Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Derivatives for Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Derivatives for Pharmaceutical Applications
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective synthetic routes. A significant breakthrough in this domain is detailed in patent CN111423381B, which discloses a novel preparation method for 2-trifluoromethyl substituted imidazole compounds. These heterocyclic structures are pivotal in medicinal chemistry, serving as core scaffolds for numerous bioactive molecules, including antihistamines like Alcaftadine and various kinase inhibitors. The introduction of a trifluoromethyl group often enhances metabolic stability, lipophilicity, and bioavailability, making these compounds highly sought after by R&D teams globally. This new methodology leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing cheap and easily obtained starting materials to achieve high reaction efficiency.
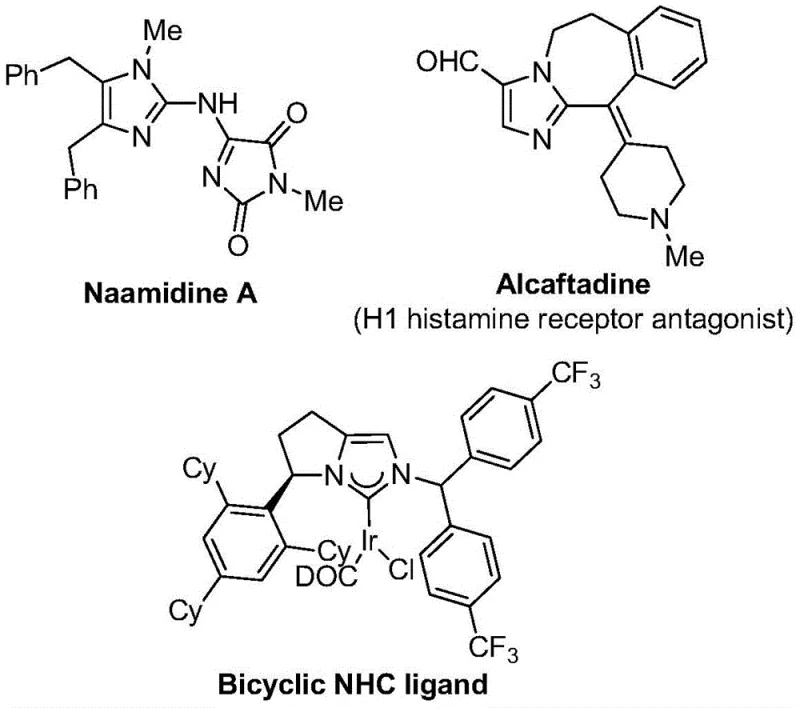
Traditionally, the synthesis of nitrogen-containing heterocycles functionalized with trifluoromethyl groups has relied on reacting synthons like trifluorodiazoethane with suitable substrates. However, these conventional approaches often suffer from significant limitations, including the use of hazardous reagents, harsh reaction conditions, and limited substrate scope. The instability and potential explosiveness of diazo compounds pose serious safety risks in large-scale manufacturing environments. Furthermore, many existing methods require elevated temperatures or strong bases that are incompatible with sensitive functional groups, leading to complex purification processes and reduced overall yields. These factors collectively increase the cost of goods sold (COGS) and extend the lead time for delivering high-purity pharmaceutical intermediates to clients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical reliance on direct trifluoromethylation using unstable synthons creates a bottleneck in the supply chain for complex heterocyclic APIs. Conventional routes frequently involve multi-step sequences with poor atom economy, generating substantial chemical waste that requires expensive disposal protocols. The incompatibility of these harsh conditions with diverse functional groups limits the chemical space that medicinal chemists can explore, forcing them to design around synthetic limitations rather than biological optimization. Additionally, the purification of products from these reactions often necessitates rigorous chromatographic separation to remove toxic byproducts and unreacted hazardous materials, further driving up operational costs and extending production timelines.
The Novel Approach
In stark contrast, the method described in patent CN111423381B offers a transformative solution by employing a mild, one-pot tandem reaction strategy. This innovative route utilizes trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts as key building blocks. The reaction proceeds smoothly at a remarkably low temperature of 30°C, significantly reducing energy consumption compared to traditional high-temperature reflux methods. By avoiding hazardous diazo reagents and utilizing stable, commercially available precursors, this process enhances operational safety and simplifies regulatory compliance. The broad substrate compatibility allows for the synthesis of diversified imidazole derivatives with various substituents at the 1 and 5 positions, providing medicinal chemists with unparalleled flexibility in drug design.
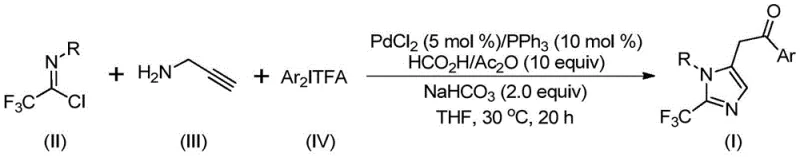
Mechanistic Insights into Palladium-Catalyzed Carbonylation and Cyclization
The success of this synthesis lies in its elegant catalytic cycle, which orchestrates multiple bond-forming events in a single operation. The mechanism initiates with the formation of a trifluoroacetamidine intermediate via an intermolecular carbon-nitrogen bond promoted by the base. Subsequently, the palladium catalyst, generated in situ from palladium chloride and triphenylphosphine, facilitates the activation of the alkyne moiety in propargylamine. This leads to the formation of an alkenyl palladium intermediate, which undergoes isomerization to a more stable alkyl palladium species. Crucially, the system employs formic acid and acetic anhydride as a safe and controllable source of carbon monoxide, enabling a carbonylation step that inserts a carbonyl group into the growing molecular framework.
Following carbonylation, the diaryl iodonium salt participates in an oxidative addition step, generating a high-valent tetravalent palladium intermediate. This step is pivotal for introducing the aryl group at the 5-position of the imidazole ring. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl-substituted imidazole product and regenerates the active palladium catalyst. This intricate dance of organometallic transformations ensures high regioselectivity and minimizes the formation of side products. The use of sodium bicarbonate as a mild additive helps neutralize acidic byproducts, maintaining the integrity of the catalyst and preventing decomposition of sensitive intermediates, thereby ensuring a clean impurity profile suitable for pharmaceutical applications.
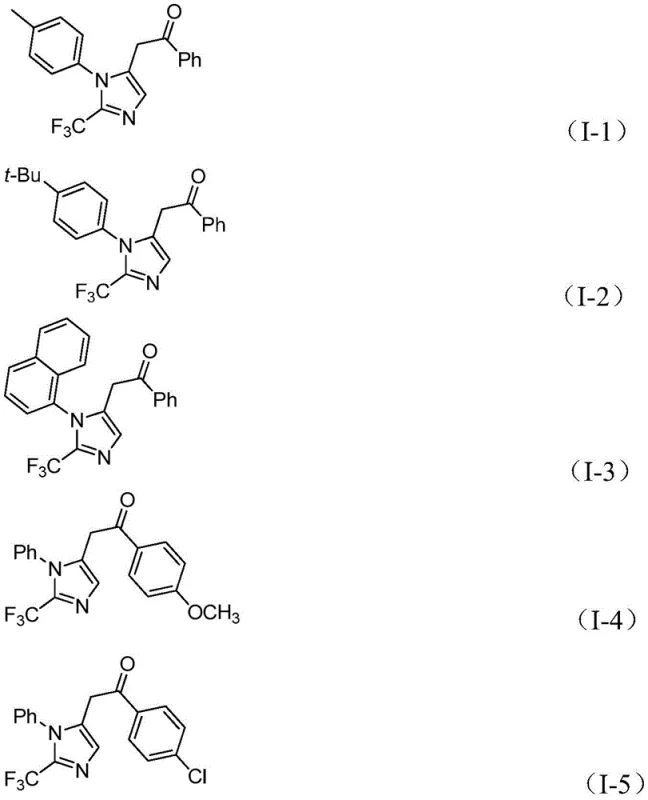
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this advanced synthetic route requires precise control over reaction parameters to maximize yield and purity. The process is designed to be robust, tolerating a wide range of electronic and steric variations in the starting materials. Whether utilizing electron-rich or electron-deficient aryl groups on the imidoyl chloride or the iodonium salt, the system maintains high efficiency. For R&D teams looking to replicate or scale this chemistry, the protocol involves a straightforward mixing of reagents in an aprotic solvent like tetrahydrofuran (THF), followed by a controlled reaction period. The simplicity of the workup procedure, involving filtration and standard column chromatography, makes this method highly attractive for rapid library synthesis and process development.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid, trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt in an organic solvent like THF.
- Stir the reaction mixture at a mild temperature of 30°C for 16 to 24 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers distinct strategic advantages that align with the goals of cost reduction and supply reliability. The shift towards milder reaction conditions and safer reagents directly translates to lower operational risks and reduced overhead costs associated with safety infrastructure. By eliminating the need for specialized equipment to handle explosive diazo compounds, manufacturers can utilize standard reactor setups, thereby increasing throughput capacity without significant capital expenditure. Furthermore, the high atom economy and simplified purification steps reduce the volume of solvent and silica gel required, contributing to a more sustainable and environmentally compliant manufacturing process.
- Cost Reduction in Manufacturing: The utilization of cheap and readily available starting materials, such as aromatic amines and propargylamine, significantly lowers the raw material costs compared to exotic fluorinated synthons. The mild reaction temperature of 30°C drastically reduces energy consumption for heating and cooling, leading to substantial utility savings over large production batches. Additionally, the high reaction efficiency and yields minimize the loss of valuable intermediates, optimizing the overall material balance and reducing the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against functional group variations ensures consistent quality across different batches, even when sourcing raw materials from different suppliers. The ability to synthesize diverse derivatives from a common set of precursors allows for agile response to changing market demands or clinical trial requirements. This flexibility reduces the risk of supply disruptions caused by the scarcity of specialized reagents, ensuring a continuous flow of high-quality intermediates to downstream customers.
- Scalability and Environmental Compliance: The process has been demonstrated to be expandable to the gram level with clear potential for industrial scale-up, addressing the critical need for commercial quantities during late-stage drug development. The avoidance of hazardous reagents and the generation of less toxic waste streams simplify waste treatment protocols, aligning with increasingly stringent global environmental regulations. This eco-friendly profile enhances the corporate social responsibility standing of the supply chain partners and facilitates smoother regulatory approvals for the final drug product.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its adoption. The following questions address common inquiries regarding the practical application and benefits of this technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable foundation for decision-making.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method utilizes cheap and readily available starting materials such as trifluoroethylimidoyl chloride and propargylamine. It operates under extremely mild conditions (30°C) compared to harsh traditional methods, offers excellent substrate compatibility, and achieves high reaction efficiency suitable for industrial scale-up.
Q: What is the role of the diaryl iodonium salt in this reaction?
A: The diaryl iodonium salt serves as the arylating agent and oxidant in the palladium catalytic cycle. It facilitates the oxidative addition step to form a tetravalent palladium intermediate, which is crucial for constructing the final imidazole skeleton with the desired aryl substitution.
Q: Is this process scalable for commercial production?
A: Yes, the patent explicitly states that the method can be expanded to the gram level and provides possibilities for industrial large-scale production. The simple post-treatment involving filtration and column chromatography further supports its feasibility for manufacturing high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned to full-scale manufacturing. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch of 2-trifluoromethyl imidazole derivatives we supply adheres to the highest industry standards.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge synthesis technology for their pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for efficiency, cost, and reliability in the competitive global market.
