Scalable Production of 2-Trifluoromethyl Benzimidazoles via Heterogeneous Copper Catalysis
Introduction to Advanced Heterogeneous Catalysis in Benzimidazole Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN113429349A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted benzimidazole compounds utilizing a heterogeneous copper-doped carbon nitride catalyst. This technology represents a significant leap forward from traditional homogeneous catalysis, addressing critical pain points regarding catalyst recovery and product purity. By leveraging a cascade cyclization reaction between trifluoroacetimidoyl chloride and various amines, this process achieves high efficiency under mild conditions. For R&D directors and procurement specialists, this patent outlines a pathway to reliable pharmaceutical intermediate supplier status by ensuring consistent quality and reduced environmental footprint through recyclable catalytic systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted benzimidazoles has relied on methods that present substantial challenges for large-scale manufacturing. Traditional routes often involve the condensation of o-phenylenediamine with trifluoroacetic acid derivatives or reductive cyclization of o-nitroanilines, which frequently suffer from severe reaction conditions and narrow substrate scopes. Furthermore, earlier copper-catalyzed approaches typically employed homogeneous catalysts, creating a significant bottleneck in downstream processing. The removal of residual copper from the final active pharmaceutical ingredient (API) intermediate requires expensive and time-consuming purification steps, such as specialized scavenging resins or multiple recrystallizations. These limitations not only inflate the cost of goods sold (COGS) but also introduce variability in the impurity profile, posing risks for regulatory compliance in drug development pipelines.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a copper-doped graphitic carbon nitride (Cu/g-C3N4) heterogeneous catalyst to drive the cascade cyclization. This method transforms the reaction landscape by enabling simple filtration for catalyst separation, thereby eliminating the need for complex metal removal protocols. The reaction proceeds efficiently in polar aprotic solvents like DMF at moderate temperatures ranging from 70°C to 90°C. As illustrated in the general reaction scheme below, the process couples readily available trifluoroacetimidoyl chlorides with a diverse array of amines to yield the target benzimidazole scaffolds with exceptional yields.
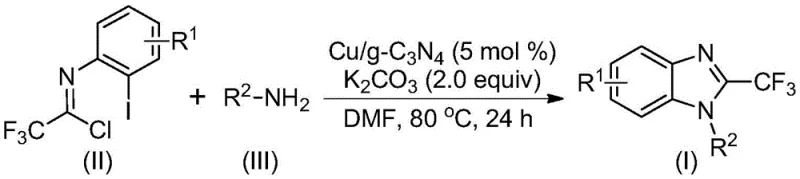
The versatility of this system is evident in its tolerance for various functional groups, allowing for the synthesis of derivatives with alkyl, aryl, and halogen substituents without compromising reaction efficiency. This flexibility is crucial for medicinal chemists exploring structure-activity relationships (SAR) who require rapid access to diverse analog libraries.
Mechanistic Insights into Cu/g-C3N4 Catalyzed Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between the heterogeneous catalyst surface and the organic substrates. Initially, the trifluoroacetimidoyl chloride undergoes a nucleophilic addition-elimination reaction with the amine to form an amidine intermediate. Subsequently, the copper species embedded within the carbon nitride matrix facilitates the insertion into the carbon-iodine bond of the aryl ring. This step is critical as it generates a reactive copper-aryl complex that promotes intramolecular cyclization. Under the basic conditions provided by potassium carbonate, hydrogen iodide is eliminated, leading to the formation of a bivalent or trivalent cyclic copper intermediate. The final step involves a reductive elimination that releases the 2-trifluoromethyl substituted benzimidazole product and regenerates the active catalytic site on the carbon nitride support.

From an impurity control perspective, the heterogeneous nature of the catalyst plays a vital role. Unlike homogeneous systems where metal leaching can lead to unpredictable side reactions or metal-complexed impurities, the solid-supported copper confines the catalytic activity to the surface. This confinement effect minimizes non-specific background reactions and ensures a cleaner crude reaction profile. Additionally, the use of mild bases like potassium carbonate prevents the degradation of sensitive functional groups that might occur under stronger alkaline conditions, further enhancing the purity of the final intermediate. The robustness of the carbon nitride support also contributes to the stability of the catalyst over multiple cycles, maintaining consistent selectivity and conversion rates.
How to Synthesize 2-Trifluoromethyl Benzimidazole Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory discovery and pilot plant operations. The procedure eliminates the need for inert atmosphere techniques often required by sensitive homogeneous catalysts, as the reaction tolerates standard Schlenk tube conditions effectively. By optimizing the molar ratios of the imidoyl chloride, amine, base, and catalyst, manufacturers can achieve near-quantitative conversions. The following guide outlines the standardized steps derived from the patent examples to ensure reproducible results.
- Combine copper-doped carbon nitride (Cu/g-C3N4), potassium carbonate, trifluoroacetimidoyl chloride, and the desired amine in an organic solvent such as DMF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the heterogeneous catalyst, mix the filtrate with silica gel, and perform column chromatography to isolate the pure 2-trifluoromethyl substituted benzimidazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this heterogeneous catalytic process offers tangible strategic benefits beyond mere chemical yield. The shift from homogeneous to heterogeneous catalysis fundamentally alters the cost structure of manufacturing complex heterocycles. By removing the dependency on expensive ligand systems and simplifying the workup procedure, the overall processing time is drastically reduced. This efficiency translates directly into lower operational expenditures and improved throughput capacity, allowing suppliers to respond more agilely to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the recyclability of the Cu/g-C3N4 catalyst. In traditional processes, the loss of precious metal catalysts into the waste stream represents a significant financial drain. Here, the solid catalyst can be recovered via simple filtration and reused, substantially lowering the raw material cost per kilogram of product. Furthermore, the elimination of rigorous metal scavenging steps reduces the consumption of auxiliary chemicals and solvents, leading to comprehensive cost optimization across the entire production lifecycle.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically trifluoroacetimidoyl chlorides and various amines, are commercially available and inexpensive commodity chemicals. This abundance ensures a stable supply chain that is less susceptible to the bottlenecks often associated with specialized reagents. The robustness of the reaction conditions also means that production is less likely to be interrupted by minor variations in utility supplies or environmental controls, guaranteeing consistent delivery schedules for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes often exacerbates waste management issues, but this method inherently mitigates such risks. The heterogeneous catalyst generates less hazardous waste compared to soluble metal salts, simplifying effluent treatment and disposal. The high atom economy of the cyclization reaction, combined with the potential for solvent recovery, aligns perfectly with modern green chemistry principles. This environmental compatibility not only reduces regulatory burdens but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its implementation. The following questions address common inquiries regarding the practical application and scope of the patented technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for process development teams.
Q: What are the advantages of using Cu/g-C3N4 over homogeneous copper catalysts?
A: The use of copper-doped carbon nitride (Cu/g-C3N4) as a heterogeneous catalyst allows for easy separation via simple filtration, enabling catalyst recycling and significantly reducing heavy metal contamination in the final pharmaceutical intermediate compared to traditional homogeneous systems.
Q: What is the substrate scope for this benzimidazole synthesis?
A: The method demonstrates broad substrate tolerance, successfully accommodating various amines including aliphatic chains (n-butyl, cyclohexyl, isopropyl) and aromatic groups (benzyl, phenyl), as well as aryl rings with electron-withdrawing or donating substituents like chloro, bromo, or trifluoromethyl groups.
Q: What are the optimal reaction conditions described in the patent?
A: The optimal conditions involve reacting the substrates in DMF solvent at 80°C for 24 hours using potassium carbonate as the base, which provides high yields (up to 99%) while maintaining mild operational parameters suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our technical team has extensively analyzed the potential of the heterogeneous catalytic route described in CN113429349A and is fully prepared to implement this advanced methodology. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from gram-scale research to industrial manufacturing. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the low metal content and high chemical purity required for GMP-grade intermediates.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to obtain specific COA data for our benzimidazole portfolio and discuss route feasibility assessments that can accelerate your timeline to market while optimizing your overall budget.
