubilizing properties and reaction promotion capabilities. The detailed standardized synthesis steps involve the sequential or simultaneous addition of the three distinct building blocks along with the necessary additives, maintaining a strict molar ratio to ensure complete conversion. For those interested in the precise operational details, the detailed standardized synthesis steps are provided in the guide below.
- Combine palladium chloride catalyst, TFP ligand, sodium carbonate base, and the formic acid/acetic anhydride CO source mixture in an organic solvent such as THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative into the reaction vessel under stirring.
- Maintain the reaction mixture at 30°C for 12 to 20 hours, followed by filtration, silica gel treatment, and column chromatography purification to isolate the final biheterocyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology offers profound strategic advantages that extend beyond mere chemical novelty. The shift towards this multi-component reaction directly addresses several pain points associated with traditional pharmaceutical intermediate sourcing, particularly regarding cost stability and supply continuity. By replacing hazardous gaseous reagents with liquid alternatives and utilizing commodity chemicals as starting materials, the process inherently de-risks the supply chain against volatile raw material markets and regulatory crackdowns on hazardous substance transport. This translates into a more predictable procurement landscape where lead times can be optimized, and the reliance on specialized, high-cost reagents is drastically diminished, fostering a more resilient supply network capable of withstanding global disruptions.
- Cost Reduction in Manufacturing: The economic implications of this technology are substantial, primarily driven by the elimination of expensive and dangerous carbon monoxide gas infrastructure. Traditional carbonylation processes often require dedicated high-pressure reactors and extensive safety monitoring systems, which represent significant capital expenditure and ongoing maintenance costs. By utilizing a formic acid and acetic anhydride mixture as the CO source, this method allows manufacturers to utilize standard glass-lined or stainless steel reactors, significantly lowering the barrier to entry and operational overhead. Furthermore, the starting materials, such as propargylamine and acrylamide derivatives, are commercially available in bulk quantities at competitive prices, and the use of palladium chloride, a relatively inexpensive palladium source compared to other complexes, further drives down the raw material cost per kilogram of the final product, enabling significant cost reduction in API manufacturing without sacrificing quality.
- Enhanced Supply Chain Reliability: Supply chain reliability is bolstered by the broad availability and stability of the requisite reagents. Unlike specialized organometallic precursors that may have long lead times or single-source dependencies, the components of this reaction system are widely produced by multiple chemical suppliers globally. This redundancy ensures that production schedules are not held hostage by the shortage of a single niche ingredient. Additionally, the mild reaction conditions (30°C) reduce the energy load on manufacturing facilities, allowing for more flexible production scheduling and reducing the risk of thermal runaways or equipment failures that could halt production lines. The robustness of the reaction across various substrates means that a single production line can be adapted to synthesize a wide array of analogues, enhancing the agility of the supply chain to respond to changing market demands for different drug candidates.
- Scalability and Environmental Compliance: The patent data explicitly demonstrates the successful expansion of this reaction to the gram scale, providing a clear proof-of-concept for industrial scalability. The simplified work-up procedure, involving filtration and standard column chromatography, avoids complex extraction protocols or cryogenic conditions that are difficult to manage on a multi-ton scale. From an environmental compliance standpoint, the avoidance of toxic CO gas aligns with increasingly stringent global safety and environmental regulations, reducing the permitting burden and liability exposure for manufacturing sites. The high atom economy of the multi-component approach also results in less chemical waste generation per unit of product, supporting sustainability goals and reducing the costs associated with waste disposal and treatment, making it an environmentally responsible choice for large-scale chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, derived directly from the experimental data and beneficial effects outlined in the intellectual property documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for your specific production needs, ensuring that all stakeholders have a clear understanding of the process capabilities and limitations before initiating pilot studies or commercial contracts.
Q: What are the safety advantages of this carbonylation method compared to traditional techniques?
A: Unlike conventional carbonylation reactions that require toxic and hazardous carbon monoxide gas under high pressure, this patented method utilizes a safe formic acid and acetic anhydride mixture to generate carbon monoxide in situ, significantly enhancing operational safety and reducing infrastructure requirements.
Q: Does this synthesis method support a wide range of substrate functional groups?
A: Yes, the process demonstrates excellent substrate compatibility, tolerating various substituents such as halogens, alkyl groups, alkoxy groups, nitro groups, and trifluoromethyl groups on the aromatic rings, allowing for the diverse design of biheterocyclic libraries for drug discovery.
Q: Is this synthetic route suitable for large-scale industrial production?
A: The patent explicitly confirms that the method has been successfully expanded to gram-scale reactions with high efficiency, indicating strong potential for commercial scale-up due to the use of cheap, readily available starting materials and mild reaction conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN115353511A for accelerating drug discovery and development pipelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory benchtop to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle complex multi-step syntheses and sensitive catalytic reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates. We are committed to leveraging our technical expertise to bring this innovative carbonyl-bridged biheterocyclic scaffold to market faster and more cost-effectively than ever before.
We invite you to collaborate with us to explore how this technology can enhance your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data for related analogues and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global pharmaceutical market through superior chemical manufacturing solutions.
