Advanced Silver-Catalyzed Isoquinoline Synthesis for Commercial Scale Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries continuously demand efficient pathways to access bioactive heterocyclic scaffolds, among which the isoquinoline skeleton stands out due to its profound physiological activities. Patent CN109678798B, published in early 2023, introduces a robust synthetic methodology for constructing these valuable isoquinoline compounds through a tandem reaction sequence. This innovation addresses the critical need for streamlined manufacturing processes that can support the rigorous purity standards required for active pharmaceutical ingredients and high-value intermediates. By leveraging a silver-catalyzed system, the disclosed method facilitates the transformation of readily available o-alkynyl aldehydes and p-hydroxybenzylamines into complex target structures with remarkable efficiency. For R&D directors and procurement specialists, understanding the technical nuances of this patent is essential for evaluating its potential integration into existing supply chains and production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of isoquinoline derivatives has relied heavily on extraction from natural plant sources or multi-step total synthesis routes that often suffer from significant inefficiencies. Traditional extraction methods are inherently limited by the seasonal availability of raw botanical materials, leading to unpredictable supply chain disruptions and fluctuating costs that complicate long-term procurement planning. Furthermore, natural extracts often contain a complex matrix of impurities that require extensive and costly downstream purification processes to meet the stringent regulatory specifications for pharmaceutical use. On the synthetic side, classical cyclization strategies frequently necessitate the use of harsh reaction conditions, toxic reagents, or expensive transition metal catalysts that pose environmental and safety challenges in a commercial manufacturing setting. These legacy methods often result in suboptimal yields and generate substantial chemical waste, thereby increasing the overall cost of goods sold and environmental compliance burdens for production facilities.
The Novel Approach
In contrast, the methodology disclosed in patent CN109678798B presents a streamlined tandem reaction protocol that significantly simplifies the construction of the isoquinoline core. This novel approach utilizes a silver nitrate catalyst in an ethanol solvent system, operating under mild thermal conditions that are far more compatible with large-scale industrial equipment. The reaction proceeds through an elegant imidization, cyclization, and oxidative elimination sequence, effectively merging multiple synthetic transformations into a single operational step. This consolidation of steps not only reduces the total processing time but also minimizes the handling of intermediate materials, thereby lowering the risk of material loss and contamination. The use of ethanol as a solvent further enhances the green chemistry profile of the process, aligning with modern sustainability goals while reducing solvent recovery costs. For supply chain managers, this translates to a more reliable and cost-effective manufacturing route that mitigates the risks associated with complex multi-step syntheses.
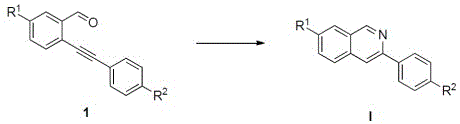
Mechanistic Insights into AgNO3-Catalyzed Cyclization
The core of this technological advancement lies in the specific mechanistic pathway facilitated by the silver catalyst, which activates the alkyne moiety for nucleophilic attack by the amine source. The reaction initiates with the formation of an imine intermediate between the aldehyde group of the o-alkynyl aldehyde and the amino group of the p-hydroxybenzylamine. Subsequently, the silver species coordinates with the alkyne triple bond, increasing its electrophilicity and promoting an intramolecular cyclization event that constructs the heterocyclic ring system. This is followed by an oxidative elimination step that restores aromaticity and delivers the final isoquinoline product with high regioselectivity. The p-hydroxybenzylamine serves a dual role in this process, acting not only as the nitrogen source but also facilitating the oxidative elimination through its inherent electronic properties. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters such as catalyst loading and temperature to maximize throughput and minimize byproduct formation in a commercial reactor.
Furthermore, the versatility of this synthetic route is demonstrated by its tolerance to a wide array of functional groups on both the aldehyde and amine components, ensuring broad applicability across different drug discovery programs. The patent data indicates that substituents such as halogens, alkyl groups, and alkoxy groups at various positions on the aromatic rings do not significantly impede the reaction progress. This functional group tolerance is a critical attribute for R&D teams, as it allows for the rapid generation of diverse analog libraries for structure-activity relationship studies without the need for extensive protecting group strategies. The ability to introduce diverse substituents like chlorine, fluorine, or methyl groups at the R1 and R2 positions enables the fine-tuning of the physicochemical properties of the final drug candidate. Consequently, this method supports the efficient exploration of chemical space, accelerating the identification of lead compounds with optimal pharmacokinetic profiles.
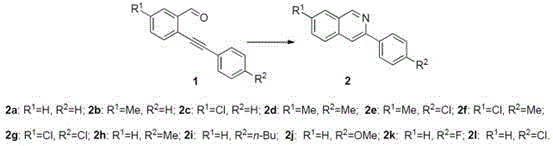
How to Synthesize Isoquinoline Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters outlined in the patent examples to ensure consistent quality and yield. The process begins with the precise weighing of o-alkynyl aldehyde, p-hydroxybenzylamine, and the silver nitrate catalyst, maintaining the specified molar ratios to drive the reaction to completion. The mixture is then dissolved in ethanol and heated to the optimal temperature range, with continuous monitoring via thin-layer chromatography to determine the exact endpoint of the reaction. Once the starting materials are fully consumed, the reaction mixture is cooled, and the solvent is removed under reduced pressure to isolate the crude product. The final purification is achieved through column chromatography, which separates the target isoquinoline from any minor byproducts or residual catalyst, ensuring the high purity required for downstream applications.
- Dissolve o-alkynyl aldehyde, p-hydroxybenzylamine, and silver nitrate catalyst in ethanol solvent with a molar ratio of 1: 1-5:0.1-3.
- Stir the reaction mixture at a temperature range of 60-100 degrees Celsius while monitoring progress via TLC until completion.
- Cool the reactant to room temperature, remove solvent by rotary evaporation, and purify the residue via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this silver-catalyzed synthesis route offers substantial strategic advantages for procurement managers and supply chain directors focused on cost optimization and reliability. The use of ethanol as a primary solvent eliminates the need for expensive, hazardous, or difficult-to-recycle organic solvents, thereby significantly reducing waste disposal costs and environmental compliance overhead. Additionally, the starting materials, specifically o-alkynyl aldehydes and p-hydroxybenzylamines, are generally accessible through established chemical supply networks, ensuring a stable raw material base that is less susceptible to geopolitical or agricultural supply shocks. The simplified one-pot nature of the reaction reduces the requirement for intermediate isolation and purification steps, which directly translates to lower labor costs and reduced equipment occupancy time in the manufacturing facility. These factors collectively contribute to a more lean and agile production model that can respond quickly to market demands.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and the use of a recoverable silver catalyst system lead to a drastic simplification of the production workflow. By consolidating imidization, cyclization, and oxidation into a single vessel operation, the process minimizes material transfer losses and reduces the consumption of auxiliary reagents. This streamlined approach inherently lowers the operational expenditure associated with heating, cooling, and agitation over extended periods, resulting in substantial cost savings per kilogram of product. Furthermore, the high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted as byproducts, which further enhances the overall cost-effectiveness of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials and a robust catalytic system ensures a high degree of supply continuity for critical pharmaceutical intermediates. Unlike natural extraction methods that are vulnerable to crop failures or seasonal variations, this synthetic route can be executed year-round in controlled industrial environments. The scalability of the reaction from milligram to kilogram scales without significant loss in efficiency provides procurement teams with the confidence to secure long-term supply agreements. This reliability is paramount for maintaining uninterrupted production schedules for downstream drug products, mitigating the risk of stockouts that could impact patient access and revenue.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reaction conditions that are easily transferable from laboratory glassware to large-scale stainless steel reactors. The use of ethanol, a green solvent, aligns with increasingly stringent environmental regulations regarding volatile organic compound emissions and waste generation. The reduced complexity of the workup procedure also minimizes the volume of chemical waste generated, simplifying the waste treatment process and lowering the environmental footprint of the manufacturing site. This compliance with green chemistry principles not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isoquinoline synthesis technology in industrial settings. These answers are derived directly from the experimental data and technical disclosures within patent CN109678798B to provide accurate guidance for decision-makers. Understanding these details helps stakeholders assess the feasibility of adopting this route for their specific product pipelines and supply chain requirements. The information covers critical aspects such as reaction optimization, substrate compatibility, and comparative advantages over legacy methods.
Q: What are the optimal reaction conditions for this isoquinoline synthesis?
A: The patent specifies using ethanol as the solvent with silver nitrate as the catalyst at temperatures between 60-100°C, preferably 80°C, achieving yields up to 83%.
Q: What is the scope of substituents allowed on the isoquinoline ring?
A: The method tolerates various groups including hydrogen, halogens (Cl, F), alkyl (Me, n-Bu), and alkoxy (OMe) at both R1 and R2 positions.
Q: How does this method compare to traditional extraction from natural products?
A: This synthetic route offers a more controlled and scalable alternative to natural extraction, providing consistent purity and avoiding supply fluctuations associated with biological sources.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes like the one described in CN109678798B for the production of high-quality pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of isoquinoline compound delivered adheres to the highest industry standards. We understand the complexities involved in transitioning a patent methodology into a commercially viable process and are dedicated to supporting our clients through every stage of development and manufacturing.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this silver-catalyzed method can optimize your supply chain and reduce overall production costs. Let us help you secure a reliable source of high-purity isoquinoline intermediates that drive your drug development programs forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
