Advanced Heterogeneous Catalysis for Scalable Production of High-Purity Pharmaceutical Intermediates
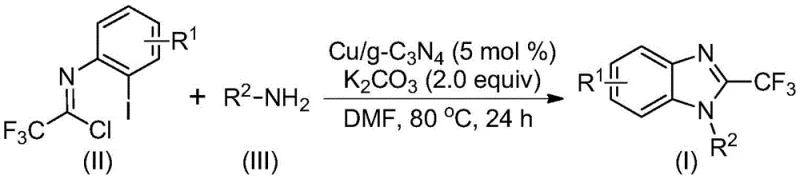
The Chinese patent CN113429349A introduces a transformative heterogeneous catalytic methodology for synthesizing structurally diverse 2-trifluoromethyl substituted benzimidazole compounds that serve as critical pharmacophores in modern drug discovery pipelines due to their enhanced bioavailability and metabolic stability imparted by strategic fluorine substitution. This innovative process directly addresses persistent industry challenges associated with conventional synthetic approaches that typically require harsh reaction conditions exceeding safe operational thresholds or involve expensive transition metal catalysts necessitating complex purification protocols. By implementing copper-doped carbon nitride as a recyclable heterogeneous catalyst system operating under mild thermal parameters between 70–90°C in dimethylformamide solvent medium, the method achieves exceptional reaction efficiency while maintaining operational simplicity through straightforward workup procedures involving filtration and chromatography. The catalyst's demonstrated recyclability through three consecutive cycles without significant activity loss represents a substantial advancement in sustainable manufacturing practices that directly impacts both environmental footprint reduction and production economics. This patent establishes a robust foundation for producing high-purity pharmaceutical intermediates meeting stringent regulatory requirements while offering significant advantages in supply chain reliability through simplified raw material sourcing from commercially available precursors. The strategic implementation of this technology enables manufacturers to overcome historical barriers in producing fluorinated heterocycles essential for next-generation therapeutic agents targeting oncology and central nervous system disorders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethyl benzimidazoles have historically been constrained by multiple critical limitations that impede their adoption in commercial manufacturing environments including severe reaction conditions requiring elevated temperatures above safe operational limits or cryogenic environments that increase energy consumption and safety risks. These methods frequently employ homogeneous copper catalysts that necessitate complex separation procedures involving multiple extraction steps and specialized equipment to remove trace metal residues below regulatory thresholds of one part per million. The narrow substrate scope observed in prior art approaches restricts structural diversity by failing to accommodate various functional groups commonly found in pharmaceutical intermediates such as halogens or alkyl chains beyond simple methyl groups. Additionally, conventional processes often utilize expensive or difficult-to-source starting materials like trifluoroacetic anhydride that require specialized handling protocols due to their corrosive nature and moisture sensitivity. The absence of catalyst recyclability in homogeneous systems creates significant economic burdens through continuous catalyst consumption while generating substantial waste streams requiring costly disposal procedures that conflict with modern green chemistry principles. These combined limitations have historically prevented pharmaceutical manufacturers from achieving both cost-effective production scales and consistent product quality required for regulatory approval processes.
The Novel Approach
The patented methodology overcomes these limitations through an elegant heterogeneous catalytic system utilizing copper-doped carbon nitride that operates under significantly milder conditions within the optimal temperature range of 70–90°C while maintaining high conversion efficiency across diverse substrate combinations. This innovation eliminates the need for complex metal removal protocols by enabling straightforward catalyst separation through simple filtration techniques that preserve product integrity without introducing additional purification steps. The strategic use of commercially available trifluoroethylimidoyl chloride and amines as starting materials provides exceptional flexibility in designing structurally diverse benzimidazole derivatives with varying functional groups including halogens and alkyl chains while maintaining consistent high yields across different substrate combinations. The process demonstrates remarkable operational simplicity through its one-pot reaction design that integrates nucleophilic addition and cyclization steps without requiring intermediate isolation or specialized equipment. Crucially, the heterogeneous catalyst maintains substantial activity through three reuse cycles as validated by experimental data showing only partial efficiency reduction after multiple applications which directly translates to reduced raw material consumption and lower environmental impact compared to single-use homogeneous systems. This approach represents a paradigm shift toward sustainable manufacturing practices that align with industry demands for both economic viability and regulatory compliance in producing high-value pharmaceutical intermediates.
Mechanistic Insights into Cu/g-C₃N₄-Catalyzed Cyclization
The catalytic mechanism begins with nucleophilic addition of amine to trifluoroethylimidoyl chloride forming an amidine intermediate followed by oxidative addition where copper inserts into the carbon-halogen bond creating a key organocopper species. Subsequent base-mediated dehydrohalogenation generates a bivalent copper complex that undergoes intramolecular cyclization through nitrogen coordination to form a trivalent copper intermediate essential for ring closure. The final reductive elimination step releases the target benzimidazole product while regenerating the active copper species completing the catalytic cycle without requiring additional oxidants or reductants. This mechanism operates efficiently due to the unique electronic properties of copper-doped carbon nitride which provides optimal Lewis acidity for substrate activation while maintaining structural stability under reaction conditions through its graphitic framework that prevents copper leaching during multiple reuse cycles. The heterogeneous nature of this catalytic system ensures precise control over reaction pathways by minimizing undesired side reactions commonly observed in homogeneous systems where free copper ions can promote decomposition pathways.
Impurity control is achieved through multiple synergistic mechanisms inherent to this heterogeneous system including precise temperature regulation within the optimal range of 70–90°C that prevents thermal decomposition pathways observed at higher temperatures in conventional methods. The selective activation of substrates by copper-doped carbon nitride minimizes competing reactions such as hydrolysis or dimerization that typically generate impurities requiring extensive purification efforts. The absence of strong acids or bases eliminates common side reactions like dehalogenation or over-reduction that compromise product purity in alternative synthetic routes. Furthermore, the recyclable nature of the catalyst prevents accumulation of metal impurities that would otherwise require additional purification steps beyond standard chromatography protocols. This integrated approach ensures consistent production of high-purity intermediates meeting pharmaceutical quality standards without necessitating specialized analytical monitoring beyond routine quality control procedures.
How to Synthesize Trifluoromethyl Benzimidazoles Efficiently
This patented methodology provides a robust framework for manufacturing high-purity trifluoromethyl benzimidazole intermediates through a carefully optimized sequence that balances reaction efficiency with operational practicality for commercial scale-up environments. The process leverages readily available starting materials including copper-doped carbon nitride catalyst synthesized from economical precursors such as copper sulfate pentahydrate and carbon nitride through established procedures described in the patent documentation. Detailed standardized synthesis steps are provided below to ensure consistent implementation across different production scales while maintaining stringent quality control parameters required for pharmaceutical applications.
- Combine copper-doped carbon nitride catalyst (5 mol%), potassium carbonate (2.0 equiv), trifluoroethylimidoyl chloride (II), and amine (III) in DMF solvent under nitrogen atmosphere with precise stoichiometric ratios.
- Heat the reaction mixture to controlled temperature range of 70–90°C while maintaining vigorous stirring for optimal reaction duration between 18–30 hours to ensure complete cyclization.
- Execute post-treatment protocol involving catalyst filtration through celite bed followed by silica gel mixing and column chromatography purification to isolate high-purity product meeting pharmaceutical standards.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route delivers substantial value across procurement and supply chain functions by addressing critical pain points associated with traditional manufacturing approaches for fluorinated heterocyclic intermediates essential in pharmaceutical production pipelines. The strategic implementation of this technology directly responds to industry demands for more resilient supply chains capable of delivering consistent quality while navigating volatile raw material markets through its reliance on commercially available starting materials with multiple sourcing options.
- Cost Reduction in Manufacturing: The elimination of expensive homogeneous catalysts requiring complex removal protocols significantly reduces raw material expenses while minimizing waste treatment costs associated with metal-contaminated streams; additionally, the simplified workup procedure using standard filtration techniques instead of specialized extraction equipment lowers capital investment requirements and operational complexity across production facilities.
- Enhanced Supply Chain Reliability: The use of readily available starting materials including potassium carbonate and common amines from multiple global suppliers mitigates single-source dependency risks while enabling flexible production scheduling; furthermore, the robustness of the process across different batch sizes ensures consistent output quality regardless of scale transitions from development to commercial manufacturing volumes.
- Scalability and Environmental Compliance: The straightforward scale-up pathway from laboratory to commercial production is facilitated by the absence of hazardous reagents or extreme process conditions requiring specialized containment systems; additionally, the recyclable catalyst system reduces environmental impact through decreased waste generation streams while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement specialists and supply chain managers regarding implementation of this patented technology for manufacturing pharmaceutical intermediates; answers are derived directly from experimental data and process descriptions documented in Chinese patent CN113429349A.
Q: How does heterogeneous catalysis improve scalability versus homogeneous methods?
A: The copper-doped carbon nitride catalyst enables straightforward separation through simple filtration without complex extraction procedures required for homogeneous systems, allowing seamless transition from laboratory-scale reactions to commercial production volumes while maintaining consistent product quality through multiple reuse cycles.
Q: What substrate functional groups demonstrate compatibility with this synthetic route?
A: The method exhibits exceptional functional group tolerance across diverse substrates including alkyl chains (n-butyl to n-hexyl), aryl groups (phenyl, benzyl), halogens (Cl, Br), and trifluoromethyl substituents on both imidoyl chloride and amine components without requiring protective groups.
Q: How does catalyst recyclability translate to commercial cost advantages?
A: With demonstrated retention of catalytic activity through three reuse cycles while maintaining high conversion rates, the system eliminates continuous catalyst replenishment costs and reduces waste generation streams associated with homogeneous catalyst disposal protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Benzimidazole Supplier
Our company leverages this patented technology to deliver exceptional value through extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities; our rigorous QC labs implement comprehensive analytical protocols ensuring consistent product quality meeting all pharmacopeial standards for critical intermediates used in therapeutic agent manufacturing. NINGBO INNO PHARMCHEM's specialized expertise in heterogeneous catalysis processes enables seamless technology transfer from laboratory development to full-scale commercial production without compromising on quality or yield consistency across different batch sizes.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team which will provide detailed insights into potential efficiency gains specific to your manufacturing requirements; please contact us directly to obtain specific COA data and route feasibility assessments tailored to your production needs.
