Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Intermediates for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access fluorinated heterocycles, which are critical scaffolds in modern drug design. Patent CN111423381A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing the long-standing challenges associated with introducing the trifluoromethyl group into nitrogen-containing heterocycles. This innovation leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts as key starting materials. Unlike traditional methods that often rely on hazardous or expensive trifluoromethyl synthons, this approach operates under remarkably mild conditions, specifically at 30°C, ensuring high reaction efficiency and exceptional substrate compatibility. For R&D directors and procurement specialists, this represents a significant shift towards safer, more cost-effective manufacturing routes for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with synthetic difficulties and safety concerns. Conventional literature methods predominantly rely on synthons such as trifluorodiazoethane, which poses significant explosion hazards and handling complexities in a commercial setting. Alternatively, the use of trifluoroethylimide acid halides has been reported, but their application has remained relatively narrow and underexplored due to perceived limitations in reactivity and scope. Traditional protocols often require harsh reaction conditions, elevated temperatures, or stoichiometric amounts of expensive reagents, which drastically inflate the cost of goods sold (COGS) and complicate waste management. Furthermore, many existing routes suffer from poor functional group tolerance, limiting the structural diversity accessible to medicinal chemists who need to rapidly iterate on lead compounds containing the metabolically stable trifluoromethyl motif.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by employing a palladium-catalyzed carbonylation series reaction that is both efficient and versatile. By combining trifluoroethylimidoyl chloride with propargylamine and diaryliodonium salts in the presence of a PdCl2/PPh3 catalyst system, the process achieves the construction of the imidazole core with high precision. The reaction utilizes a formic acid and acetic anhydride mixture as a carbon monoxide surrogate, eliminating the need for high-pressure CO gas cylinders, which significantly enhances operational safety. As illustrated in the general reaction scheme below, this one-pot transformation seamlessly integrates C-N bond formation, isomerization, and carbonylation steps.
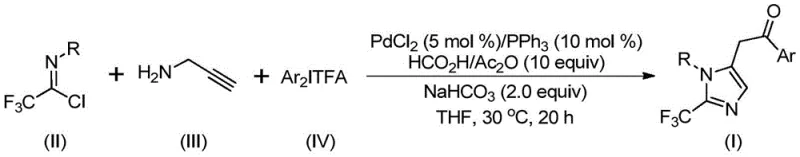
This novel approach not only simplifies the operational workflow but also broadens the utility of trifluoroethylimide acid halides, transforming them from niche reagents into powerful building blocks for diverse chemical libraries. The ability to conduct this transformation at a mild 30°C further underscores its potential for energy savings and reduced thermal degradation of sensitive intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic pathway is crucial for R&D teams aiming to optimize this process for specific analogs. The reaction is proposed to initiate with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, yielding a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization, followed by palladium-catalyzed aminopalladation of the alkyne moiety to generate an alkenyl palladium intermediate. A subsequent isomerization step converts this into a more stable alkyl palladium species. The critical carbonylation event occurs under the influence of carbon monoxide released in situ from the formic acid and acetic anhydride mixture, forming an acyl palladium intermediate. Finally, oxidative addition of the diaryliodonium salt generates a tetravalent palladium species, which undergoes reductive elimination to release the final 2-trifluoromethyl substituted imidazole product and regenerate the active catalyst.
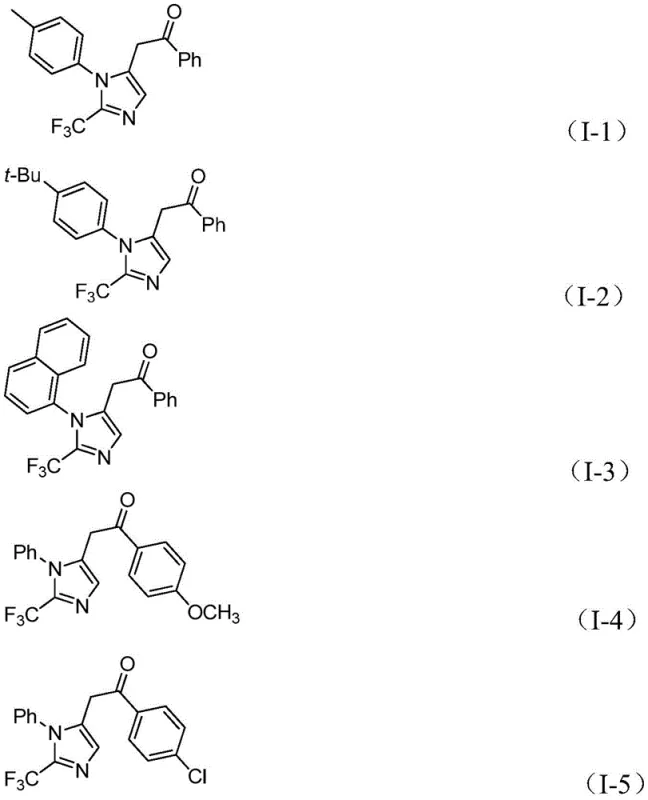
From an impurity control perspective, the mild temperature profile (30°C) is instrumental in suppressing side reactions such as polymerization of the alkyne or decomposition of the diazo-like intermediates that might occur at higher temperatures. The use of sodium bicarbonate as a mild base ensures that acid-sensitive functional groups on the aromatic rings remain intact. The broad substrate scope is evidenced by the successful synthesis of various derivatives, including those with electron-donating groups like methoxy and tert-butyl, as well as electron-withdrawing groups like chloro and nitro. The structural diversity achievable through this method is highlighted by the specific examples shown below, demonstrating the robustness of the catalytic cycle across different electronic environments.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The standardized protocol for synthesizing these valuable intermediates involves a straightforward mixing procedure in a Schlenk tube or standard reactor. The process begins with the combination of the palladium catalyst, ligand, base, and CO source in an aprotic organic solvent such as tetrahydrofuran (THF), which has been identified as the optimal medium for maximizing conversion rates. Following the addition of the three key organic substrates, the mixture is stirred at 30°C for a duration of 16 to 24 hours. Post-reaction processing is equally simple, involving filtration to remove inorganic salts, followed by silica gel treatment and purification via column chromatography. For detailed operational parameters and specific molar ratios optimized for different substrates, please refer to the technical guide below.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction vessel under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the accessibility and cost profile of the raw materials. Trifluoroethylimidoyl chloride can be rapidly synthesized from inexpensive aromatic amines, carbon tetrachloride, and trifluoroacetic acid, all of which are commodity chemicals available from multiple global suppliers. Similarly, propargylamine and diaryliodonium salts are commercially available or easily prepared from aryl boronic acids and aryl iodides, ensuring a resilient supply chain that is not dependent on single-source exotic reagents.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of low-loading palladium catalysts (5 mol%) and common ligands like triphenylphosphine, avoiding the need for expensive proprietary ligand systems. Furthermore, the elimination of high-pressure equipment required for gaseous carbon monoxide reduces capital expenditure (CAPEX) for reactor setup. The mild reaction temperature of 30°C significantly lowers energy consumption compared to traditional reflux conditions, contributing to substantial operational cost savings over large-scale production runs. The high yields reported, reaching up to 96% for certain substrates, minimize raw material waste and maximize throughput per batch.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials mitigates the risk of supply disruptions often associated with specialized fluorinating agents. The robustness of the reaction conditions allows for flexibility in sourcing; for instance, various substituted aromatic amines can be used to generate the necessary imidoyl chloride precursors, providing procurement teams with multiple vendor options. The scalability of the method, demonstrated from milligram to gram scales in the patent data, suggests a smooth path to kilogram and ton-scale production without the need for extensive process re-engineering, ensuring consistent supply continuity for downstream API manufacturing.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers significant advantages. The avoidance of explosive trifluorodiazoethane removes a major safety hazard from the manufacturing floor. The use of THF as a solvent allows for established recycling protocols, reducing volatile organic compound (VOC) emissions. Additionally, the simplified workup procedure, which avoids complex extraction sequences or hazardous quenching steps, reduces the volume of chemical waste generated. This aligns with modern green chemistry principles and facilitates easier regulatory compliance for facilities operating under strict environmental guidelines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: What are the key advantages of this trifluoromethyl imidazole synthesis method?
A: This method utilizes cheap and readily available starting materials such as trifluoroethylimidoyl chloride and propargylamine. It operates under mild conditions (30°C) with high reaction efficiency and excellent substrate compatibility, allowing for the synthesis of diverse derivatives.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method can be expanded to the gram level and provides the possibility for industrial large-scale production applications due to its simple operation and convenient post-processing steps.
Q: What types of substituents are tolerated in this reaction?
A: The reaction demonstrates broad functional group tolerance, successfully accommodating aryl groups with substituents such as methyl, tert-butyl, chlorine, bromine, trifluoromethyl, and nitro groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
As the demand for fluorinated heterocycles continues to surge in the development of next-generation therapeutics, having a manufacturing partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging advanced catalytic technologies like the palladium-catalyzed carbonylation described in CN111423381A to deliver high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from clinical trials to market launch. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-trifluoromethyl imidazole meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis capabilities can accelerate your drug development timeline while ensuring supply security.
