Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Manufacturing
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that possess high biological activity and metabolic stability. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These nitrogen-containing fused ring systems are pivotal in medicinal chemistry, serving as core structures for numerous drug candidates exhibiting anti-cancer, anticonvulsant, and anti-inflammatory properties. The strategic introduction of a trifluoromethyl group into these heterocycles enhances electronegativity, lipophilicity, and bioavailability, making them highly desirable targets for modern drug discovery programs. This technical insight report analyzes the feasibility of this iron-catalyzed protocol for commercial adoption.
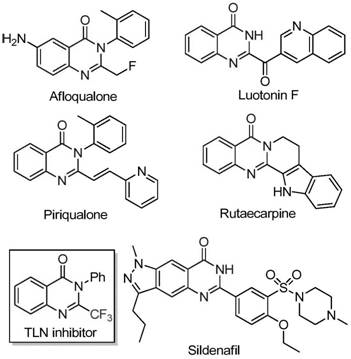
Traditional synthetic methodologies for constructing trifluoromethylated quinazolinones often rely on cyclization reactions involving expensive synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with anthranilamides or isatoic anhydrides. While effective in academic settings, these conventional approaches frequently suffer from severe reaction conditions, narrow substrate scope, and prohibitive costs associated with the fluorinating agents. Furthermore, the reliance on precious metal catalysts or harsh reagents complicates the purification process, often necessitating extensive downstream processing to remove toxic metal residues to meet regulatory standards for active pharmaceutical ingredients (APIs). These limitations create significant bottlenecks for procurement teams aiming to secure cost-effective and reliable supply chains for complex intermediates.
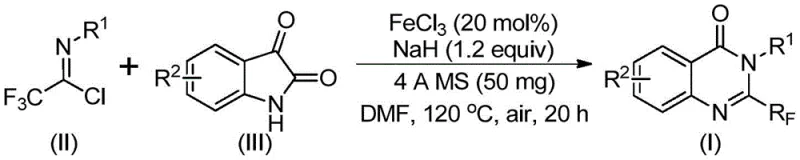
In stark contrast, the novel approach outlined in the patent utilizes readily available trifluoroethylimidoyl chloride and isatin derivatives as starting materials, catalyzed by inexpensive ferric chloride (FeCl3). This method operates under relatively mild conditions, initially at 40°C and subsequently heated to 120°C in an aprotic solvent like DMF, under an air atmosphere. The use of iron, an earth-abundant base metal, represents a paradigm shift towards greener and more sustainable chemistry. The reaction mechanism involves an alkali-promoted formation of carbon-nitrogen bonds followed by an iron-catalyzed decarbonylation and cyclization. This pathway not only simplifies the operational procedure but also drastically improves the economic viability of the synthesis by eliminating the need for exotic reagents.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this innovation lies in the efficient activation of the C-N bond formation and subsequent cyclization mediated by the iron catalyst. The reaction initiates with the interaction between the trifluoroethylimidoyl chloride and the isatin derivative in the presence of sodium hydride, which acts as a base to deprotonate the isatin nitrogen. This generates a nucleophilic species that attacks the electrophilic carbon of the imidoyl chloride. The ferric chloride then facilitates the intramolecular cyclization and the crucial decarbonylation step, leading to the aromatization of the quinazolinone ring system. The tolerance of the catalyst for various functional groups, including halogens (fluoro, bromo, chloro) and electron-donating groups (methyl, methoxy), ensures that a wide array of substituted quinazolinones can be accessed without protecting group strategies.
From an impurity control perspective, this mechanism offers distinct advantages. The use of 4A molecular sieves in the reaction mixture helps to sequester moisture, which is critical for maintaining the activity of the sodium hydride and preventing hydrolysis of the sensitive imidoyl chloride intermediate. The high conversion rates observed, with yields reaching up to 93% for certain substrates like the 5-fluoro derivative, indicate a clean reaction profile with minimal side-product formation. This high selectivity reduces the burden on downstream purification, allowing for simpler workup procedures involving filtration and standard column chromatography. For R&D directors, this implies a more predictable impurity profile, facilitating faster method validation and regulatory filing.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The synthesis protocol described in the patent provides a straightforward roadmap for laboratory and pilot-scale production. The process begins with the precise weighing of ferric chloride (20 mol%) and sodium hydride (1.2 equivalents), which are added to a reaction vessel along with 4A molecular sieves. The key reactants, trifluoroethylimidoyl chloride and the chosen isatin derivative, are then introduced into an organic solvent, preferably DMF, to ensure complete dissolution. The detailed standardized synthesis steps, including specific temperature ramps and reaction times optimized for maximum yield, are provided in the technical guide below.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in DMF solvent.
- Stir the reaction mixture at 40°C for 8-10 hours, then increase temperature to 120°C and react for 18-20 hours under air atmosphere.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl substituted quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iron-catalyzed methodology presents substantial opportunities for cost optimization and risk mitigation. The primary driver for cost reduction is the replacement of expensive noble metal catalysts and specialized fluorinating reagents with commodity chemicals. Ferric chloride is ubiquitous in the chemical industry and costs a fraction of palladium or rhodium catalysts. Additionally, the starting materials—isatin and aromatic amines used to prepare the imidoyl chloride—are bulk chemicals with stable global supply chains, reducing the risk of raw material shortages that often plague specialty syntheses.
- Cost Reduction in Manufacturing: The elimination of precious metals removes the need for expensive metal scavenging resins and complex analytical testing for residual heavy metals, which are mandatory for API release. This simplification of the downstream process translates directly into lower manufacturing overheads. Furthermore, the reaction proceeds in air, removing the capital expenditure and operational costs associated with maintaining strict inert gas (nitrogen or argon) atmospheres typically required for sensitive organometallic transformations.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the tolerance to air and moisture (managed by molecular sieves), makes the process highly transferable between different manufacturing sites. This flexibility allows for multi-sourcing strategies, ensuring continuity of supply even if one facility faces disruptions. The use of common solvents like DMF further enhances logistical ease, as these are standard items in any fine chemical inventory, avoiding the need for specialized solvent handling infrastructure.
- Scalability and Environmental Compliance: The patent explicitly notes the potential for industrial scale application, supported by the successful gram-level synthesis of diverse derivatives. From an environmental standpoint, the use of iron aligns with green chemistry principles, reducing the ecological footprint of the manufacturing process. This is increasingly important for meeting the sustainability goals of major pharmaceutical clients who are auditing their suppliers' environmental, social, and governance (ESG) performance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this technology for commercial production.
Q: What are the advantages of using FeCl3 over precious metal catalysts for quinazolinone synthesis?
A: Using ferric chloride (FeCl3) significantly reduces raw material costs compared to palladium or rhodium catalysts. It is earth-abundant, non-toxic, and simplifies the removal of heavy metal residues, which is critical for meeting stringent pharmaceutical purity specifications.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides possibilities for industrial scale application. The use of cheap, commercially available starting materials like isatin and the tolerance for air atmosphere facilitate large-scale manufacturing.
Q: What is the substrate scope for this trifluoromethylation reaction?
A: The method demonstrates good functional group tolerance. It works effectively with various substituted isatins (including halogens like F, Br, Cl and alkyl groups) and different aryl substituents on the imidoyl chloride, allowing for the design of diverse quinazolinone libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug development timelines. Our team of expert chemists has extensively evaluated the iron-catalyzed cyclization route described in CN111675662B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone meets the highest international standards.
We invite you to collaborate with us to leverage this cost-effective technology for your next drug candidate. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for custom synthesis projects, ensuring a seamless transition from bench-scale discovery to commercial supply.
