Advanced Synthesis of Quinoline Derivatives for Scalable Erlotinib Intermediate Production
Advanced Synthesis of Quinoline Derivatives for Scalable Erlotinib Intermediate Production
The pharmaceutical industry's relentless pursuit of more efficient oncology treatments has placed significant emphasis on the optimization of kinase inhibitor supply chains. Patent CN112469711A introduces a groundbreaking synthetic methodology for preparing 4-(4-fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxy-7-hydroxyquinoline derivatives, which serve as critical precursors for Erlotinib. This technical disclosure addresses long-standing challenges in heterocyclic chemistry, specifically targeting the elimination of cumbersome purification steps and the mitigation of persistent impurity profiles. By leveraging a strategic reduction-cyclization sequence, the inventors have established a pathway that not only enhances chemical purity to levels exceeding 99% but also drastically streamlines the operational workflow. For global procurement leaders and R&D directors, this represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms for high-value tyrosine kinase inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex quinoline-indole hybrids has been plagued by multi-step sequences that suffer from low overall yields and significant impurity burdens. Prior art, such as the methodology disclosed in CN102159078A, typically relies on a four-step reaction sequence starting from 7-benzyloxy-4-chloro-6-methoxyquinoline. A critical bottleneck in these traditional routes is the generation of hydroxylamine impurities during the penultimate reaction stages. These nitrogen-oxygen species are notoriously difficult to separate from the desired product, often necessitating resource-intensive column chromatography purification. Furthermore, the harsh conditions sometimes required in older protocols can lead to the degradation of sensitive functional groups, resulting in a broader impurity spectrum that complicates regulatory compliance and increases the cost of goods sold due to material loss.
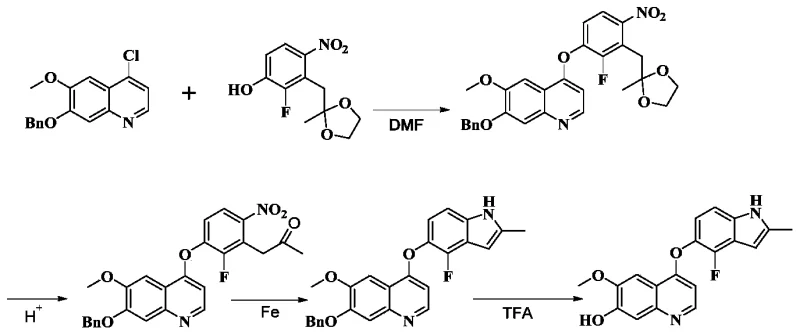
The Novel Approach
In stark contrast, the novel approach detailed in CN112469711A circumvents these inefficiencies through a clever manipulation of oxidation states and protecting group strategies. The core innovation lies in the conversion of a nitro-substituted precursor directly into the amino-intermediate, followed by an in situ or sequential cyclization to form the indole ring. This strategy effectively bypasses the formation of stable hydroxylamine by-products that plague earlier methods. By utilizing a nitro-reduction pathway, the synthesis achieves a higher degree of atom economy and operational simplicity. The process is designed to be telescoped or performed in minimal steps, thereby reducing solvent consumption and processing time. This streamlined architecture not only improves the final yield but also ensures that the crude product possesses a purity profile amenable to simple crystallization rather than chromatographic separation.
Mechanistic Insights into Catalytic Reduction and Cyclization
The mechanistic elegance of this synthesis is rooted in the selective reduction of the aromatic nitro group in the presence of other potentially reducible functionalities. The patent outlines the use of catalytic transfer hydrogenation, employing palladium on carbon (Pd/C) alongside ammonium formate as a hydrogen donor. This system operates under mild thermal conditions, typically between 30°C and 40°C, which preserves the integrity of the quinoline core and the ether linkages. The reduction proceeds via the formation of surface-bound hydrogen species on the palladium catalyst, which sequentially reduce the nitro group (-NO2) to the amine (-NH2) without accumulating the hydroxylamine (-NHOH) intermediate to any significant extent. This kinetic control is vital for maintaining high purity, as the rapid conversion prevents the side reactions associated with partially reduced nitrogen species.
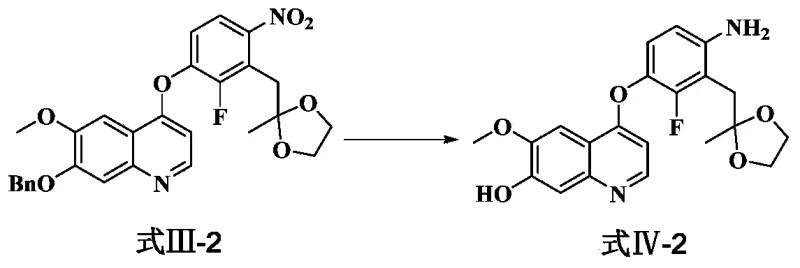
Following the reduction, the subsequent cyclization step leverages the nucleophilicity of the newly formed aniline nitrogen to attack an adjacent electrophilic center, facilitated by acid catalysis. The patent specifies the use of hydrochloric acid in methanol at elevated temperatures (60-70°C) to effect this transformation. This acidic environment serves a dual purpose: it catalyzes the ring closure to form the indole moiety and simultaneously cleaves acid-labile protecting groups, such as ketals or benzyl ethers, depending on the specific substrate variant. This tandem deprotection-cyclization event is a masterstroke of process chemistry, as it consolidates two distinct chemical transformations into a single operational unit. The result is a robust process that minimizes handling losses and ensures that the final product, the hydroxyquinoline derivative, is obtained with exceptional purity, often reported above 99.5% in the provided examples.
How to Synthesize 4-(4-fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxy-7-hydroxyquinoline Efficiently
The practical execution of this synthesis requires precise control over reaction parameters to maximize the benefits of the novel pathway. The process begins with the coupling of the quinoline chloride and the nitrophenol derivative, followed by the critical reduction and cyclization steps described in the mechanistic section. Operators must adhere to strict stoichiometric ratios, particularly regarding the reducing agent and the acid catalyst, to ensure complete conversion. The detailed standardized synthesis steps, including specific solvent volumes, temperature ramps, and workup procedures like filtration and crystallization, are outlined in the comprehensive guide below to ensure reproducibility and safety in a GMP environment.
- Perform nucleophilic substitution between 7-benzyloxy-4-chloro-6-methoxyquinoline and a nitrophenol derivative using an organic base at elevated temperatures (135°C) to form the nitro-intermediate.
- Execute catalytic transfer hydrogenation using palladium on carbon and ammonium formate to reduce the nitro group to an amino group while simultaneously removing protecting groups.
- Conduct acid-catalyzed cyclization and deprotection using hydrochloric acid in methanol to finalize the indole ring formation and yield the target hydroxyquinoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented synthesis route offers profound economic and logistical advantages. The primary value driver is the complete elimination of column chromatography, a unit operation that is notoriously expensive, slow, and difficult to scale in a commercial setting. By replacing chromatography with crystallization and filtration, the process drastically reduces solvent usage, silica gel waste, and processing time. This transition from batch chromatographic purification to continuous or semi-continuous crystallization significantly lowers the variable costs associated with manufacturing. Furthermore, the high purity achieved directly from the reactor reduces the need for re-processing or recycling of off-spec material, thereby enhancing overall material throughput and yield efficiency.
- Cost Reduction in Manufacturing: The removal of column chromatography translates directly into substantial cost savings by eliminating the need for expensive stationary phases and large volumes of high-purity solvents. Additionally, the use of readily available catalysts like palladium on carbon and common reagents like ammonium formate ensures that raw material costs remain stable and predictable. The simplified workflow reduces labor hours and equipment occupancy time, allowing for higher throughput in existing manufacturing facilities without the need for capital-intensive new infrastructure.
- Enhanced Supply Chain Reliability: The robustness of this chemical route enhances supply chain resilience by reducing the number of critical process steps where failures can occur. Fewer steps mean fewer opportunities for deviation, leading to more consistent batch-to-batch quality and reliable delivery schedules. The use of standard chemical reagents that are globally sourced mitigates the risk of supply disruptions associated with specialized or exotic reagents. This reliability is crucial for maintaining the continuity of API production for life-saving oncology medications like Erlotinib.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns with green chemistry principles by minimizing waste generation and solvent intensity. The avoidance of chromatographic silica waste and the reduction in solvent volume lower the environmental footprint of the manufacturing process, facilitating easier compliance with increasingly stringent environmental regulations. The scalability of the reaction conditions, which utilize standard glass-lined or stainless steel reactors, ensures that the process can be seamlessly transferred from pilot scale to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within patent CN112469711A, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new synthesis method improve upon previous erlotinib intermediate routes?
A: Unlike prior art methods such as CN102159078A which require four steps and generate difficult-to-remove hydroxylamine impurities, this patented process utilizes a direct reduction-cyclization strategy. This eliminates the need for column chromatography, significantly simplifying purification and achieving purities exceeding 99%.
Q: What specific catalysts and conditions are used for the nitro reduction step?
A: The process employs catalytic transfer hydrogenation using 10% palladium on carbon (Pd/C) with ammonium formate as the hydrogen donor in solvents like dichloromethane or methanol. This mild condition (30-40°C) ensures selective reduction without affecting other sensitive functional groups on the quinoline scaffold.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the method is explicitly designed for industrial scalability. By avoiding column chromatography and utilizing robust reagents like iron powder or Pd/C which are easily filtered, the process reduces operational complexity and waste generation, making it ideal for commercial-scale API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxy-7-hydroxyquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and production of next-generation oncology therapeutics. Our team of expert process chemists has extensively analyzed the methodology presented in CN112469711A and possesses the technical capability to implement this advanced synthesis route at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of quinoline derivative meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this innovative technology for your Erlotinib supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
