Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Global Pharmaceutical Manufacturing
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Global Pharmaceutical Manufacturing
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These heterocyclic scaffolds are ubiquitous in medicinal chemistry, serving as the core structure for numerous bioactive molecules exhibiting anti-cancer, anticonvulsant, anti-inflammatory, and antifungal properties. The strategic introduction of a trifluoromethyl group into these frameworks is particularly valuable, as it significantly enhances the electronegativity, lipophilicity, and metabolic stability of the final drug candidates. This patent presents a robust methodology that leverages inexpensive iron catalysis to construct these complex fused-ring systems from readily available starting materials, marking a substantial advancement over traditional synthetic approaches.
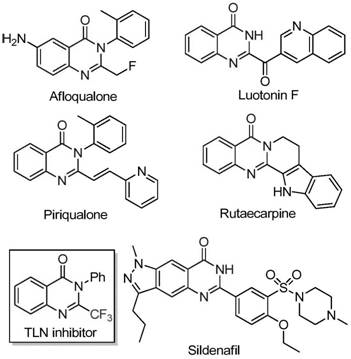
Quinazolinone derivatives represent a privileged class of nitrogen-containing fused ring heterocycles that are foundational to modern drug discovery. As illustrated by known bioactive molecules such as Afloqualone and Luotonin F, the quinazolinone core is versatile and potent. However, the conventional synthesis of trifluoromethyl-substituted variants has historically been plagued by significant bottlenecks. Traditional methods often rely on the cyclization of synthons like trifluoroacetic anhydride or ethyl trifluoroacetate with substrates such as anthranilamide or isatoic anhydride. While chemically feasible, these legacy routes are frequently characterized by severe reaction conditions, the requirement for expensive and specialized fluorinating reagents, and generally low yields that hinder industrial viability. Moreover, the narrow substrate scope of these older methods limits the ability of medicinal chemists to rapidly generate diverse libraries for structure-activity relationship (SAR) studies, creating a bottleneck in the early stages of drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical reliance on precious metal catalysts or harsh fluorinating agents has created a persistent barrier to the cost-effective manufacturing of high-purity quinazolinone intermediates. Conventional protocols often necessitate the use of stoichiometric amounts of expensive reagents, which not only drives up the bill of materials but also generates substantial chemical waste, complicating downstream processing and environmental compliance. For instance, methods utilizing trifluoroacetic anhydride often require strict anhydrous conditions and cryogenic temperatures to control exotherms, demanding specialized reactor infrastructure that increases capital expenditure. Furthermore, the purification of the final product from these reactions can be arduous, often requiring multiple recrystallizations or extensive chromatography to remove trace impurities and residual fluorine sources, thereby reducing the overall process mass intensity (PMI) and operational efficiency.
The Novel Approach
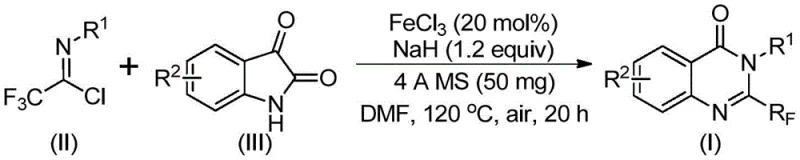
In stark contrast, the methodology outlined in patent CN111675662B introduces a paradigm shift by utilizing trifluoroethylimidoyl chloride and isatin derivatives as the primary building blocks. This novel approach capitalizes on the inherent reactivity of these substrates to form the quinazolinone core through a streamlined cascade sequence. By employing ferric chloride as a catalyst, the process bypasses the need for costly noble metals, aligning perfectly with the principles of green chemistry and economic manufacturing. The reaction demonstrates exceptional functional group tolerance, accommodating a wide array of substituents including alkyl, alkoxy, nitro, and various halogens on the aromatic rings without compromising yield. This versatility allows for the rapid synthesis of diverse analogues, such as the specific compound (I-4) shown below, facilitating faster lead optimization cycles for pharmaceutical partners.

Mechanistic Insights into FeCl3-Catalyzed Cyclization
The success of this synthetic route lies in the intricate interplay between the base-promoted nucleophilic attack and the subsequent iron-catalyzed cyclization. The reaction initiates with the formation of a carbon-nitrogen bond between the trifluoroethylimidoyl chloride and the isatin substrate, facilitated by sodium hydride. This initial step generates a trifluoroacetamidine intermediate, which serves as the precursor for the ring-closing event. The presence of ferric chloride is critical at this stage, acting as a Lewis acid to activate the carbonyl functionality and promote the decarbonylation and cyclization steps necessary to aromatize the system. This mechanistic pathway is highly efficient, avoiding the formation of stable byproducts that typically plague similar condensation reactions, thus ensuring a cleaner reaction profile and higher crude purity.
Furthermore, the mechanistic robustness of this iron-catalyzed system provides significant advantages regarding impurity control, a key concern for R&D directors overseeing process development. The use of 4A molecular sieves in the reaction mixture plays a dual role: it acts as a desiccant to sequester water generated during the reaction, preventing hydrolysis of the sensitive imidoyl chloride starting material, and it helps to drive the equilibrium towards the desired product. The tolerance of the catalytic system to air atmosphere, as opposed to requiring rigorous inert gas blanketing, suggests that the active iron species are relatively stable and resistant to oxidation under the reaction conditions. This stability minimizes the formation of oxidative degradation products, simplifying the impurity profile and reducing the burden on analytical quality control teams during batch release testing.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The operational simplicity of this patented process makes it highly attractive for technology transfer from laboratory bench to pilot plant. The procedure involves a straightforward one-pot sequence where reagents are combined in a polar aprotic solvent such as DMF, heated in a two-stage temperature profile, and worked up via standard filtration and chromatography techniques. This eliminates the need for intermediate isolation or hazardous reagent handling, streamlining the workflow for process chemists. The detailed standardized synthesis steps, including precise molar ratios and temperature ramping protocols, are essential for ensuring reproducibility and safety during scale-up operations.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in DMF solvent within a reaction vessel.
- Initiate the reaction by stirring at 40°C for approximately 10 hours to facilitate initial bond formation, then increase temperature to 120°C.
- Maintain heating at 120°C for an additional 20 hours under air atmosphere to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iron-catalyzed methodology represents a strategic opportunity to optimize the cost structure and reliability of the supply chain for quinazolinone-based APIs. The substitution of expensive precious metal catalysts with commodity-grade ferric chloride results in a drastic reduction in raw material costs, directly impacting the bottom line. Additionally, the use of widely available starting materials like isatin and aromatic amines ensures that the supply chain is not vulnerable to the geopolitical or logistical bottlenecks often associated with specialized fluorinating reagents. The robustness of the reaction conditions further enhances supply continuity by reducing the risk of batch failures due to minor deviations in process parameters.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of inexpensive iron salts significantly lowers the direct material costs associated with production. Moreover, the simplified workup procedure, which avoids complex extraction or heavy metal scavenging steps, reduces solvent consumption and waste disposal fees, leading to substantial overall cost savings in the manufacturing process.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals such as ferric chloride and sodium hydride, manufacturers can mitigate the risks associated with single-source suppliers of exotic reagents. The broad availability of these inputs ensures that production schedules can be maintained even during periods of global supply chain disruption, providing a secure and consistent flow of high-quality intermediates to downstream customers.
- Scalability and Environmental Compliance: The reaction's tolerance to air and its operation in common solvents like DMF facilitate easy scale-up from gram to multi-kilogram quantities without requiring specialized pressure vessels or cryogenic equipment. This scalability, combined with the generation of less hazardous waste compared to traditional fluorination methods, supports stricter environmental compliance standards and reduces the regulatory burden on manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders evaluating this technology for their pipeline.
Q: What are the primary advantages of using ferric chloride over precious metal catalysts for quinazolinone synthesis?
A: Ferric chloride is significantly cheaper and more abundant than palladium or rhodium catalysts, drastically reducing raw material costs. Furthermore, iron catalysts often exhibit excellent functional group tolerance and eliminate the need for complex heavy metal removal steps required for pharmaceutical grade APIs.
Q: Can this synthesis method accommodate diverse substituents on the isatin ring?
A: Yes, the patented method demonstrates broad substrate scope, successfully tolerating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens (F, Cl, Br) and nitro groups at various positions on the aromatic ring.
Q: Is the reaction sensitive to moisture or oxygen during the scale-up process?
A: The protocol utilizes 4A molecular sieves to manage moisture effectively and operates under air atmosphere rather than requiring strict inert gas protection, which simplifies the engineering controls needed for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in accelerating drug development timelines. Our team of expert process chemists has extensively evaluated the iron-catalyzed cyclization method described in patent CN111675662B and confirmed its potential for robust commercial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trials to market launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone intermediate meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced technology for your next pharmaceutical project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that optimize both the quality and economics of your supply chain.
